3.8www.00271
www.00271 时间:2021-03-23 阅读:()
RESEARCHARTICLEOpenAccessEpidemiologyofrotavirusinfectionamongyoungchildrenwithacutediarrhoeainBurkinaFasoIsidoreJOBonkoungou1,2*,IdrissaSanou3,4,FabienneBon5,BenoitBenon6,SheickOCoulibaly2,4,KaisaHaukka7,AlfredSTraoré1,NicolasBarro1AbstractBackground:Inanticipationofvaccineintroduction,weassessedepidemiologyofrotavirusdiseaseamongchildrenvisitingmedicalcentreduetoacutediarrhoeainOuagadougou,BurkinaFaso.
Methods:BetweenNovember2008andFebruary2010,stoolspecimensfrom447childrenlessthan5yearsofagesufferingfromdiarrhoeaweretestedforthepresenceofrotavirusbyantigendetectionusinganimmunochromatographictest.
Sociodemographic,environmentalandclinicalfactorswereassessedduringthestudy.
Results:Rotavirusantigenwasdetectedin151(33.
8%)ofthepatients.
Mostofthecases(94.
2%)wereinchildren<24monthsofage.
Feverandvomitingwerethesymptomsmostcommonlyreportedinassociationwithrotavirusdiarrhoeaandthepatientswereoftenhospitalized.
Rotavirus-associateddiarrhoeaoccurredmostlyduringtheseasonfromDecembertoApril(dryseason).
Rotavirusinfectionwassignificantlylessfrequentinbreast-fedthanamongbottle-fedbabies.
Conclusions:TheresultsofthisstudyunderscoretheneedtocontrolrotavirusinfectionsamongyoungchildreninBurkinaFasoandmayargueadecisionontheintroductionofrotavirusvaccineinBurkinaFaso.
BackgroundRotavirusisamajorcauseofacutegastroenteritisininfantsandyoungchildrenworldwide[1].
Ithasbeenestimatedthatabout39%ofchildhooddiarrhoeahospita-lizationsarecausedbyrotavirusesandnearlyhalfamil-lionchildrendiefromrotavirusinfectionseachyear[2].
Furthermore,rotavirusmortalityisconcentratedinthedevelopingcountriesontheAsiansubcontinent,Africa,andLatinAmericawhereaccesstohealthcarefacilitiesislimited[3].
Thismayresultinasignificantdiseasebur-denandeconomiceffectofdirectmedicalcosts,lossofwork,qualityoflifeandmortality.
Inotherdiarrhoealdiseases,improvementofhygieneandsanitationmayreducetheincidence,butthesemeasuresareunlikelysuf-ficientforrotaviruscontrol.
Vaccinationistheonlycon-trolmeasurelikelytohaveasignificantimpactontheincidenceofseverelydehydratingrotavirusdisease[4].
Twonewlive-attenuatedrotavirusvaccines(RotarixandRotaTeq)havedemonstratedverygoodsafetyandeffi-cacyprofilesinlargeclinicaltrialsintheWesternindus-trializedcountriesandinLatinAmerica[5-9].
Trialsonthesevaccinesarenowinprogressinsub-SaharanAfricatoassesstheireffectivenessandefficacy.
InBurkinaFaso,veryfewdataonillnesscausedbyrotavirushavebeenpublishedandthesestudiesindi-catedthat14%ofacutediarrhoeainchildrenundertheageof5yearsisduetorotavirusinfectionbutepide-miologicaldataarestillincomplete[10].
Theobjectivesofthisstudyweretodescribe,forthefirsttime,epidemiologyofrotavirusdiseaseamongchil-drenvisitingalocalhealthcentrebecauseofacutegas-troenteritisinOuagadougouinBurkinaFasotoprovidebackgroundknowledgeonthediseasebeforevaccineintroductionandtoinformthepolicymakersontheneedfortheintroductionofnewrotavirusvaccines.
MethodsStudypopulationandspecimensThestudywasconductedatCentreMédicalavecAntenneChirugicale(CMA)duSecteur30inthecapital*Correspondence:ouindgueta@gmail.
com1LaboratoiredeBiochimieetBiologieMoléculaire,CRSBAN/UFR-SVT,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFasoFulllistofauthorinformationisavailableattheendofthearticleBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/942010Bonkoungouetal;licenseeBioMedCentralLtd.
ThisisanOpenAccessarticledistributedunderthetermsoftheCreativeCommonsAttributionLicense(http://creativecommons.
org/licenses/by/2.
0),whichpermitsunrestricteduse,distribution,andreproductioninanymedium,providedtheoriginalworkisproperlycited.
cityofOuagadougou,BurkinaFaso.
CMAdusecteur30,locatedintheBogodogodistrictisoneofthefoursec-ondaryhealthcarecentersinOuagadougouanditspae-diatricwardhasacapacityof30bedsandadmitsover2260childreneachyear.
Ouagadougouhasapopulationofnearlytwomillions,whereastheBogodogodistricthasapopulationofabout548000with81000(15%)chil-dren<5yearsofagehttp://www.
sante.
gov.
bf.
ThestudyprotocolwasapprovedbytheEthicsCommitteeofBurkinaFaso.
Parentsofallthepaediatricpatientswereinformedonthestudydetailsandtheiroralconsentwasobtainedbeforestoolspecimenandepidemiologicaldatacollectionduringthecourseoftreatment.
Writtencon-sentwasobtainedfromparentsofthecontrolgroup.
Allchildrenundertheageof5yearvisitingthepaediatricservicefortreatmentofgastroenteritisfromNovember2008toFebruary2010wereincludedinthestudy.
Diar-rhoeawasdefinedasthepassageofthreeormorelooseorwaterystoolsinthepreceding24h.
Duringthisstudy,471patientswereincludedassoonastheywereseenbyaphysicianandfreshstoolsampleswerecol-lectedandtransferredtotheMicrobiologyLaboratoryattheNationalPublicHealthLaboratory,Ouagadougou,forrotavirusandadenovirusdetection.
Controlstoolsampleswerecollectedfrom60randomlyselectedchil-drencomingtothesamehealthcentreforroutineimmunizationandnotpresentinggastroenteritissymp-toms.
Theageofthecontrolwaspairedwiththepatient'sage.
Informationregardingtheage,sex,typeofnutrition(breast-fedand/orbottle-fed),hospitalizationandclinicalsymptomssuchasfever,vomitinganddehy-dration,andthecharacteristicsofstoolwererecordedforeachchild.
Alsohygienefactorssuchassourceofdrinkingwaterofthechildwerecollectedduringthestudy.
DetectionofrotavirusinstoolsamplesAllstoolsampleswereanalyzedforgroupArotavirususingonesteprotavirusandadenovirusserotypes40/41testfordeterminationofrotavirusandadenovirusinhumanfeces(SDBiolineRota/Adeno;Standarddiag-nostics,Inc.
,Korea)followingthemanufacturer'sinstructions.
Statisticalanalysesc2testwasusedtoanalyzethedataandthepvaluelessthan0.
05wasconsideredstatisticallysignificant.
ResultsRotavirusprevalenceOutofthe471childrenwithacutediarrheainitiallyincludedinthestudy,24weresubsequentlyexcludedbecausefor19ofthemnosamplewascollectedandfor5ofthemnoepidemiologicaldatawereavailable.
