fectedww.4399.com
ww.4399.com 时间:2021-03-22 阅读:()
2668TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June2014TargetingCD137enhancestheefficacyofcetuximabHolbrookE.
Kohrt,1A.
DimitriosColevas,1RochHouot,1,2,3KippWeiskopf,1MatthewJ.
Goldstein,1PederLund,1AntoniaMueller,1IditSagiv-Barfi,1AurelienMarabelle,1,4RuthLira,1EmilyTroutner,1LoriRichards,1AmandaRajapaska,1JonathanHebb,1CariadChester,1ErinWaller,1AntonOstashko,1Wen-KaiWeng,1LiepingChen,5DebraCzerwinski,1Yang-XinFu,6JohnSunwoo,1andRonaldLevy11DepartmentofMedicine,DivisionofOncology,StanfordUniversity,Stanford,California,USA.
2CHURennes,ServiceHématologieClinique,Rennes,France.
3INSERM,U917,Rennes,France.
4CentredeRechercheenCancérologiedeLyon,UMRINSERMU1052,CNRS5268,CentreLéonBérard,UniversitédeLyon,Lyon,France.
5DepartmentofImmunobiology,YaleCancerCenter,NewHaven,Connecticut,USA.
6DepartmentofPathologyandCommitteeonImmunology,UniversityofChicago,Chicago,Illinois,USA.
Treatmentwithcetuximab,anEGFR-targetingIgG1mAb,resultsinbeneficial,yetlimited,clinicalimprove-mentforpatientswithheadandneck(HN)canceraswellascolorectalcancer(CRC)patientswithWTKRAStumors.
Antibody-dependentcell-mediatedcytotoxicity(ADCC)byNKcellscontributestotheefficacyofcetuximab.
ThecostimulatorymoleculeCD137(4-1BB)isexpressedfollowingNKandmemoryTcellactiva-tion.
WefoundthatisolatedhumanNKcellssubstantiallyincreasedexpressionofCD137whenexposedtocetuximab-coated,EGFR-expressingHNandCRCcelllines.
Furthermore,activationofCD137withanago-nisticmAbenhancedNKcelldegranulationandcytotoxicity.
Inmultiplemurinexenograftmodels,includ-ingEGFR-expressingcancercells,HNcells,andKRAS-WTandKRAS-mutantCRC,combinedcetuximabandanti-CD137mAbadministrationwassynergisticandledtocompletetumorresolutionandprolongedsurvival,whichwasdependentonthepresenceofNKcells.
Inpatientsreceivingcetuximab,thelevelofCD137oncir-culatingandintratumoralNKcellswasdependentonpostcetuximabtimeandhostFcyRIIIapolymorphism.
Interestingly,theincreaseinCD137-expressingNKcellsdirectlycorrelatedtoanincreaseinEGFR-specificCD8+Tcells.
TheseresultssupportdevelopmentofasequentialantibodyapproachagainstEGFR-expressingmalignanciesthatfirsttargetsthetumorandthenthehostimmunesystem.
IntroductionColorectalcarcinoma(CRC)isestimatedtoremainthethirdmostcommoncancer,affectingover140,000livesandleadingtoover50,000deathsintheUnitedStatesin2013alone.
Thoughcasesofheadandneck(HN)cancerare3-foldlesscommon,theincidencecontinuestoriseduetoagrowingpopulationinfectedwithhumanpapillomavirus(HPV)(1).
EGFRERBB1(avianerythroblasticleu-kemiaviral[v-erb-b]oncogenehomolog,receptorforEGF)isapoorprognosticmarkerinmultiplesolidtumors,includingCRC,HN,non–smallcelllung,andpancreaticcarcinomas,andisatherapeu-tictargetofmAbs(cetuximabandpanitumumab)aswellassmallmoleculekinaseinhibitors(erlotinibandgefitinib).
Cetuximab,ahuman-mousechimericIgG1mAb,wasapprovedbytheFoodandDrugAdministrationfirstin2004andtodayisindicatedforpatientswithCRCandHNcarcinomas(2).
However,responsestocetuximabmonotherapyarelimited;only1in5patientswithmetastaticCRC(3)andonly1in10withadvancedHNcarcino-marespondtotreatment(4,5).
Despiteexpressionofthetarget,patientswithnon–smallcelllung,pancreatic,andCRCwithcodon11or12KRASmutationsfailtobenefitfromcetuximab(6).
Givenmultiplemechanismsofcetuximab'saction,avarietyofapproacheshavebeenpursuedtoenhanceitsefficacy.
Bycompeti-tivelyblockingthebindingofEGF,phosphorylationandkinaseactivationareprevented,therebyinhibitingcellgrowth,inducingapoptosis,anddecreasingproductionofmatrixmetalloproteinaseandvascularendothelialgrowthfactor(7,8).
Inadditiontotheseinvitroeffects,invivoevidenceinbothmurinemodelsandpatientssuggestscetuximab'sefficacyisduetoantibody-dependentcell-mediatedcytotoxicity(ADCC),whichrequiresimmuneeffectorcells,mainlyNKcells,bindingviatheirFcreceptor(FcγRIII,CD16)totheIgG1Fc,heavy-chain,portionofcetuximab(9–13).
TargetingEGFRbysmallmoleculesthatlackanFc,andthereforelackADCC,hasnotresultedinaclinicalbenefitinHNorCRCs.
SupportingADCCasaprimarymechanismofcetuximab'sactivityinpatients,NKcellinfiltratewithinprimarycolorectaltumorsindependentlypredictsprognosis(14).
PatientswithcolorectalandHNcarcino-masharboringahigh-affinityFcγRIIIpolymorphismhavebeenshowntorespondmorefavorablytocetuximabbothexvivowithhighercytotoxicityagainstEGFR-expressingcelllines(15)andclin-icallywithsuperiordisease-freeandoverallsurvival(15–19).
There-fore,methodstoenhanceADCC,suchasstimulatingtheinnateimmuneresponse,mayclinicallytranslatetoimprovedantitumoractivity.
AugmentingtheNKcellresponsetocetuximabtherapymayenhancetheadaptiveimmuneresponseinadditiontoinnateimmunityduetoNKcell–DC"crosstalk,"whichleadstotumorantigen–specificTcellresponsesfollowingcetuximabtherapy(20).
Wesoughttoidentifyaninducibleandtargetablecostimula-torymoleculeonNKcellsinordertoenhanceADCC.
CD137(4-1BB)isupregulatedonhumanNKcellswhentheyencounterantibody-boundtumorcells(21).
Therefore,wehypothesizedthattheantitumorefficacyofcetuximabcouldbeimprovedthroughadualantibodystrategy:firstbyinducingCD137expressiononNKcellsupontheirexposuretocetuximab-boundtumorcellsandsubsequentlybytargetingactivatedNKcellswithanagonis-ticanti-CD137mAb.
Conflictofinterest:Theauthorshavedeclaredthatnoconflictofinterestexists.
Citationforthisarticle:JClinInvest.
2014;124(6):2668–2682.
doi:10.
1172/JCI73014.
RelatedCommentary,page2371TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142669ResultsCetuximabinducesCD137upregulationonhumanNKcellsfollowingincubationwithEGFR-positivetumorcells.
CD137expressionwasinducedonthesurfaceofNKcellsfromhealthyhumansubjectsfollowingincubationwithcetuximabandEGFR-expressingcan-cercelllines(SCC6,PC1,andSCC4)(Figure1A).