Ofthe447stoolspecimensanalysed,151(33.
8%)werefoundtocontainrotavirus.
Onlyone(1.
7%)outofthe60stoolspecimenscollectedfromhealthychildrenwaspositivebyimmunochromatographictest(ICG)(p<0.
0001).
Adenoviruswasdetectedin17of447(3.
8%)stoolsam-plesandmixedinfectionswithbothrotavirusandade-noviruswereobservedin11(2.
5%)stoolsamples.
AgeandsexdistributionofpatientswithrotavirusinfectionTheagedistributionofchildrenwithrotavirusisshowninFigure1.
Mostcasesofrotavirusinfection(94.
2%)occurredamongchildrenlessthan2yearsofage.
Thehighestincidencewasobservedinchildrenbetween6and11monthsofage.
Themedianageforrotavirusinfectionwas8months.
Thereweremoremales(52.
8%)thanfemales,butthesexratioamongtherotavirusdiar-rheapatientswasnotsignificant(p=0.
1).
SeasonaldistributionofrotavirusinfectionsDuringthewholeperiodofsurveillance,theincidenceofrotavirusinfectionsvariedsignificantlyaccordingtoseasons(warmandcold)(p=0.
0001).
Rotavirusinfec-tionsoccurredmostlyduringtheseasonfromDecembertoApril,correspondingtothedryseasonandrelativelycoldperiod(Figure2).
HospitalizationandclinicalpresentationofrotavirusinfectionOfthe217outpatientchildren,48(22.
1%)wereinfectedandofthe230inpatientchildren,103(44.
8%)wereinfectedwithrotavirus.
Therotavirusinfectionpreva-lencewassignificantlyhigheramonghospitalizedchil-dren(p=0.
0001)illustratingasignificantrelationshipbetweenrateofhospitalization-severityofillnessanddiarrhoeaassociatedwithrotavirusinfection.
Inaddi-tion,feverwasthesymptommostcommonlyreportedinassociationwithrotavirusdiarrhoea(82.
1%),followedbyvomiting(72.
8%)anddehydration(48.
3%)(Table1).
NutritionanddrinkingwaterAmongtheinfants≤9monthsofage,whohadrota-virusdiarrhoeaandforwhomthemethodoffeedingwasrecorded,55outof154werebreast-fed(35.
7%)and32outof61werebottle-fed(52.
5%)(p=0.
01).
Analysisofhygienefactorssuchasthesourceofdrinkingwatershowedthatchildrendrinkingmunicipalwaterweremoreaffectedbyrotavirusdiarrhoeathanchildrendrinkingmineralwatersoldinbottles(p=0.
0001)(Table1).
DiscussionThiswasthefirststudyinBurkinaFasotoinvestigatetheprevalence,clinicalcharacteristicandriskfactorsofBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page2of6rotavirusgastroenteritisamongchildren.
Regardlessourresults,rotavirusdiarrhoeaappearstobeamajorpublichealthproblemforchildreninBurkinaFaso,asintheotherdevelopingcountries.
Ourresultsshowthatasig-nificantproportionofacutediarrhoeaisduetorotavirus(33.
8%)androtavirusmayberesponsibleforalmostone-half(44.
8%)ofallhospitalizationsfordiarrheainchildren<5yearsofageinBurkinaFaso.
ThedetectedprevalenceappearstobesimilartothosereportedfromotherWestAfricancountries,whichrangedfrom33%to39%[11-13].
Inaddition,acumulativeexperiencefrom15AfricancountriessuggestedthatrotavirusisthemostimportantcauseofseverediarrhoeainAfricanchildren[14].
Asobservedintheotherpartsoftheworld,thebur-denofrotavirusdiseaseispredominantlybornebychil-drenlessthan2yearsofage[15]withahighincidenceamongchildren6-11monthsofage.
Thiscanbeexplainedbytheprotectiveeffectofmaternalantibodiesin<6monthsold,andthedevelopmentofnaturalimmunityafterrepeatedinfectionsinchildrenover2yearsofage[16,17].
OurresultsshowedthatrotavirusoccurredmostlyduringtheseasonfromDecembertoApril,correspond-ingtothedryseasonandrelativelycoldperiod,ashasbeenreportedfromNorthernGhananearBurkinaFaso[13]andGuinea-Bissau[11].
SomestudiesconductedinotherAfricancountriesindicatedthatrotavirusinfec-tionsarepresentthroughouttheyear,butwithmuchhigherprevalenceinacertainperiodofayear[14].
Comparisonoftheclinicalcharacteristicsandseverityoftheacutegastroenteritisamongtherotavirus-positiveandrotavirus-negativepatientsindicatedthatvomiting,feveranddehydrationweremorefrequentlyobservedamongdiarrhealchildrenwithrotavirusthanamongthosewithoutrotavirusinfection,asreportedintheothercountries[11,18].
Ourconfirmationofapreviousobservationmadeintheotherpartsoftheworldthatduringthefirstyearoflifebreastfeedingisassociatedwithalowerincidenceofrotavirusdiarrhoealepisodesaddstothemultitudeofbenefitsthathavebeenassociatedwithbreastfeeding[19,20].
Inaddition,ithasbeenshownthatevenifthebreast-fedinfantsgetinfectedwithrotavirus,amilderdiseaseoccursandhospitalizationrateissignificantlylower[21].
Anotherimportantissue,whichwasshowninthisstudy,isasignificantassociationbetweenrotavirusdiar-rheaandmunicipaldrinkingwater.
ThismaybeduetothepossiblecontaminationofmunicipalwaterforDetectionrate(%)Age(months)6.
0%47.
0%23.
2%0.
6%4.
0%18%0.
6%0.
6%0.
0%0.
0%Figure1Proportionofrotavirusinfectionsbyagegroupsamongthe447childrensufferingofgastroenteritis,betweenNovember2008toFebruary2010.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page3of6Figure2Seasonalityoftheacutegastroenteritisandtherotavirusinfectionsamongthe447childrensufferingofgastroenteritis,betweenNovember2008toFebruary2010.
Table1ClinicalandepidemiologicalfeaturesofchildrenwithandwithoutrotavirusdetectedinthediarrhealstoolsampleEpidemiologicalandclinicalcharacteristicsRotavirusDiarrheaNonRotavirusDiarrheap(N=151)(N=296)SexMale71(47.
0%)165(55.
7%)p=0.
1Female80(53.
0%)131(44.
8%)Meanage(months)10.
916.
6PatientstatusInpatient103(44.
8%)127(55.
2%)p=0.
0001Outpatient48(22.
1%)169(77.
9%)SymptomFever124(82.
1%)161(54.
4%)p=0.
0001Vomiting110(72.
8%)80(27.
0%)p=0.
0001Dehydration73(48.
3%)54(18.
2%)p=0.
0001Season*Warm5(2.
9%)168(97.
1%)p=0,0001Cold146(53.
3%)128(46.
7%)Breastfeeding**Yes59(59.
0%)105(75.
5%)p=0.
010No41(41.
0%)34(24.
5%)DrinkingwatersourceMunicipalsupply108(71.
5%)142(48.