ThisCD137upregulationrequiredthepresenceofbothanEGFR-expressingcellandanEGFR-targetingmAb,aslittleeffectonCD137expres-sionwasobservedwithcetuximaborwithEGFR-expressingcan-cercelllinesalone.
Similarly,NKcellexpressionofCD137didnotincreasefollowingculturewithanon-EGFR–targetingmAb,rituximab,whichtargetsCD20,eveninthepresenceoftheEGFR-expressingcells(Figure1,BandC).
TheinductionofCD137occurredpreferentiallyinCD56dimcomparedwithCD56hiNKcellsandamongthissubsetwasassociatedwithaconcurrentdecreaseintheexpressionoftheFcγRIII(CD16)(Figure1,A–C).
Anti-CD137agonisticmAbincreasescetuximab-mediatedNKcellcytotoxicityontumorcellsandDCcytokinesecretion.
TodeterminewhetherCD137isapotentialtherapeutictargetforenhancingNKcellfunction,NKcellswerefirstactivatedtoexpressCD137bytheirexposuretoEGFR-expressingcancercellsandcetuximab.
ActivatedCD137-expressingNKcellswerethenreisolatedandtestedfortheirabilitytoperformADCCagainstEGFR-expressingcancercells(Fig-ure2,A–F).
ActivatedNKcellsshowedenhancedADCCfollowinganti-CD137mAbstimulation,asmeasuredbyapoptosis(Figure2,A–C)andchromiumrelease(Figure2,D–F)fromEGFR-expressingcancercells.
Thoughtheanti-CD137mAbenhancedcytotoxicity,specificallyADCC,enhancedIFN-γsecretion,anotherusualmea-sureofNKcellfunction,incontrast,wasnotobserved(Supple-mentalFigure1,A–C;supplementalmaterialavailableonlinewiththisarticle;doi:10.
1172/JCI73014DS1).
However,inthepresenceofactivatedNKcells,EGFR-expressingcancercells,cetuximab,andimmatureDCs(iDCs),anti-CD137agonisticmAbenhancedsecre-tionofIL-12,IFN-γ,andTNF-α(SupplementalFigure1,D–F).
Anti-CD137agonisticmAbenhancesantitumoractivityofcetuximabinvivoagainstbothKRAS-mutantandKRAS-WTtumors.
WetestedtheabilityofcetuximabtoinduceCD137expressiononmurineNKcellsinvivoinanEGFR-expressingxenotransplantmodel(SCC6).
Weadministeredcetuximab21daysfollowingimplantationoftumorandmeasuredtheexpressionofCD137onNKcellsintheperipheralblood,spleen,andtumor(Figure3A).
Ineachofthe3compartmentsandatseveraltimepoints,weobservedupregulatedexpressionofCD137onNKcells(Figure3,BandC).
Interestingly,atthetumorsite,CD137expressionpeakedat24hoursfollowingFigure1CetuximabinducesCD137upregulationonhumanNKcellsfollowingincubationwithEGFR-positivetumorcells.
PeripheralbloodfromhealthydonorswasanalyzedforCD137expressiononCD3–CD56+NKcellsafter24-hourculturewithEGFR-positivetumorcelllinesSCC6,PC1,andSCC4,andcetuximaborrituximab.
(A)PercentageofNKcellsdividedbyquadranttodelineatesubsetsofCD3–CD56brightandCD3–CD56dimexpressingCD137fromarepresentativehealthydonorafter24-hourculturewiththeEGFR-positivetumorcelllinePC1andcetuximab.
(B)PercentageofCD137expressiononNKcellsfrom3healthydonorsafter24-hourculturewiththeEGFR-positivetumorcelllineSCC6,PC1,orSCC4,andcetuximaborrituximab.
(C)CD16expressiononNKcellsfroma3healthydonorsafter24-hourculturewiththeEGFR-positivetumorcelllineSCC6,PC1,orSCC4,andcetuximaborrituximab.
*P90%purityasdefinedbyCD3–CD56+flowcytometry)andactivation(>50%expressionofCD137).
EGFR-expressingcancercelllinesincludingSCC6(AandD),PC1(BandE),andSCC4(CandF)wereculturedfor18hourswithpreactivated,purifiedNKcellsinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml).
CellswerewashedandincubatedwithannexinVand7-AADtodeterminepercentageofapoptotictumorcellsbyannexinVand7-AADstaining.
Chromium-labeledEGFR-expressingcancercelllineswereculturedfor4hourswithpreactivated,purifiedNKcellsinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs.
Shownispercentagelysisoftargetcellsbychromiumreleaseatvaryingeffector(activatedNKcells)/targetcellratiosculturedwithmediumalone(circles),anti-CD137(upside-downtriangle),cetux-imab(triangle),orcetuximabandanti-CD137(square)antibodies.
*P=0.
002,SCC6(A);*P=0.
007,PC1(B);*P=0.
007,SCC4(C);*P=0.
021,SCC6(D);*P=0.
011,PC1(E);*P=0.
049,SCC4(F).
TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142671wassuperiortoconcurrentcombinationdosingineffectonbothtumorgrowthandsurvival.
ThistherapeuticschemawastestedonbothKRAS-WT(T84)andKRAS-mutant(HCT116G13D)tumorsanddemonstratedasimilartherapeuticbenefittothesequentialcombinationofcetuximabfollowedbyanti-CD137mAb,withnearcompletetumorregressionandallanimalssurviving100daysaftertumorimplantation(Figure4,D–F).
Enhancementoftheantitumoractivityofcetuximabbyanti-CD137agonisticmAbisdependentonNKcellsandCD8+Tcells.
Theefficacyweobservedwithcombinationcetuximabandanti-CD137mAbtherapyinathymicmicestronglyimplicatestheinnateimmunesysteminthemechanismofaction.
Inordertodeterminewhethertheadaptiveimmuneresponsealsoplaysarole,weappliedthecombinationtherapytoasyngeneictumormodelinimmuno-competentBALB/cmice,usingatumorcellline,TUBO,trans-fectedwithhumanEGFR,previouslydeterminedtobesensitivetocetuximabtreatment(Figure5Aandref.
22).
ThiscelllineisHER2/neu-dependentandderivedfromBALB/cmicetransgenicfortheneuoncogene.
Inthismodel,anti-CD137mAbtherapyalonehadnoeffectontumorgrowthandcetuximabmonothera-pysignificantlyslowedtumorgrowth,whilethesequentialcom-binationofcetuximabfollowedbyanti-CD137mAbledtotumorregressionandimprovedsurvival,withallmiceinthisgroupaliveatday100(Figure5,BandC).
DepletionofCD4+Tcellsormacrophageshadnoinfluenceonefficacy(Figure5,DandE).
Ashypothesized,depletionofNKcellsalmostcompletelyabrogatedthetherapeuticbenefit.
Butsurpris-ingly,depletionofCD8+TcellspartiallyreducedtheefficacyoftheFigure3CetuximabmAbinducesCD137expressioninvivo.
nu/numicewereinoculatedwith1*106SCC6tumorcellss.
c.
ontheleftflank,andcetux-imabwasadministeredonday21aftertumorinoculation(A,schema).
(B)RepresentativeCD137expressiononCD3–DX5+cellsisolatedfromthespleenorperipheralblood24hoursfollowingcetuximabtreatmentorisotypecontrolinnon–tumor-bearingmice(n=3micepergroup).