0%)p=0.
0001Bottledwater43(28.
5%)154(52.
0%)*Percentageofrotavirusdiarrheaversusnon-rotavirusdiarrheaforbothseasons.
**Percentageofrotavirusdiarrheaversusnon-rotavirusdiarrheaaccordingtomethodoffeedingamonginfants≤9monthsofage.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page4of6humanconsumptionorininter-humancontaminationwhichdrinkingwatermaybeapotentialriskofrota-virustransmission.
Rotavirushasbeendescribedasacausativeagentinseveralwaterborneoutbreaksintheindustrializedcountries[22-25],indicatinggoodsurvivalofrotavirusinwater.
InBurkinaFaso,theevaluationofdrinkingwaterqualitydoesnotrequiretestingforrota-virusbutourresultsshowtheimportanceofincludingroutinevirologicalanalysisofdrinkingwaterduringrotavirusseason.
ConclusionsInconclusion,thisstudyprovidesinformationontheepidemiologyandtheextentofrotavirusinfectionsinBurkinaFaso.
Ourresultsindicatethatgastroenteritiscausedbyrotavirusinthecountryisanimportanthealthproblem,particularlyamongchildrenlessthan2yearsofageandduringthecoldseason.
ThesedatawillbeusefulformakinganinformeddecisionabouttheintroductionofrotavirusvaccineinBurkinaFasoandwillprovideabaselineagainstwhichtheimpactofthevaccineintroductioncanbemeasuredinthefuture.
AcknowledgementsThisstudywassupportedbytheLaboratoryofBiochemstryandMolecularBiologyofCRSBAN/UFR-SVT(UniversityofOuagadougou)andtheNationalPublicHealthLaboratoryinBurkinaFaso.
WethankthestaffofCentreMédicalduSecteur30deOuagadougou,allthechildrenandtheirparentswhoparticipatedinthisresearch.
WeexpressourgratitudetoDrFidèleTiendrébéogoforthetechnicalassistanceinthestatisticalanalysis.
Authordetails1LaboratoiredeBiochimieetBiologieMoléculaire,CRSBAN/UFR-SVT,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFaso.
2LaboratoireNationaldeSantéPublique,09BP24,Ouagadougou,BurkinaFaso.
3LaboratoiredeBactériologieetVirologieduCHU-YalgadoOuédraogo,03BP7022Ouagadougou,BurkinaFaso.
4UFRSciencesdelaSanté,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFaso.
5LaboratoireInteractionsMuqueuses-AgentsTransmissibles-UFRMédecine,21079Dijon,France.
6Servicedepédiatrie,CentreMédicalduSecteur30deOuagadougou,Ouagadougou,BurkinaFaso.
7DepartmentofInfectiousDiseaseSurveillanceandControl,BacteriologyUnit,NationalInstituteforHealthandWelfare,P.
O.
Box30,00271Helsinki,Finland.
Authors'contributionsIJOB,ISandNBconceivedthestudy;BBandIJOBwereinchargeofrecruitment,examination,treatmentandfollow-upofpatients,controlsandundertooklaboratoryanalysis;IJOB,ISandNBanalyzedthedataandpreparedthemanuscript;NB,IS,FB,SOC,KHandASTsecuredthestudyexecutionandprovidedideasandcommentsduringmanuscriptpreparation.
Allauthorshavereadandapprovedthefinalmanuscript.
CompetinginterestsTheauthorsdeclarethattheyhavenocompetinginterests.
Received:10July2010Accepted:20December2010Published:20December2010References1.
DesselbergerU,Wolleswinkel-vandenBoschJ,MrukowiczJ,RodrigoC,GiaquintoC,VesikariT:RotavirustypesinEuropeandtheirsignificancefor638vaccinations.
PediatrInfectDisJ2006,25(1):S30-41.
2.
ParasharUD,HummelmanEG,BreseeJS,MillerMA,GlassRI:Globalillnessanddeathscausedbyrotavirusdiseaseinchildren.
EmergInfectDis2003,9:565-5772.
3.
PhuaKB,EmmanuelSC,GohP,QuakSH,LeeBW,HanHH,WardRL,BernsteinDI,DeVosB,BockHL:Arotavirusvaccineforinfants:theAsianexperience.
AnnAcadMedSingapore2006,35(1):38-47.
4.
DennehyPH:Rotavirusvaccines:anoverview.
ClinMicrobiolRev2008,21:198-208.
5.
Ruiz-PalaciosGM,Pérez-SchaelI,VelázquezFR,AbateH,BreuerT,ClemensSC,CheuvartB,EspinozaF,GillardP,InnisBL,CervantesY,LinharesAC,LópezP,Macías-ParraM,Ortega-BarríaE,RichardsonV,Rivera-MedinaDM,RiveraL,SalinasB,Pavía-RuzN,SalmerónJ,RüttimannR,TinocoJC,RubioP,NuezE,GuerreroML,YarzábalJP,DamasoS,TornieporthN,Sáez-LlorensX,VergaraRF,VesikariT,BouckenoogheA,ClemensR,DeVosB,O'RyanM,HumanRotavirusVaccineStudyGroup:Safetyandefficacyofanattenuatedvaccineagainstsevererotavirusgastroenteritis.
NEnglJMed2006,354:11-22.
6.
VesikariT,MatsonDO,DennehyP,VanDammeP,SantoshamM,RodriguezZ,DallasMJ,HeyseJF,GoveiaMG,BlackSB,ShinefieldHR,ChristieCD,YlitaloS,ItzlerRF,CoiaML,OnoratoMT,AdeyiBA,MarshallGS,GotheforsL,CampensD,KarvonenA,WattJP,O'BrienKL,DiNubileMJ,ClarkHF,BoslegoJW,OffitPA,HeatonPM:Safetyandefficacyofpentavalenthuman-bovine(WC3)reassortantrotavirusvaccineinpreventingrotavirusgastroenteritisandreducingassociatedhealthcareresourceutilization.
NEnglJMed2006,354:23-33.
7.
VesikariT,KarvonenA,PrymulaR,SchusterV,TejedorJC,CohenR,MeuriceF,HanHH,DamasoS,BouckenoogheA:Efficacyofhumanrotavirusvaccineagainstrotavirusgastroenteritisduringthefirst2yearsoflifeinEuropeaninfants:randomised,double-blindcontrolledstudy.
Lancet2007,370(9601):1757-1763.
8.
BlockSL,VesikariT,GoveiaMG,RiversSB,AdeyiBA,DallasMJ,BauderJ,BoslegoJW,HeatonPM:Efficacy:immunogenicity,andsafetyofthepentavalenthuman-bovine(WC3)reassortantRotavirusVaccineattheendofshelflife.
Pediatrics2007,119(1):11-18.
9.
ParasharUD,GibsonCJ,BresseJS,GlassRI:Rotavirusandseverechildhooddiarrhea.
EmergInfectDis2006,12(2):304-306.
10.
BonkoungouOJI,SanouI,BarroN,ToéL,SanfoMS,Ouédraogo/TraoréR,TraoreAS:ElectrophoretypescharacterizationofhumanRotavirusintwopediatricsservices,Ouagadougou,BurkinaFaso.
JMedSci2008,8:371-377.
11.
RodriguesA,deCarvalhoM,MonteiroS,MikkelsonSC,AabyP,MolbakkK,FischerTK:HospitalsurveillanceofrotavirusinfectionandnosocomialtransmissionofrotavirusdiseaseamongchildreninGuinea-Bissau.
PediatrInfectDisJ2007,26(3):233-237.
12.
deVilliersFPR,SawyerrTN,deVilliersGK:TheincidenceandclinicalpresentationofinfantilerotavirusdiarrhoeainSierraLeone.
SAfrMedJ2009,99(4):249-252.
13.
BinkaFN,AntoFK,OduroAR,AwiniEA,NazzarAK,ArmahGE,AsmahRH,HallAJ,CuttsF,AlexanderN,BrownD,GreenJ,GrayJ,Iturriza-GómaraM,NavrongoRotavirusResearchGroup:IncidenceandriskfactorsofpaediatricrotavirusdiarrhoeainNorthernGhana.
TropMedIntHealth2003,8(9):840-846.
14.
CunliffeNA,KilgorePE,BreseeJS,SteeleAD,LuoN,HartCA,GlassRI:EpidemiologyofrotavirusdiarrheainAfrica:areviewtoassesstheneedforrotavirusimmunization.
BullWHO1998,76(5):525-37.
15.
DeZoysaI,FeachemRG:Interventionsforthecontrolofdiarrhoealdiseaseamongyoungchildren:rotavirusandcholeraimmunization.
BullWHO1985,63(3):569-583.
16.
OffitPA:Hostfactorsassociatedwithprotectionagainstrotavirusdisease:theskiesareclearing.
JInfectDis1996,174(Suppl1):S59-S64.
17.
JiangB,GentschJR,GlassRI:Theroleofserumantibodiesintheprotectionagainstrotavirusdisease:anoverview.
ClinInfectDis2002,34(10):1351-1361.
18.
ZarnaniAH,ModarresS,JadaliF,SabahiF,MoazzeniSM,VazirianF:RoleofrotavirusesinchildrenwithacutediarrheainTehran,Iran.
JClinVirology2004,29(3):189-193.
19.
NaficyAB,Abu-ElyazeedR,HolmesJL,RaoRM,SavarinoJS,KimY,WierzbaTF,PeruskiL,LeeYJ,GentschJR,GlassRI,ClemensJD:EpidemiologyofRotavirusDiarrheainEgyptianChildrenandImplicationsforDiseaseControl.
AmJEpidemiol1999,150(7):770-777.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page5of620.
ClemensJ,RaoM,AhmedF,WardR,HudaS,ChakrabortyJ,YunusM,KhanMR,AliM,KayB,vanLoonF,SackD:Breast-feedingandtheriskoflife-threateningrotavirusdiarrhea:preventionorpostponementPediatrics1993,92(5):680-685.
21.
KapikianAZ,YasutakaH,ChanokRM:Rotaviruses.
Field'sVirology.
4edition.
Philadelphia:LippincottWilliamsandWilkinsCo;2001,1787-834.
22.
KukkulaM,ArstilaP,KlossnerML,MaunulaL,BonsdorffCH,JaatinenP:Waterborneoutbreakofviralgastroenteritis.
ScandJInfectDis1997,29(4):415-418.
23.
VillenaC,GabrieliR,PintoRM,GuixS,DoniaD,BuonomoE,PalombiL,CenkoF,BinoS,BoschA,DiviziaM:AlargeinfantilegastroenteritisoutbreakinAlbaniacausedbymultipleemergingrotavirusgenotypes.
EpidemiolInfect2003,131(3):1105-1110.
24.
BorchardtMA,HaasNL,HuntRJ:Vulnerabilityofdrinking-waterwellsinLaCrosse,Wisconsin,toenteric-viruscontaminationfromsurfacewatercontributions.
ApplEnvironMicrobiol2004,70(10):5937-5946.
25.
MartinelliD,PratoR,ChironnaM,SallustioA,CaputiG,ConversanoM,CiofiDegliAttiM,D'AnconaFP,GerminarioCA,QuartoM:LargeoutbreakofviralgastroenteritiscausedbycontaminateddrinkingwaterinApulia,Italy,May-October2006.
EuroSurveill2007,12(16):Article1.
Pre-publicationhistoryThepre-publicationhistoryforthispapercanbeaccessedhere:http://www.
biomedcentral.
com/1471-2431/10/94/prepubdoi:10.
1186/1471-2431-10-94Citethisarticleas:Bonkoungouetal.
:EpidemiologyofrotavirusinfectionamongyoungchildrenwithacutediarrhoeainBurkinaFaso.
BMCPediatrics201010:94.
SubmityournextmanuscripttoBioMedCentralandtakefulladvantageof:ConvenientonlinesubmissionThoroughpeerreviewNospaceconstraintsorcolorgurechargesImmediatepublicationonacceptanceInclusioninPubMed,CAS,ScopusandGoogleScholarResearchwhichisfreelyavailableforredistributionSubmityourmanuscriptatwww.
biomedcentral.
com/submitBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page6of6
Methods:BetweenNovember2008andFebruary2010,stoolspecimensfrom447childrenlessthan5yearsofagesufferingfromdiarrhoeaweretestedforthepresenceofrotavirusbyantigendetectionusinganimmunochromatographictest.
Sociodemographic,environmentalandclinicalfactorswereassessedduringthestudy.
Results:Rotavirusantigenwasdetectedin151(33.
8%)ofthepatients.
Mostofthecases(94.
2%)wereinchildren<24monthsofage.
Feverandvomitingwerethesymptomsmostcommonlyreportedinassociationwithrotavirusdiarrhoeaandthepatientswereoftenhospitalized.
Rotavirus-associateddiarrhoeaoccurredmostlyduringtheseasonfromDecembertoApril(dryseason).
Rotavirusinfectionwassignificantlylessfrequentinbreast-fedthanamongbottle-fedbabies.
Conclusions:TheresultsofthisstudyunderscoretheneedtocontrolrotavirusinfectionsamongyoungchildreninBurkinaFasoandmayargueadecisionontheintroductionofrotavirusvaccineinBurkinaFaso.
BackgroundRotavirusisamajorcauseofacutegastroenteritisininfantsandyoungchildrenworldwide[1].
Ithasbeenestimatedthatabout39%ofchildhooddiarrhoeahospita-lizationsarecausedbyrotavirusesandnearlyhalfamil-lionchildrendiefromrotavirusinfectionseachyear[2].
Furthermore,rotavirusmortalityisconcentratedinthedevelopingcountriesontheAsiansubcontinent,Africa,andLatinAmericawhereaccesstohealthcarefacilitiesislimited[3].
Thismayresultinasignificantdiseasebur-denandeconomiceffectofdirectmedicalcosts,lossofwork,qualityoflifeandmortality.
Inotherdiarrhoealdiseases,improvementofhygieneandsanitationmayreducetheincidence,butthesemeasuresareunlikelysuf-ficientforrotaviruscontrol.
Vaccinationistheonlycon-trolmeasurelikelytohaveasignificantimpactontheincidenceofseverelydehydratingrotavirusdisease[4].