(C)RepresentativeCD3–DX5+cellsisolatedfromthespleen,peripheralblood,ortumoranalyzedforCD137expression24,72,or168hoursfol-lowingcetuximabtreatmentorisotypecontrolintumor-bearingmice(n=3micepergroup).
ForBandC,numbersindicatepercentageofcellsaboveandbelowupperlimitofisotypecontrol.
2672TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June2014Figure4Anti-CD137agonisticmAbenhancesantitumoractivityofcetuximabinvivoagainstKRAS-mutantandKRAS-WTEGFR-expressingtumors.
nu/numicewereinoculatedwith1*106SCC6tumorcells(KRAS-WT)s.
c.
ontheleftflank,andcetuximaband/oranti-CD137mAbwasadminis-teredstartingonday21aftertumorinoculation(A,schema).
(BandC)Followingtumorinoculationonday0,micereceivedeitherratIgGcontrolonday21(circles),cetuximabonday21(squares),anti-CD137antibodyonday22(diamonds),cetuximabonday21andanti-CD137antibodyonday21(upside-downtriangles),orcetuximabonday21andanti-CD137antibodyonday22(triangles),witheachinjectionrepeatedweeklyforatotalof4injections.
Mice(10pergroup)werethenmonitoredfortumorgrowth(B)andoverallsurvival(C).
nu/numicewereinoculatedwith1*106T84(KRAS-WT)orHCT116(KRAS-mutant)tumorcellss.
c.
ontheleftflank,andcetuximaband/oranti-CD137mAbwasadministeredstartingonday21aftertumorinoculation(D,schema).
(EandF)Aftertumorinoculation,miceinoculatedwithHCT116thenreceivedeitherratIgGcontrolonday21(blackcircles)orcetuximabonday21andanti-CD137antibodyonday22(bluecircles),witheachinjectionrepeatedweeklyforatotalof4injections.
MiceinoculatedwithT84thenreceivedeitherratIgGcontrolonday21(blackdiamonds)orcetuximabonday21andanti-CD137antibodyonday22(reddiamonds),witheachinjectionrepeatedweeklyforatotalof4injections.
Mice(10pergroup)werethenmonitoredfortumorgrowth(E)andoverallsurvival(F).
*P50%expressionofCD137).
TheactivatedNKcellswerethenculturedwithadditionalEGFR-expressingcancercelllines(SCC6,PC1,orSCC4)ataratioof1:1.
After18hours,thesupernatantswereremovedandassayedforIFN-γbyELISA(HumanInterferon-γBDOptEIAELISASetandBDOptEIAReagentSetB;BDBiosciences—Pharmingen).
CellsfromthesecultureswereincubatedwithFITC–annexinVand7-AAD(bothBDBiosciences)andanalyzedbyflowcytometry.
ForwardandsidescatterwereusedtogateouttheNKcells,andfluorescencewasusedtodeter-minethepercentageofapoptotictumorcellsbyannexinVand7-AADstaining.
NKcellcytotoxicitywasadditionallymeasuredbyachromiumWehavestudiedpatientswithHNcarcinomareceivingthera-pywithcetuximab.
WefoundNKcellsincirculationandinthetumorsthathaveupregulatedCD137.
WehavealsofoundthatFcγRIIIpolymorphismscorrelatewiththemagnitudeofCD137upregulation,withincreasedCD137associatedwiththehigh-affinityalleles.
WeobservedasignificantincreaseinNKcellCD137expressionfollowingcetuximaboccurringwithin24hours;insomepatients,itwaselevatedaslongas1week.
Theincreasedexpressionintheperipheralbloodandwithinthetumorwashet-erogeneous,withasubsetofpatientsdemonstratingminimalupregulation.
ThoughtheongoingandplannedphaseIandI/IIclinicaltrialsdonotlimitenrollmentbasedonCD137expres-sion,wespeculatethatpatientswithminimalupregulationofCD137maynotbenefitfromanti-CD137mAbtherapy.
Toiden-tifypredictorsofCD137upregulation,theFcγRIIIpolymorphismofallpatientswastypedandstratifiedbypresenceofasinglehigh-affinityallele.
PatientswithHNcancerwitheitherV/VorV/FdemonstratedgreaterCD137upregulationcomparedwithpatientswith2low-affinityalleles.
Withcontinuedenrollmentinthisphase0study,wewillidentifyclinicalandpathologicchar-acteristicsthatpredicttheheterogeneityinCD137upregulation.
Bydoingso,wehopetocreateanomogrampredictiveofCD137expression,whichwithvalidationinongoingclinicaltrialsandcorrelationwithclinicalresponsetoanti-CD137mAbtherapy,mayprovideapredictivebiomarkerforpatientselectionandindi-viduallytailoredtreatmentschedules.
Bychangingtheparadigmofimprovingantibodytechnology—notbycreatingsuperiortumor-targetingmAbs,ratherbytarget-ingthesecondaryimmuneresponse—wehaveidentifiedathera-peuticstrategythatisselectivetothetumormicroenvironmentandefficaciousinmultiplemodels,includingthoseresistanttotreatment.
Theapproachisdependentuponsequentialtarget-ingoffirst,thetumor,bycetuximab,andsecond,theactivatedNKcells,byanti-CD137agonisticmAbs.
ThisstrategyinitiatesamorerobustinnateimmuneresponsewithaugmentedADCCaswellasadaptiveimmunitythroughlikelyenhancedcrosstalkbetweenNKcells,DCs,andtumorantigen–specificCD8+Tcells.
ByinvestigatingpredictorsofCD137expression,suchasFcγRIIIpolymorphism,inpatientsreceivingcetuximabmonotherapy,wehopetoidentifybiomarkersthatwillbetestedalongwiththecen-tralhypothesisofthisnewstrategyinaplannedcombinationtrialwithananti-CD137agonisticantibody.
MethodsCelllinesandculture.
ThehumanEGFR-expressingcancercelllinesSCC4,SCC6,andPC1wereobtainedfromJohnSunwooatStanfordUniversity.
SCC4isanupperaerodigestivetractsquamouscellcarcinomawithmuta-tionsinCDKN2A,NF1,andTP53andWTKRAS,EGFR,MET,NRAS,andPTEN.
SCC6isanupperaerodigestivetractsquamouscellcarcinomawithmutationsinCDKN2AandWTNF1,TP53,KRAS,EGFR,ET,NRAS,andPTEN.
PC1isapancreaticadenocarcinomawithmutationsinCDKN2A,KRAS(G12D),andTP53andWTEGFR,NRAS,MET,NF1,andPTEN.
HumanEGFR-positivecancercelllinesT84(KRAS-WT),HCT116(KRASG13Dmutant),andA20(alymphomacelllineusedasanegativecontrol)wereobtainedfromATCC.
T84isaCRCwithmutationsinAPCandTP53andWTNF1,KRAS,NRAS,MET,PTEN,andEGFR.
HCT116isaCRCwithmutationsinKRAS(G13D),APC,andTP53andWTNF1,NRAS,MET,PTEN,andEGFR.
TheTUBO-EGFRmurinecancercelllinewasobtainedfromYang-XinFuattheUniversityofChicago.
TheSCC4,SCC6,PC1,andT84celllineswereculturedina1:1mixtureofDMEM:F12supplemented2680TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June2014precipitateredcells.
Leukocyte-containingsupernatantwasremovedandcentrifuged,andtheremainingredcellswerelysedwithACKLysisBuffer(QualityBiological).