Twonewlive-attenuatedrotavirusvaccines(RotarixandRotaTeq)havedemonstratedverygoodsafetyandeffi-cacyprofilesinlargeclinicaltrialsintheWesternindus-trializedcountriesandinLatinAmerica[5-9].
Trialsonthesevaccinesarenowinprogressinsub-SaharanAfricatoassesstheireffectivenessandefficacy.
InBurkinaFaso,veryfewdataonillnesscausedbyrotavirushavebeenpublishedandthesestudiesindi-catedthat14%ofacutediarrhoeainchildrenundertheageof5yearsisduetorotavirusinfectionbutepide-miologicaldataarestillincomplete[10].
Theobjectivesofthisstudyweretodescribe,forthefirsttime,epidemiologyofrotavirusdiseaseamongchil-drenvisitingalocalhealthcentrebecauseofacutegas-troenteritisinOuagadougouinBurkinaFasotoprovidebackgroundknowledgeonthediseasebeforevaccineintroductionandtoinformthepolicymakersontheneedfortheintroductionofnewrotavirusvaccines.
MethodsStudypopulationandspecimensThestudywasconductedatCentreMédicalavecAntenneChirugicale(CMA)duSecteur30inthecapital*Correspondence:ouindgueta@gmail.
com1LaboratoiredeBiochimieetBiologieMoléculaire,CRSBAN/UFR-SVT,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFasoFulllistofauthorinformationisavailableattheendofthearticleBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/942010Bonkoungouetal;licenseeBioMedCentralLtd.
ThisisanOpenAccessarticledistributedunderthetermsoftheCreativeCommonsAttributionLicense(http://creativecommons.
org/licenses/by/2.
0),whichpermitsunrestricteduse,distribution,andreproductioninanymedium,providedtheoriginalworkisproperlycited.
cityofOuagadougou,BurkinaFaso.
CMAdusecteur30,locatedintheBogodogodistrictisoneofthefoursec-ondaryhealthcarecentersinOuagadougouanditspae-diatricwardhasacapacityof30bedsandadmitsover2260childreneachyear.
Ouagadougouhasapopulationofnearlytwomillions,whereastheBogodogodistricthasapopulationofabout548000with81000(15%)chil-dren<5yearsofagehttp://www.
sante.
gov.
bf.
ThestudyprotocolwasapprovedbytheEthicsCommitteeofBurkinaFaso.
Parentsofallthepaediatricpatientswereinformedonthestudydetailsandtheiroralconsentwasobtainedbeforestoolspecimenandepidemiologicaldatacollectionduringthecourseoftreatment.
Writtencon-sentwasobtainedfromparentsofthecontrolgroup.
Allchildrenundertheageof5yearvisitingthepaediatricservicefortreatmentofgastroenteritisfromNovember2008toFebruary2010wereincludedinthestudy.
Diar-rhoeawasdefinedasthepassageofthreeormorelooseorwaterystoolsinthepreceding24h.
Duringthisstudy,471patientswereincludedassoonastheywereseenbyaphysicianandfreshstoolsampleswerecol-lectedandtransferredtotheMicrobiologyLaboratoryattheNationalPublicHealthLaboratory,Ouagadougou,forrotavirusandadenovirusdetection.
Controlstoolsampleswerecollectedfrom60randomlyselectedchil-drencomingtothesamehealthcentreforroutineimmunizationandnotpresentinggastroenteritissymp-toms.
Theageofthecontrolwaspairedwiththepatient'sage.
Informationregardingtheage,sex,typeofnutrition(breast-fedand/orbottle-fed),hospitalizationandclinicalsymptomssuchasfever,vomitinganddehy-dration,andthecharacteristicsofstoolwererecordedforeachchild.
Alsohygienefactorssuchassourceofdrinkingwaterofthechildwerecollectedduringthestudy.
DetectionofrotavirusinstoolsamplesAllstoolsampleswereanalyzedforgroupArotavirususingonesteprotavirusandadenovirusserotypes40/41testfordeterminationofrotavirusandadenovirusinhumanfeces(SDBiolineRota/Adeno;Standarddiag-nostics,Inc.
,Korea)followingthemanufacturer'sinstructions.
Statisticalanalysesc2testwasusedtoanalyzethedataandthepvaluelessthan0.
05wasconsideredstatisticallysignificant.
ResultsRotavirusprevalenceOutofthe471childrenwithacutediarrheainitiallyincludedinthestudy,24weresubsequentlyexcludedbecausefor19ofthemnosamplewascollectedandfor5ofthemnoepidemiologicaldatawereavailable.
Ofthe447stoolspecimensanalysed,151(33.
8%)werefoundtocontainrotavirus.
Onlyone(1.
7%)outofthe60stoolspecimenscollectedfromhealthychildrenwaspositivebyimmunochromatographictest(ICG)(p<0.
0001).
Adenoviruswasdetectedin17of447(3.
8%)stoolsam-plesandmixedinfectionswithbothrotavirusandade-noviruswereobservedin11(2.
5%)stoolsamples.
AgeandsexdistributionofpatientswithrotavirusinfectionTheagedistributionofchildrenwithrotavirusisshowninFigure1.
Mostcasesofrotavirusinfection(94.
2%)occurredamongchildrenlessthan2yearsofage.
Thehighestincidencewasobservedinchildrenbetween6and11monthsofage.
Themedianageforrotavirusinfectionwas8months.
Thereweremoremales(52.
8%)thanfemales,butthesexratioamongtherotavirusdiar-rheapatientswasnotsignificant(p=0.
1).
SeasonaldistributionofrotavirusinfectionsDuringthewholeperiodofsurveillance,theincidenceofrotavirusinfectionsvariedsignificantlyaccordingtoseasons(warmandcold)(p=0.
0001).
Rotavirusinfec-tionsoccurredmostlyduringtheseasonfromDecembertoApril,correspondingtothedryseasonandrelativelycoldperiod(Figure2).
HospitalizationandclinicalpresentationofrotavirusinfectionOfthe217outpatientchildren,48(22.
1%)wereinfectedandofthe230inpatientchildren,103(44.
8%)wereinfectedwithrotavirus.
Therotavirusinfectionpreva-lencewassignificantlyhigheramonghospitalizedchil-dren(p=0.
0001)illustratingasignificantrelationshipbetweenrateofhospitalization-severityofillnessanddiarrhoeaassociatedwithrotavirusinfection.
Inaddi-tion,feverwasthesymptommostcommonlyreportedinassociationwithrotavirusdiarrhoea(82.
1%),followedbyvomiting(72.
8%)anddehydration(48.
3%)(Table1).
NutritionanddrinkingwaterAmongtheinfants≤9monthsofage,whohadrota-virusdiarrhoeaandforwhomthemethodoffeedingwasrecorded,55outof154werebreast-fed(35.
7%)and32outof61werebottle-fed(52.
5%)(p=0.
01).
Analysisofhygienefactorssuchasthesourceofdrinkingwatershowedthatchildrendrinkingmunicipalwaterweremoreaffectedbyrotavirusdiarrhoeathanchildrendrinkingmineralwatersoldinbottles(p=0.
0001)(Table1).
DiscussionThiswasthefirststudyinBurkinaFasotoinvestigatetheprevalence,clinicalcharacteristicandriskfactorsofBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page2of6rotavirusgastroenteritisamongchildren.