Splenocytesandsubcutaneoustumorsfrom3micepertreatmentgroupwereresected24,72,or168hoursfollowingcetux-imabtreatmentorisotypecontrolintumor-bearingmiceandmechani-callydigestedintoasingle-cellsuspension.
CellswerestainedwithmAbstoevaluateCD137expressiononNKcellsbyCD3–DX5+expressionbyflowcytometry.
SubcutaneousTUBOandTUBO-EGFRtumorsfrom5micepertreatmentgroupwereresectedonday24aftertumorinoculationandanalyzedforNKcell,Tcell,andDCinfiltrationbyflowcytometryofasingle-cellsuspensionfollowingmechanicaldigestion.
Thetumor-infil-tratingDCpopulationwasmarkedbyCD11c+andhighsurfaceexpres-sionofMHCclassIandclassIIproteinswithexpressionofcostimula-torymoleculesCD80(B7.
1)andCD86(B7.
2).
PBMCscollectedonday24aftertumorinoculationfrom5micepergroupinthe2tumormodelwerepurifiedasdescribedandthencoculturedwithmedium,1*106irradiatedA20,TUBO,orTUBO-EGFRcellsfor24hourswith0.
5mganti-mouseCD28mAb(BDBiosciences—Pharmingen)andinthepresenceofmonen-sin(Golgistop;BDBiosciences)forthelast5hoursat37°Cand5%CO2.
IntracellularIFN-γexpressionwasassessedwithBDCytofix/CytopermPlusKitperinstructions.
DepletionofCD4+andCD8+Tcells,NKcells,andmacrophages.
Anti-CD4mAb,anti-CD8mAb,anti-asialoGM1mAb(WakoChemicals),andlipo-somalclodronate(Weissmanlaboratory,StanfordUniversity)wereusedtodepleteCD4+Tcell,CD8+Tcell,NKcell,andmacrophageactivityinvivo,respectively.
AsciticfluidwasharvestedfromSCIDmicebearinghybrid-omasGK1.
5-andGK2.
43-producinganti-CD4(ratIgG2b)andanti-CD8(ratIgG2b)mAbs.
TheascitesweredilutedinsterilePBS.
Depletinganti-CD4mAb,anti-CD8mAb,andanti-asialoGM1mAbwereinjectedi.
p.
onday–1andday0oftumorinoculation,andevery5daysthereafteruntilday45atadoseof500μgperinjectionofanti-CD4mAboranti-CD8mAband50μlperinjectionofanti-asialoGM1mAb.
Liposomalclodronatewasinjectedretroorbitallyonday–2atadoseof200μlandonday0oftumorinoculation,followedbyevery4daysuntilday44atadoseof100μlperinjection.
Thedepletionconditionswerevalidatedbyflowcytometryofbloodshowingmorethan95%depletionofeachcellsubset.
Antibodiesforflowcytometry.
mAbstohumanantigenswereusedforstain-ingofhumanPBMCsincludingthefollowing:CD16FITC,CD4FITC,CD8FITC,CD137ligandPE,CD56PE,CD3PerCP,anti-EGFRAPC,CD137APC,APC-H7,FITC–annexinV,and7-AAD(allfromBD).
HIV-tetramerPEandEGFR-tetramer(YLN)PEwereprovidedbytheNIHTet-ramerCoreFacility(Atlanta,Georgia,USA)aswellasMarkDavis(Stan-fordUniversity).
ThefollowingmAbstomouseantigenswereused:CD8FITC(BD),CD4FITC(BD),CD137ligandPE(eBioscience),CD137PE(eBioscience),CD3PerCP(BD),DX5APC(eBioscience),CD11c-PE(BD),MHCIAPC(BD),MHCIIAPC(BD),B7.
1APC(BD),B7.
2FITC(BD),andF4/80APC(eBioscience).
StainedcellswerecollectedonaFACSCaliburoraLSRII3-lasercytometer(BD),anddatawereanalyzedusingCytobank(http://www.
cytobank.
org)(61).
AnalysisofFcγRIIIapolymorphisms.
GenomicDNAwaspreparedfromperipheralbloodmononuclearcellsusingaDNAextractionkit(QIAGEN).
GenotypingofFcγRIIIa158V/Fpolymorphismwasperformedbyapoly-merasechainreactionfollowedbyallele-specificrestrictionenzymediges-tion(62).
AllgenotypingofFcγRIIIapolymorphismwasconfirmedbydirectsequencingoftheregionofinterest.
Statistics.
Prismsoftware(GraphPad)wasusedtoanalyzetumorgrowthandcomparisonofcellpopulationstodeterminestatisticalsignificanceofdifferences(datapresentedasmean±SEM)betweengroupsbyapplyinga2-tailed,unpairedStudent'sttestor2-wayANOVAwithBonferroni'scor-rectionformultiplecomparisons.
Kaplan-Meierplotswereusedtoanalyzereleaseassay(60):targettumorcellswerelabeledwith150μCi51Crper1*106cellsfor2hours.
Activated,purified,andnonpurifiedNKcellswerepreparedasdescribedaboveandwerethenaddedatvariableeffec-tor/targetcellratiosfrom1:1to50:1.
Percentageoflysiswasdeterminedafter4hoursofcultureinthepresenceofanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml).
Allassayswereperformedintriplicatewith3independentNKcellsamples.
InvitroDCfunctionalassays.
AutologousiDCswerepreparedasprevi-ouslydescribed(52).
iDCpopulationswerecheckedforCD1aandCD80expressionandlackofCD14expression.
ActivatedandpurifiedNKcellswerepreparedasdescribedabove.
ActivationofNKcellswasconfirmedbyflowcytometry(>50%expressionofCD137).
TheactivatedNKcellswerethenculturedwithautologousiDCsandEGFR-expressingcancercelllines(SCC6,PC1,orSCC4)ataratioof1:1:1for18hoursinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml);supernatantwasharvestedandanalyzedbyELISAforIFN-γ,IL-12p40,andTNF-α(humanIFN-γ,IL-12p40,andTNR-αBDOptEIAELISASetsandBDOptEIAReagentSetB;BDPharmingen).
Allassayswereperformedintriplicatewith3independentNK:iDCcellsamples.
Tumortransplantationandantibodytherapy.
SCC6cancercellswereimplanteds.
c.
into5-to6-week-oldfemaleathymicnu/numiceatadoseof1*106cellsin50μlofPBS(BDBiosciences).
mAbwereadministeredbyi.
p.
injections.
Cetuximabwasgiveni.
p.
at200μgperinjection,andanti-CD137mAbwasgiveni.
p.
at150μgperinjectionatvarioustimesaftertumorinoculation.
TodeterminetheefficacyagainstKRAS-WTandmutanttumors,nu/numicewereinoculatedwith1*106T84(KRAS-WT)tumorcellsor1*106HCT116(KRASG13Dmutant)tumorcellss.
c.
ontheleftflank.
Todeterminetheimmunecellsubsetsnecessaryforefficacy,BALB/cmicewereinoculatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflank.
Cetuximabandanti-CD137mAbwereadministeredaftertumorinoculationasabove.
Tocharacterizetheadaptiveimmuneresponsefollowingcombinationtherapy,BALB/cmicewereinoculatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflankand1*106TUBO-EGFRtumorcellss.
c.
ontherightflank.
Cetuximabandanti-CD137mAbwereadministeredaftertumorinoculationasabove.
Tumorgrowthwasmea-suredbycalipertwiceaweekandexpressedastheproductoflengthbywidthinsquarecentimeters.
Miceweresacrificedwhentumorsizereached4cm2orwhentumorsitesulcerated.
Allinvivomodelswerepilotedwith5micepergroupandrepeatedwith10micepergroup,exceptinthe2-tumormodel,inwhich15micewereinoculatedand5pergroupsacri-ficedonday24fortumor-infiltratinglymphocyteanalysisandperipheralbloodcollectionforassessmentofCD8+Tcellfunctionexvivo.
Adoptiveimmunitytransferexperiments(Winnassay).
BALB/cmicewereinoc-ulatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflankandtreat-edwitheithercetuximabmonotherapyondays14,21,28,and35orsequen-tialcetuximabandanti-CD137mAbcombinationwithcetuximabdosedasinthemonotherapygroupandanti-CD137mAbonthesubsequentday.
Allmiceweresacrificed50daysafterbeinginoculatedwithtumorcells.
TotalorCD8-selectedsuspensionswerepreparedbyCD8a+TcellIsolationKit(MiltenyiBiotecGmbH)andpurificationassessedbyflowcytometricanalysisandconfirmedasmorethan95%.
Totalorinvitro–purifiedCD8+splenocyteswereadmixedwithTUBOorTUBO-EGFRtumorcells(100:1ratio)andcoinjectedintonaiveBALB/cmice.
Theresultswereexpressedaspercentageofanimalsthatweretumor-freeovertime.
DetectionofCD137-expressingcellsinvivo,tumor-infiltratingimmunecellsinvivo,andIFN-γproductionassayexvivo.
Bloodwascollectedfromthetailvein,anticoagulatedwith2mMEDTAinPBS,thendiluted1:1with2%DextranT500(Pharmacosmos)inPBSandincubatedat37°Cfor45minutestoTheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142681ardHughesMedicalInstituteResearchTrainingFellowship.
R.
HouotissupportedbyfellowshipsfromtheFoundationdeFranceandtheAssociationpourlaRecherchesurleCancer.
H.
E.
KohrtisaDamonRunyonClinicalInvestigatorandissupportedbyfellowshipsfromtheAmericanSocietyofHematologyandtheAmericanSocietyofClinicalOncologyandDepartmentofDefenseBreastCancerpostdoctoralfellowshipaward(26687).
K.
WeiskopfissupportedbytheStanfordUniversityMedicalSci-entistTrainingProgram.
R.
LevyisanAmericanCancerSocietyClinicalResearchProfessor.
ReceivedforpublicationSeptember3,2013,andacceptedinrevisedformMarch6,2014.
Addresscorrespondenceto:HolbrookKohrt,DivisionofOncol-ogy,269CampusDrive,CCSR1105,StanfordUniversityMedi-calCenter,Stanford,California94305-5151,USA.
Phone:650.
725.
6452;Fax:650.
736.
1454;E-mail:Kohrt@stanford.
edu.
survival.
Comparisonsofsurvivalcurvesweremadeusingthelog-ranktest.
CorrelationcoefficientswerecalculatedforcomparisonofpercentageofEGFR+CD8+TcellsandpercentageofCD137+NKcells(datapresentedasmean±SEM).
P<0.
05wasconsideredsignificant.
Fortumorburdens,comparisonsofmeansweredonebyANOVA.
Studyapproval.
HumanperipheralbloodandprimarybreasttumorsampleswereobtainedunderareviewedprotocolapprovedbyStanfordUniversity'sAdministrativePanelsonHumanSubjects(NCT01114256)andfollowinginformedconsentfrompatientsinaccordancewiththeDec-larationofHelsinki.
AllanimalexperimentswereapprovedbytheStanfordAdministrativePanelonLaboratoryAnimalCareandconductedinaccor-dancewithNIHguidelines.
AcknowledgmentsThisworkwassupportedbytheCaliforniaBreastCancerResearchProgram,EUREKA(R01CA153248-01),theReliableCancerTherapyFund,andNadia'sGiftFoundationattheStan-fordCancerCenter.
M.
J.
GoldsteinissupportedbytheHow-1.
AmericanCancerSociety.
CancerFactsandFigures2013.
Atlanta,Georgia,USA:AmericanCancerSociety;2013.
2.
LeeCM,TannockIF.
Thedistributionofthethera-peuticmonoclonalantibodiescetuximabandtrastuzumabwithinsolidtumors.
BMCCancer.
2010;10:255.
3.
DeRoockW,DeVriendtV,NormannoN,Ciardi-elloF,TejparS.
KRAS,BRAF,PIK3CA,andPTENmutations:implicationsfortargetedtherapiesinmetastaticcolorectalcancer.
LancetOncol.
2011;12(6):594–603.
4.
BaselgaJ,etal.
PhaseIImulticenterstudyoftheantiepidermalgrowthfactorreceptormonoclo-nalantibodycetuximabincombinationwithplatinum-basedchemotherapyinpatientswithplatinum-refractorymetastaticand/orrecurrentsquamouscellcarcinomaoftheheadandneck.
JClinOncol.
2005;23(24):5568–5577.
5.
VermorkenJB,etal.
Open-label,uncontrolled,mul-ticenterphaseIIstudytoevaluatetheefficacyandtoxicityofcetuximabasasingleagentinpatientswithrecurrentand/ormetastaticsquamouscellcar-cinomaoftheheadandneckwhofailedtorespondtoplatinum-basedtherapy.
JClinOncol.
2007;25(16):2171–2177.
6.
LiuL,CaoY,TanA,LiaoC,GaoF.
Cetuximab-basedtherapyversusnon-cetuximabtherapyforadvancedcancer:ameta-analysisof17randomizedcontrolledtrials.
CancerChemotherPharmacol.
2010;65(5):849–861.
7.
LiX,LuY,LiangK,PanT,MendelsohnJ,FanZ.
Requirementofhypoxia-induciblefactor-1alphadown-regulationinmediatingtheantitumoractiv-ityoftheanti-epidermalgrowthfactorreceptormonoclonalantibodycetuximab.
MolCancerTher.
2008;7(5):1207–1217.
8.
HuangSM,LiJ,HarariPM.
Molecularinhibitionofangiogenesisandmetastaticpotentialinhumansquamouscellcarcinomasafterepidermalgrowthfactorreceptorblockade.
MolCancerTher.
2002;1(7):507–514.
9.
ClynesRA,TowersTL,PrestaLG,RavetchJV.
InhibitoryFcreceptorsmodulateinvivocytoxicityagainsttumortargets.
NatMed.
2000;6(4):443–446.
10.
KuraiJ,etal.
Antibody-dependentcellularcytotoxicitymediatedbycetuximabagainstlungcan-cercelllines.
ClinCancerRes.
2007;13(5):1552–1561.
11.
SteinerP,etal.
Tumorgrowthinhibitionwithcetuximabandchemotherapyinnon-smallcelllungcancerxenograftsexpressingwild-typeandmutatedepidermalgrowthfactorreceptor.
ClinCancerRes.
2007;13(5):1540–1551.
12.
RabenD,etal.
Theeffectsofcetuximabaloneandincombinationwithradiationand/orchemotherapyinlungcancer.
ClinCancerRes.
2005;11(2pt1):795–805.
13.
WilsonNS,etal.
AnFcγreceptor-dependentmechanismdrivesantibody-mediatedtarget-receptorsignalingincancercells.