Regardlessourresults,rotavirusdiarrhoeaappearstobeamajorpublichealthproblemforchildreninBurkinaFaso,asintheotherdevelopingcountries.
Ourresultsshowthatasig-nificantproportionofacutediarrhoeaisduetorotavirus(33.
8%)androtavirusmayberesponsibleforalmostone-half(44.
8%)ofallhospitalizationsfordiarrheainchildren<5yearsofageinBurkinaFaso.
ThedetectedprevalenceappearstobesimilartothosereportedfromotherWestAfricancountries,whichrangedfrom33%to39%[11-13].
Inaddition,acumulativeexperiencefrom15AfricancountriessuggestedthatrotavirusisthemostimportantcauseofseverediarrhoeainAfricanchildren[14].
Asobservedintheotherpartsoftheworld,thebur-denofrotavirusdiseaseispredominantlybornebychil-drenlessthan2yearsofage[15]withahighincidenceamongchildren6-11monthsofage.
Thiscanbeexplainedbytheprotectiveeffectofmaternalantibodiesin<6monthsold,andthedevelopmentofnaturalimmunityafterrepeatedinfectionsinchildrenover2yearsofage[16,17].
OurresultsshowedthatrotavirusoccurredmostlyduringtheseasonfromDecembertoApril,correspond-ingtothedryseasonandrelativelycoldperiod,ashasbeenreportedfromNorthernGhananearBurkinaFaso[13]andGuinea-Bissau[11].
SomestudiesconductedinotherAfricancountriesindicatedthatrotavirusinfec-tionsarepresentthroughouttheyear,butwithmuchhigherprevalenceinacertainperiodofayear[14].
Comparisonoftheclinicalcharacteristicsandseverityoftheacutegastroenteritisamongtherotavirus-positiveandrotavirus-negativepatientsindicatedthatvomiting,feveranddehydrationweremorefrequentlyobservedamongdiarrhealchildrenwithrotavirusthanamongthosewithoutrotavirusinfection,asreportedintheothercountries[11,18].
Ourconfirmationofapreviousobservationmadeintheotherpartsoftheworldthatduringthefirstyearoflifebreastfeedingisassociatedwithalowerincidenceofrotavirusdiarrhoealepisodesaddstothemultitudeofbenefitsthathavebeenassociatedwithbreastfeeding[19,20].
Inaddition,ithasbeenshownthatevenifthebreast-fedinfantsgetinfectedwithrotavirus,amilderdiseaseoccursandhospitalizationrateissignificantlylower[21].
Anotherimportantissue,whichwasshowninthisstudy,isasignificantassociationbetweenrotavirusdiar-rheaandmunicipaldrinkingwater.
ThismaybeduetothepossiblecontaminationofmunicipalwaterforDetectionrate(%)Age(months)6.
0%47.
0%23.
2%0.
6%4.
0%18%0.
6%0.
6%0.
0%0.
0%Figure1Proportionofrotavirusinfectionsbyagegroupsamongthe447childrensufferingofgastroenteritis,betweenNovember2008toFebruary2010.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page3of6Figure2Seasonalityoftheacutegastroenteritisandtherotavirusinfectionsamongthe447childrensufferingofgastroenteritis,betweenNovember2008toFebruary2010.
Table1ClinicalandepidemiologicalfeaturesofchildrenwithandwithoutrotavirusdetectedinthediarrhealstoolsampleEpidemiologicalandclinicalcharacteristicsRotavirusDiarrheaNonRotavirusDiarrheap(N=151)(N=296)SexMale71(47.
0%)165(55.
7%)p=0.
1Female80(53.
0%)131(44.
8%)Meanage(months)10.
916.
6PatientstatusInpatient103(44.
8%)127(55.
2%)p=0.
0001Outpatient48(22.
1%)169(77.
9%)SymptomFever124(82.
1%)161(54.
4%)p=0.
0001Vomiting110(72.
8%)80(27.
0%)p=0.
0001Dehydration73(48.
3%)54(18.
2%)p=0.
0001Season*Warm5(2.
9%)168(97.
1%)p=0,0001Cold146(53.
3%)128(46.
7%)Breastfeeding**Yes59(59.
0%)105(75.
5%)p=0.
010No41(41.
0%)34(24.
5%)DrinkingwatersourceMunicipalsupply108(71.
5%)142(48.
0%)p=0.
0001Bottledwater43(28.
5%)154(52.
0%)*Percentageofrotavirusdiarrheaversusnon-rotavirusdiarrheaforbothseasons.
**Percentageofrotavirusdiarrheaversusnon-rotavirusdiarrheaaccordingtomethodoffeedingamonginfants≤9monthsofage.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page4of6humanconsumptionorininter-humancontaminationwhichdrinkingwatermaybeapotentialriskofrota-virustransmission.
Rotavirushasbeendescribedasacausativeagentinseveralwaterborneoutbreaksintheindustrializedcountries[22-25],indicatinggoodsurvivalofrotavirusinwater.
InBurkinaFaso,theevaluationofdrinkingwaterqualitydoesnotrequiretestingforrota-virusbutourresultsshowtheimportanceofincludingroutinevirologicalanalysisofdrinkingwaterduringrotavirusseason.
ConclusionsInconclusion,thisstudyprovidesinformationontheepidemiologyandtheextentofrotavirusinfectionsinBurkinaFaso.
Ourresultsindicatethatgastroenteritiscausedbyrotavirusinthecountryisanimportanthealthproblem,particularlyamongchildrenlessthan2yearsofageandduringthecoldseason.
ThesedatawillbeusefulformakinganinformeddecisionabouttheintroductionofrotavirusvaccineinBurkinaFasoandwillprovideabaselineagainstwhichtheimpactofthevaccineintroductioncanbemeasuredinthefuture.
AcknowledgementsThisstudywassupportedbytheLaboratoryofBiochemstryandMolecularBiologyofCRSBAN/UFR-SVT(UniversityofOuagadougou)andtheNationalPublicHealthLaboratoryinBurkinaFaso.
WethankthestaffofCentreMédicalduSecteur30deOuagadougou,allthechildrenandtheirparentswhoparticipatedinthisresearch.
WeexpressourgratitudetoDrFidèleTiendrébéogoforthetechnicalassistanceinthestatisticalanalysis.
Authordetails1LaboratoiredeBiochimieetBiologieMoléculaire,CRSBAN/UFR-SVT,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFaso.
2LaboratoireNationaldeSantéPublique,09BP24,Ouagadougou,BurkinaFaso.
3LaboratoiredeBactériologieetVirologieduCHU-YalgadoOuédraogo,03BP7022Ouagadougou,BurkinaFaso.
4UFRSciencesdelaSanté,UniversitédeOuagadougou,03BP7021,Ouagadougou,BurkinaFaso.
5LaboratoireInteractionsMuqueuses-AgentsTransmissibles-UFRMédecine,21079Dijon,France.
6Servicedepédiatrie,CentreMédicalduSecteur30deOuagadougou,Ouagadougou,BurkinaFaso.
7DepartmentofInfectiousDiseaseSurveillanceandControl,BacteriologyUnit,NationalInstituteforHealthandWelfare,P.