CancerCell.
2011;19(1):101–113.
14.
MarechalR,etal.
PutativecontributionofCD56positivecellsincetuximabtreatmentefficacyinfirst-linemetastaticcolorectalcancerpatients.
BMCCancer.
2010;10:340.
15.
Lopez-AlbaiteroA,etal.
RoleofpolymorphicFcγreceptorIIIaandEGFRexpressionlevelincetuximabmediated,NKcelldependentinvitrocytotoxicityofheadandnecksquamouscellcarcinomacells.
CancerImmunolImmunother.
2009;58(11):1853–1864.
16.
BibeauF,etal.
ImpactofFc{gamma}RIIa-Fc{gamma}RIIIapolymorphismsandKRASmuta-tionsontheclinicaloutcomeofpatientswithmet-astaticcolorectalcancertreatedwithcetuximabplusirinotecan.
JClinOncol.
2009;27(7):1122–1129.
17.
FerrisRL,JaffeeEM,FerroneS.
Tumorantigen-tar-geted,monoclonalantibody-basedimmunothera-py:clinicalresponse,cellularimmunity,andimmu-noescape.
JClinOncol.
2010;28(28):4390–4399.
18.
QiuLX,etal.
PredictiveandprognosticvalueofKRASmutationsinmetastaticcolorectalcancerpatientstreatedwithcetuximab:ameta-analysisof22studies.
EurJCancer.
2010;46(15):2781–2787.
19.
RodriguezJ,etal.
FcγreceptorpolymorphismsaspredictivemarkersofCetuximabefficacyinepider-malgrowthfactorreceptordownstream-mutatedmetastaticcolorectalcancer.
EurJCancer.
2012;48(12):1774–1780.
20.
LeeSC,SrivastavaRM,Lopez-AlbaiteroA,FerroneS,FerrisRL.
Naturalkiller(NK):dendriticcell(DC)crosstalkinducedbytherapeuticmonoclonalanti-bodytriggerstumorantigen-specificTcellimmu-nity.
ImmunolRes.
2011;50(2–3):248–254.
21.
LinW,etal.
Fc-dependentexpressionofCD137onhumanNKcells:insightsinto"agonistic"effectsofanti-CD137monoclonalantibodies.
Blood.
2008;112(3):699–707.
22.
YangX,ZhangX,MortensonED,Radkevich-BrownO,WangY,FuYX.
Cetuximab-mediatedtumorregressiondependsoninnateandadaptiveimmuneresponses.
MolTher.
2013;21(1):91–100.
23.
SchulerPJ,etal.
EGFR-specificTcellfrequenciescorrelatewithEGFRexpressioninheadandnecksquamouscellcarcinoma.
JTranslMed.
2011;9:168.
24.
SrivastavaRM,etal.
Cetuximab-activatednatu-ralkilleranddendriticcellscollaboratetotriggertumorantigen-specificT-cellimmunityinheadandneckcancerpatients.
ClinCancerRes.
2013;19(7):1858–1872.
25.
MendelsohnJ,BaselgaJ.
Statusofepidermalgrowthfactorreceptorantagonistsinthebiol-ogyandtreatmentofcancer.
JClinOncol.
2003;21(14):2787–2799.
26.
DassonvilleO,etal.
Expressionofepidermalgrowthfactorreceptorandsurvivalinupperaerodigestivetractcancer.
JClinOncol.
1993;11(10):1873–1878.
27.
KohrtHE,etal.
CD137stimulationenhancestheantilymphomaactivityofanti-CD20antibodies.
Blood.
2011;117(8):2423–2432.
28.
KohrtHE,etal.
StimulationofnaturalkillercellswithaCD137-specificantibodyenhancestrastu-zumabefficacyinxenotransplantmodelsofbreastcancer.
JClinInvest.
2012;122(3):1066–1075.
29.
MeleroI,Hervas-StubbsS,GlennieM,PardollDM,ChenL.
Immunostimulatorymonoclonalantibodiesforcancertherapy.
NatRevCancer.
2007;7(2):95–106.
30.
WilcoxRA,etal.
Cuttingedge:Expressionoffunc-tionalCD137receptorbydendriticcells.
JImmunol.
2002;168(9):4262–4267.
31.
NarazakiH,ZhuY,LuoL,ZhuG,ChenL.
CD137agonistantibodypreventscancerrecurrence:contri-butionofCD137onbothhematopoieticandnon-hematopoieticcells.
Blood.
2010;115(10):1941–1948.
32.
BaesslerT,etal.
CD137ligandmediatesoppositeeffectsinhumanandmouseNKcellsandimpairsNKcellreactivityagainsthumanacutemyeloidleukemiacells.
Blood.
2010;115(15):3058–3069.
33.
MeleroI,JohnstonJV,ShuffordWW,MittlerRS,ChenL.
NK1.
1cellsexpress4-1BB(CDw137)costimulatorymoleculeandarerequiredfortumorimmunityelicitedbyanti-4-1BBmonoclonalanti-bodies.
CellImmunol.
1998;190(2):167–172.
34.
WilcoxRA,TamadaK,StromeSE,ChenL.
Signal-ingthroughNKcell-associatedCD137promotesbothhelperfunctionforCD8+cytolyticTcellsandresponsivenesstoIL-2butnotcytolyticactivity.
JImmunol.
2002;169(8):4230–4236.
35.
ManiarA,etal.
Human{gamma}{delta}Tlympho-cytesinducerobustNKcellmediatedantitumorcytotoxicitythroughCD137engagement.
Blood.
2010;116(10):1726–1733.
36.
GongW,etal.
ImmobilizedMHCclassIchain-relatedproteinAsynergizeswithIL-15andsoluble4-1BBligandtoexpandNKcellswithhighcytotoxicityexvivo.
CellMolImmunol.
2010;7(6):477–484.
37.
KohrtHE,etal.
CD137stimulationenhancestheanti-lymphomaactivityofanti-CD20antibodies.
Blood.
2010;117(8):2423–2432.
38.
CooperMA,FehnigerTA,CaligiuriMA.
Thebiol-ogyofhumannaturalkiller-cellsubsets.
TrendsImmunol.
2001;22(11):633–640.
2682TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June201439.
BowlesJA,WeinerGJ.
CD16polymorphismsandNKactivationinducedbymonoclonalantibody-coatedtargetcells.
JImmunolMethods.
2005;304(1–2):88–99.
40.
JuelkeK,etal.
CD62LexpressionidentifiesauniquesubsetofpolyfunctionalCD56dimNKcells.
Blood.
2010;116(8):1299–1307.
41.
ManiA,etal.
AphaseIItrialoftrastuzumabincombinationwithlow-doseinterleukin-2(IL-2)inpatients(PTS)withmetastaticbreastcancer(MBC)whohavepreviouslyfailedtrastuzumab.
BreastCan-cerResTreat.
2009;117(1):83–89.
42.
AcquavellaN,etal.
Toxicityandactivityofatwicedailyhigh-dosebolusinterleukin2regimeninpatientswithmetastaticmelanomaandmetastaticrenalcellcancer.
JImmunother.
2008;31(6):569–576.
43.
GluckWL,etal.
PhaseIstudiesofinterleukin(IL)-2andrituximabinB-cellnon-hodgkin'slymphoma:IL-2mediatednaturalkillercellexpansioncorrela-tionswithclinicalresponse.
ClinCancerRes.
2004;10(7):2253–2264.
44.
AnsellSM,etal.
Phase1studyofinterleukin-12incombinationwithrituximabinpatientswithB-cellnon-Hodgkinlymphoma.
Blood.
2002;99(1):67–74.
45.
HouotR,etal.
TherapeuticeffectofCD137immu-nomodulationinlymphomaanditsenhancementbyTregdepletion.
Blood.
2009;114(16):3431–3438.
46.
KohrtHE,etal.
CD137stimulationenhancestheanti-lymphomaactivityofanti-CD20antibodies.
Blood.
2011;117(8):2423–2432.
47.
GenovesiEV,PetteyCL,CollinsJJ.
UseofadoptivetransferandWinnassayproceduresinthefurtheranalysisofantiviralacquiredimmunityinmiceprotectedagainstFriendleukemiavirus-induceddiseasebypassiveserumtherapy.
CancerRes.
1984;44(4):1489–1498.
48.
JiangD,SchwarzH.
RegulationofgranulocyteandmacrophagepopulationsofmurinebonemarrowcellsbyG-CSFandCD137protein.
PLoSOne.
2010;5(12):e15565.
49.
ZhangX,etal.
CD137promotesproliferationandsurvivalofhumanBcells.
JImmunol.
2010;184(2):787–795.
50.
MurilloO,etal.
InvivodepletionofDCimpairstheanti-tumoreffectofagonisticanti-CD137mAb.
EurJImmunol.
2009;39(9):2424–2436.
51.
SharmaRK,etal.
4-1BBligandasaneffectivemul-tifunctionalimmunomodulatorandantigendeliv-eryvehicleforthedevelopmentoftherapeuticcan-cervaccines.
CancerRes.
2010;70(10):3945–3954.
52.
PiccioliD,SbranaS,MelandriE,ValianteNM.
Con-tact-dependentstimulationandinhibitionofden-driticcellsbynaturalkillercells.
JExpMed.
2002;195(3):335–341.
53.
WehnerR,DietzeK,BachmannM,SchmitzM.
Thebidirectionalcrosstalkbetweenhumandendriticcellsandnaturalkillercells.
JInnateImmun.
2011;3(3):258–263.
54.
OllionV,etal.
Tumorcell-derivedAgcross-presen-tationinhumanDCinducedbyNKcells:mecha-nismsandspecializationoftheBDCA-3+DCsub-set.
Presentedat:15thInternationalCongressofImmunology;August22–27,2013;Milan,Italy.
AbstractP1.
08.
18.
55.
GerosaF,Baldani-GuerraB,NisiiC,MarchesiniV,CarraG,TrinchieriG.
Reciprocalactivatinginter-actionbetweennaturalkillercellsanddendriticcells.
JExpMed.
2002;195(3):327–333.
56.
MailliardRB,etal.
DendriticcellsmediateNKcellhelpforTh1andCTLresponses:two-signalrequirementfortheinductionofNKcellhelperfunction.
JImmunol.
2003;171(5):2366–2373.
57.
AgaugueS,MarcenaroE,FerrantiB,MorettaL,MorettaA.
HumannaturalkillercellsexposedtoIL-2,IL-12,IL-18,orIL-4differentlymodulateprimingofnaiveTcellsbymonocyte-derivedden-driticcells.
Blood.
2008;112(5):1776–1783.
58.
MatsuoT,NishizukaSS,IshidaK,IwayaT,IkedaM,WakabayashiG.
Analysisoftheanti-tumoreffectofcetuximabusingproteinkineticsandmousexeno-graftmodels.
BMCResNotes.
2011;4:140.
59.
WilcoxRA,etal.
ProvisionofantigenandCD137signalingbreaksimmunologicalignorance,pro-motingregressionofpoorlyimmunogenictumors.
JClinInvest.
2002;109(5):651–659.
60.
KimGG,DonnenbergVS,DonnenbergAD,Good-ingW,WhitesideTL.
Anovelmultiparametricflowcytometry-basedcytotoxicityassaysimultaneouslyimmunophenotypeseffectorcells:comparisonstoa4h51Cr-releaseassay.
JImmunolMethods.
2007;325(1–2):51–66.
61.
KotechaN,etal.
Single-cellprofilingidentifiesaberrantSTAT5activationinmyeloidmalignan-cieswithspecificclinicalandbiologiccorrelates.
CancerCell.
2008;14(4):335–343.
62.
KoeneHR,KleijerM,AlgraJ,RoosD,vondemBorneAE,deHaasM.
FcγRIIIa-158V/Fpolymor-phisminfluencesthebindingofIgGbynaturalkillercellFcγRIIIa,independentlyoftheFcγRIIIa-48L/R/Hphenotype.
Blood.
1997;90(3):1109–1114.
jci.
orgVolume124Number6June2014TargetingCD137enhancestheefficacyofcetuximabHolbrookE.
Kohrt,1A.
DimitriosColevas,1RochHouot,1,2,3KippWeiskopf,1MatthewJ.
Goldstein,1PederLund,1AntoniaMueller,1IditSagiv-Barfi,1AurelienMarabelle,1,4RuthLira,1EmilyTroutner,1LoriRichards,1AmandaRajapaska,1JonathanHebb,1CariadChester,1ErinWaller,1AntonOstashko,1Wen-KaiWeng,1LiepingChen,5DebraCzerwinski,1Yang-XinFu,6JohnSunwoo,1andRonaldLevy11DepartmentofMedicine,DivisionofOncology,StanfordUniversity,Stanford,California,USA.
2CHURennes,ServiceHématologieClinique,Rennes,France.
3INSERM,U917,Rennes,France.
4CentredeRechercheenCancérologiedeLyon,UMRINSERMU1052,CNRS5268,CentreLéonBérard,UniversitédeLyon,Lyon,France.
5DepartmentofImmunobiology,YaleCancerCenter,NewHaven,Connecticut,USA.
6DepartmentofPathologyandCommitteeonImmunology,UniversityofChicago,Chicago,Illinois,USA.
Treatmentwithcetuximab,anEGFR-targetingIgG1mAb,resultsinbeneficial,yetlimited,clinicalimprove-mentforpatientswithheadandneck(HN)canceraswellascolorectalcancer(CRC)patientswithWTKRAStumors.
Antibody-dependentcell-mediatedcytotoxicity(ADCC)byNKcellscontributestotheefficacyofcetuximab.
ThecostimulatorymoleculeCD137(4-1BB)isexpressedfollowingNKandmemoryTcellactiva-tion.
WefoundthatisolatedhumanNKcellssubstantiallyincreasedexpressionofCD137whenexposedtocetuximab-coated,EGFR-expressingHNandCRCcelllines.
Furthermore,activationofCD137withanago-nisticmAbenhancedNKcelldegranulationandcytotoxicity.
Inmultiplemurinexenograftmodels,includ-ingEGFR-expressingcancercells,HNcells,andKRAS-WTandKRAS-mutantCRC,combinedcetuximabandanti-CD137mAbadministrationwassynergisticandledtocompletetumorresolutionandprolongedsurvival,whichwasdependentonthepresenceofNKcells.
Inpatientsreceivingcetuximab,thelevelofCD137oncir-culatingandintratumoralNKcellswasdependentonpostcetuximabtimeandhostFcyRIIIapolymorphism.
Interestingly,theincreaseinCD137-expressingNKcellsdirectlycorrelatedtoanincreaseinEGFR-specificCD8+Tcells.