O.
Box30,00271Helsinki,Finland.
Authors'contributionsIJOB,ISandNBconceivedthestudy;BBandIJOBwereinchargeofrecruitment,examination,treatmentandfollow-upofpatients,controlsandundertooklaboratoryanalysis;IJOB,ISandNBanalyzedthedataandpreparedthemanuscript;NB,IS,FB,SOC,KHandASTsecuredthestudyexecutionandprovidedideasandcommentsduringmanuscriptpreparation.
Allauthorshavereadandapprovedthefinalmanuscript.
CompetinginterestsTheauthorsdeclarethattheyhavenocompetinginterests.
Received:10July2010Accepted:20December2010Published:20December2010References1.
DesselbergerU,Wolleswinkel-vandenBoschJ,MrukowiczJ,RodrigoC,GiaquintoC,VesikariT:RotavirustypesinEuropeandtheirsignificancefor638vaccinations.
PediatrInfectDisJ2006,25(1):S30-41.
2.
ParasharUD,HummelmanEG,BreseeJS,MillerMA,GlassRI:Globalillnessanddeathscausedbyrotavirusdiseaseinchildren.
EmergInfectDis2003,9:565-5772.
3.
PhuaKB,EmmanuelSC,GohP,QuakSH,LeeBW,HanHH,WardRL,BernsteinDI,DeVosB,BockHL:Arotavirusvaccineforinfants:theAsianexperience.
AnnAcadMedSingapore2006,35(1):38-47.
4.
DennehyPH:Rotavirusvaccines:anoverview.
ClinMicrobiolRev2008,21:198-208.
5.
Ruiz-PalaciosGM,Pérez-SchaelI,VelázquezFR,AbateH,BreuerT,ClemensSC,CheuvartB,EspinozaF,GillardP,InnisBL,CervantesY,LinharesAC,LópezP,Macías-ParraM,Ortega-BarríaE,RichardsonV,Rivera-MedinaDM,RiveraL,SalinasB,Pavía-RuzN,SalmerónJ,RüttimannR,TinocoJC,RubioP,NuezE,GuerreroML,YarzábalJP,DamasoS,TornieporthN,Sáez-LlorensX,VergaraRF,VesikariT,BouckenoogheA,ClemensR,DeVosB,O'RyanM,HumanRotavirusVaccineStudyGroup:Safetyandefficacyofanattenuatedvaccineagainstsevererotavirusgastroenteritis.
NEnglJMed2006,354:11-22.
6.
VesikariT,MatsonDO,DennehyP,VanDammeP,SantoshamM,RodriguezZ,DallasMJ,HeyseJF,GoveiaMG,BlackSB,ShinefieldHR,ChristieCD,YlitaloS,ItzlerRF,CoiaML,OnoratoMT,AdeyiBA,MarshallGS,GotheforsL,CampensD,KarvonenA,WattJP,O'BrienKL,DiNubileMJ,ClarkHF,BoslegoJW,OffitPA,HeatonPM:Safetyandefficacyofpentavalenthuman-bovine(WC3)reassortantrotavirusvaccineinpreventingrotavirusgastroenteritisandreducingassociatedhealthcareresourceutilization.
NEnglJMed2006,354:23-33.
7.
VesikariT,KarvonenA,PrymulaR,SchusterV,TejedorJC,CohenR,MeuriceF,HanHH,DamasoS,BouckenoogheA:Efficacyofhumanrotavirusvaccineagainstrotavirusgastroenteritisduringthefirst2yearsoflifeinEuropeaninfants:randomised,double-blindcontrolledstudy.
Lancet2007,370(9601):1757-1763.
8.
BlockSL,VesikariT,GoveiaMG,RiversSB,AdeyiBA,DallasMJ,BauderJ,BoslegoJW,HeatonPM:Efficacy:immunogenicity,andsafetyofthepentavalenthuman-bovine(WC3)reassortantRotavirusVaccineattheendofshelflife.
Pediatrics2007,119(1):11-18.
9.
ParasharUD,GibsonCJ,BresseJS,GlassRI:Rotavirusandseverechildhooddiarrhea.
EmergInfectDis2006,12(2):304-306.
10.
BonkoungouOJI,SanouI,BarroN,ToéL,SanfoMS,Ouédraogo/TraoréR,TraoreAS:ElectrophoretypescharacterizationofhumanRotavirusintwopediatricsservices,Ouagadougou,BurkinaFaso.
JMedSci2008,8:371-377.
11.
RodriguesA,deCarvalhoM,MonteiroS,MikkelsonSC,AabyP,MolbakkK,FischerTK:HospitalsurveillanceofrotavirusinfectionandnosocomialtransmissionofrotavirusdiseaseamongchildreninGuinea-Bissau.
PediatrInfectDisJ2007,26(3):233-237.
12.
deVilliersFPR,SawyerrTN,deVilliersGK:TheincidenceandclinicalpresentationofinfantilerotavirusdiarrhoeainSierraLeone.
SAfrMedJ2009,99(4):249-252.
13.
BinkaFN,AntoFK,OduroAR,AwiniEA,NazzarAK,ArmahGE,AsmahRH,HallAJ,CuttsF,AlexanderN,BrownD,GreenJ,GrayJ,Iturriza-GómaraM,NavrongoRotavirusResearchGroup:IncidenceandriskfactorsofpaediatricrotavirusdiarrhoeainNorthernGhana.
TropMedIntHealth2003,8(9):840-846.
14.
CunliffeNA,KilgorePE,BreseeJS,SteeleAD,LuoN,HartCA,GlassRI:EpidemiologyofrotavirusdiarrheainAfrica:areviewtoassesstheneedforrotavirusimmunization.
BullWHO1998,76(5):525-37.
15.
DeZoysaI,FeachemRG:Interventionsforthecontrolofdiarrhoealdiseaseamongyoungchildren:rotavirusandcholeraimmunization.
BullWHO1985,63(3):569-583.
16.
OffitPA:Hostfactorsassociatedwithprotectionagainstrotavirusdisease:theskiesareclearing.
JInfectDis1996,174(Suppl1):S59-S64.
17.
JiangB,GentschJR,GlassRI:Theroleofserumantibodiesintheprotectionagainstrotavirusdisease:anoverview.
ClinInfectDis2002,34(10):1351-1361.
18.
ZarnaniAH,ModarresS,JadaliF,SabahiF,MoazzeniSM,VazirianF:RoleofrotavirusesinchildrenwithacutediarrheainTehran,Iran.
JClinVirology2004,29(3):189-193.
19.
NaficyAB,Abu-ElyazeedR,HolmesJL,RaoRM,SavarinoJS,KimY,WierzbaTF,PeruskiL,LeeYJ,GentschJR,GlassRI,ClemensJD:EpidemiologyofRotavirusDiarrheainEgyptianChildrenandImplicationsforDiseaseControl.
AmJEpidemiol1999,150(7):770-777.
Bonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page5of620.
ClemensJ,RaoM,AhmedF,WardR,HudaS,ChakrabortyJ,YunusM,KhanMR,AliM,KayB,vanLoonF,SackD:Breast-feedingandtheriskoflife-threateningrotavirusdiarrhea:preventionorpostponementPediatrics1993,92(5):680-685.
21.
KapikianAZ,YasutakaH,ChanokRM:Rotaviruses.
Field'sVirology.
4edition.
Philadelphia:LippincottWilliamsandWilkinsCo;2001,1787-834.
22.
KukkulaM,ArstilaP,KlossnerML,MaunulaL,BonsdorffCH,JaatinenP:Waterborneoutbreakofviralgastroenteritis.
ScandJInfectDis1997,29(4):415-418.
23.
VillenaC,GabrieliR,PintoRM,GuixS,DoniaD,BuonomoE,PalombiL,CenkoF,BinoS,BoschA,DiviziaM:AlargeinfantilegastroenteritisoutbreakinAlbaniacausedbymultipleemergingrotavirusgenotypes.
EpidemiolInfect2003,131(3):1105-1110.
24.
BorchardtMA,HaasNL,HuntRJ:Vulnerabilityofdrinking-waterwellsinLaCrosse,Wisconsin,toenteric-viruscontaminationfromsurfacewatercontributions.
ApplEnvironMicrobiol2004,70(10):5937-5946.
25.
MartinelliD,PratoR,ChironnaM,SallustioA,CaputiG,ConversanoM,CiofiDegliAttiM,D'AnconaFP,GerminarioCA,QuartoM:LargeoutbreakofviralgastroenteritiscausedbycontaminateddrinkingwaterinApulia,Italy,May-October2006.
EuroSurveill2007,12(16):Article1.
Pre-publicationhistoryThepre-publicationhistoryforthispapercanbeaccessedhere:http://www.
biomedcentral.
com/1471-2431/10/94/prepubdoi:10.
1186/1471-2431-10-94Citethisarticleas:Bonkoungouetal.
:EpidemiologyofrotavirusinfectionamongyoungchildrenwithacutediarrhoeainBurkinaFaso.
BMCPediatrics201010:94.
SubmityournextmanuscripttoBioMedCentralandtakefulladvantageof:ConvenientonlinesubmissionThoroughpeerreviewNospaceconstraintsorcolorgurechargesImmediatepublicationonacceptanceInclusioninPubMed,CAS,ScopusandGoogleScholarResearchwhichisfreelyavailableforredistributionSubmityourmanuscriptatwww.
biomedcentral.
com/submitBonkoungouetal.
BMCPediatrics2010,10:94http://www.
biomedcentral.
com/1471-2431/10/94Page6of6
- 3.8www.00271相关文档
- readywww.00271
- 1.1www.00271
- "NO.","工程和计算机科学综述文集(Synthesis)各系列",
- schemewww.00271
- 2.52www.00271
- obtainedwww.00271
FlashFXP FTP工具无法连接主机常见原因及解决办法
目前,我们都在用哪个FTP软件?喜欢用的是WinSCP,是一款免费的FTP/SFTP软件。今天在帮助一个网友远程解决问题的时候看到他用的是FlashFXP FTP工具,这个工具以前我也用过,不过正版是需要付费的,但是网上有很多的绿色版本和破解版本。考虑到安全的问题,个人不建议选择破解版。但是这款软件还是比较好用的。今天主要是遇到他的虚拟主机无法通过FTP连接主机,这里我就帮忙看看到底是什么问题。一...
VoLLcloud:超便宜香港CMI大带宽vps-三网CMI直连-年付四免服务-低至4刀/月-奈飞
vollcloud LLC创立于2020年,是一家以互联网基础业务服务为主的 技术型企业,运营全球数据中心业务。致力于全球服务器租用、托管及云计算、DDOS安 全防护、数据实时存储、 高防服务器加速、域名、智能高防服务器、网络安全服务解决方案等领域的智 能化、规范化的体验服务。所有购买年付产品免费更换香港原生IP(支持解锁奈飞),商家承诺,支持3天内无条件退款(原路退回)!点击进入:vollclo...
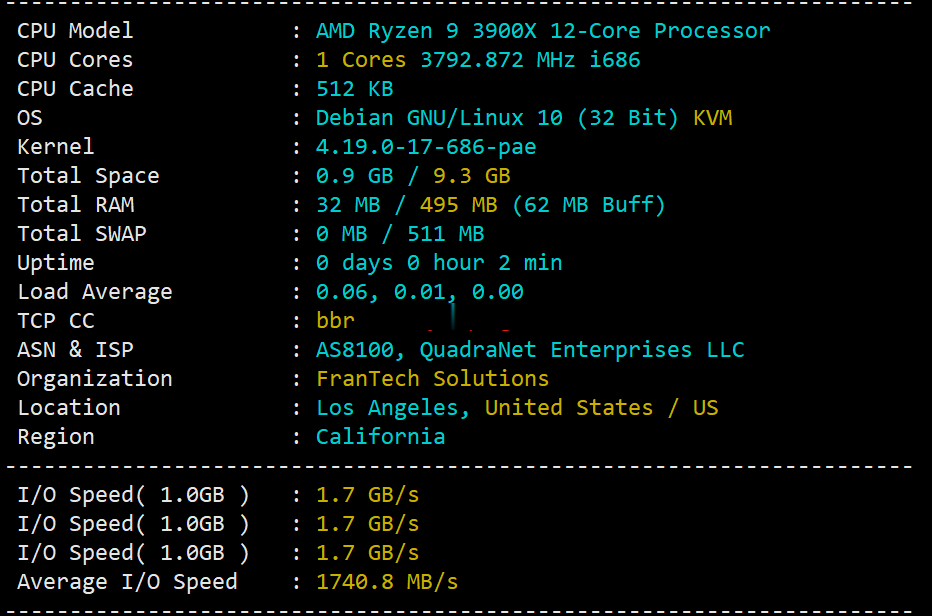
buyvm迈阿密机房VPS国内首发测评,高性能平台:AMD Ryzen 9 3900x+DDR4+NVMe+1Gbps带宽不限流量
buyvm的第四个数据中心上线了,位于美国东南沿海的迈阿密市。迈阿密的VPS依旧和buyvm其他机房的一样,KVM虚拟,Ryzen 9 3900x、DDR4、NVMe、1Gbps带宽、不限流量。目前还没有看见buyvm上架迈阿密的block storage,估计不久也会有的。 官方网站:https://my.frantech.ca/cart.php?gid=48 加密货币、信用卡、PayPal、...
www.00271为你推荐
-
今日油条油条的由来及历史今日油条油条是怎样由来广东GDP破10万亿在已披露的2017年GDP经济数据中,以下哪个省份GDP总量排名第一?xyq.163.cbg.comhttp://xyq.cbg.163.com/cgi-bin/equipquery.py?act=buy_show_equip_info&equip_id=475364&server_id=625 有金鱼贵吗?同ip网站同IP网站9个越来越多,为什么?www.yahoo.com.hk香港有什么有名的娱乐门户网站吗?同一服务器网站同一服务器上的域名/网址无法访问www.haole012.comhttp://fj.qq.com/news/wm/wm012.htm 这个链接的视频的 第3分20秒开始的 背景音乐 是什么?www.se333se.com米奇网www.qvod333.com 看电影的效果好不?haole10.com空人电影网改网址了?www.10yyy.cn是空人电影网么