TheseresultssupportdevelopmentofasequentialantibodyapproachagainstEGFR-expressingmalignanciesthatfirsttargetsthetumorandthenthehostimmunesystem.
IntroductionColorectalcarcinoma(CRC)isestimatedtoremainthethirdmostcommoncancer,affectingover140,000livesandleadingtoover50,000deathsintheUnitedStatesin2013alone.
Thoughcasesofheadandneck(HN)cancerare3-foldlesscommon,theincidencecontinuestoriseduetoagrowingpopulationinfectedwithhumanpapillomavirus(HPV)(1).
EGFRERBB1(avianerythroblasticleu-kemiaviral[v-erb-b]oncogenehomolog,receptorforEGF)isapoorprognosticmarkerinmultiplesolidtumors,includingCRC,HN,non–smallcelllung,andpancreaticcarcinomas,andisatherapeu-tictargetofmAbs(cetuximabandpanitumumab)aswellassmallmoleculekinaseinhibitors(erlotinibandgefitinib).
Cetuximab,ahuman-mousechimericIgG1mAb,wasapprovedbytheFoodandDrugAdministrationfirstin2004andtodayisindicatedforpatientswithCRCandHNcarcinomas(2).
However,responsestocetuximabmonotherapyarelimited;only1in5patientswithmetastaticCRC(3)andonly1in10withadvancedHNcarcino-marespondtotreatment(4,5).
Despiteexpressionofthetarget,patientswithnon–smallcelllung,pancreatic,andCRCwithcodon11or12KRASmutationsfailtobenefitfromcetuximab(6).
Givenmultiplemechanismsofcetuximab'saction,avarietyofapproacheshavebeenpursuedtoenhanceitsefficacy.
Bycompeti-tivelyblockingthebindingofEGF,phosphorylationandkinaseactivationareprevented,therebyinhibitingcellgrowth,inducingapoptosis,anddecreasingproductionofmatrixmetalloproteinaseandvascularendothelialgrowthfactor(7,8).
Inadditiontotheseinvitroeffects,invivoevidenceinbothmurinemodelsandpatientssuggestscetuximab'sefficacyisduetoantibody-dependentcell-mediatedcytotoxicity(ADCC),whichrequiresimmuneeffectorcells,mainlyNKcells,bindingviatheirFcreceptor(FcγRIII,CD16)totheIgG1Fc,heavy-chain,portionofcetuximab(9–13).
TargetingEGFRbysmallmoleculesthatlackanFc,andthereforelackADCC,hasnotresultedinaclinicalbenefitinHNorCRCs.
SupportingADCCasaprimarymechanismofcetuximab'sactivityinpatients,NKcellinfiltratewithinprimarycolorectaltumorsindependentlypredictsprognosis(14).
PatientswithcolorectalandHNcarcino-masharboringahigh-affinityFcγRIIIpolymorphismhavebeenshowntorespondmorefavorablytocetuximabbothexvivowithhighercytotoxicityagainstEGFR-expressingcelllines(15)andclin-icallywithsuperiordisease-freeandoverallsurvival(15–19).
There-fore,methodstoenhanceADCC,suchasstimulatingtheinnateimmuneresponse,mayclinicallytranslatetoimprovedantitumoractivity.
AugmentingtheNKcellresponsetocetuximabtherapymayenhancetheadaptiveimmuneresponseinadditiontoinnateimmunityduetoNKcell–DC"crosstalk,"whichleadstotumorantigen–specificTcellresponsesfollowingcetuximabtherapy(20).
Wesoughttoidentifyaninducibleandtargetablecostimula-torymoleculeonNKcellsinordertoenhanceADCC.
CD137(4-1BB)isupregulatedonhumanNKcellswhentheyencounterantibody-boundtumorcells(21).
Therefore,wehypothesizedthattheantitumorefficacyofcetuximabcouldbeimprovedthroughadualantibodystrategy:firstbyinducingCD137expressiononNKcellsupontheirexposuretocetuximab-boundtumorcellsandsubsequentlybytargetingactivatedNKcellswithanagonis-ticanti-CD137mAb.
Conflictofinterest:Theauthorshavedeclaredthatnoconflictofinterestexists.
Citationforthisarticle:JClinInvest.
2014;124(6):2668–2682.
doi:10.
1172/JCI73014.
RelatedCommentary,page2371TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142669ResultsCetuximabinducesCD137upregulationonhumanNKcellsfollowingincubationwithEGFR-positivetumorcells.
CD137expressionwasinducedonthesurfaceofNKcellsfromhealthyhumansubjectsfollowingincubationwithcetuximabandEGFR-expressingcan-cercelllines(SCC6,PC1,andSCC4)(Figure1A).
ThisCD137upregulationrequiredthepresenceofbothanEGFR-expressingcellandanEGFR-targetingmAb,aslittleeffectonCD137expres-sionwasobservedwithcetuximaborwithEGFR-expressingcan-cercelllinesalone.
Similarly,NKcellexpressionofCD137didnotincreasefollowingculturewithanon-EGFR–targetingmAb,rituximab,whichtargetsCD20,eveninthepresenceoftheEGFR-expressingcells(Figure1,BandC).
TheinductionofCD137occurredpreferentiallyinCD56dimcomparedwithCD56hiNKcellsandamongthissubsetwasassociatedwithaconcurrentdecreaseintheexpressionoftheFcγRIII(CD16)(Figure1,A–C).
Anti-CD137agonisticmAbincreasescetuximab-mediatedNKcellcytotoxicityontumorcellsandDCcytokinesecretion.
TodeterminewhetherCD137isapotentialtherapeutictargetforenhancingNKcellfunction,NKcellswerefirstactivatedtoexpressCD137bytheirexposuretoEGFR-expressingcancercellsandcetuximab.
ActivatedCD137-expressingNKcellswerethenreisolatedandtestedfortheirabilitytoperformADCCagainstEGFR-expressingcancercells(Fig-ure2,A–F).
ActivatedNKcellsshowedenhancedADCCfollowinganti-CD137mAbstimulation,asmeasuredbyapoptosis(Figure2,A–C)andchromiumrelease(Figure2,D–F)fromEGFR-expressingcancercells.
Thoughtheanti-CD137mAbenhancedcytotoxicity,specificallyADCC,enhancedIFN-γsecretion,anotherusualmea-sureofNKcellfunction,incontrast,wasnotobserved(Supple-mentalFigure1,A–C;supplementalmaterialavailableonlinewiththisarticle;doi:10.
1172/JCI73014DS1).
However,inthepresenceofactivatedNKcells,EGFR-expressingcancercells,cetuximab,andimmatureDCs(iDCs),anti-CD137agonisticmAbenhancedsecre-tionofIL-12,IFN-γ,andTNF-α(SupplementalFigure1,D–F).
Anti-CD137agonisticmAbenhancesantitumoractivityofcetuximabinvivoagainstbothKRAS-mutantandKRAS-WTtumors.
WetestedtheabilityofcetuximabtoinduceCD137expressiononmurineNKcellsinvivoinanEGFR-expressingxenotransplantmodel(SCC6).
Weadministeredcetuximab21daysfollowingimplantationoftumorandmeasuredtheexpressionofCD137onNKcellsintheperipheralblood,spleen,andtumor(Figure3A).
Ineachofthe3compartmentsandatseveraltimepoints,weobservedupregulatedexpressionofCD137onNKcells(Figure3,BandC).
Interestingly,atthetumorsite,CD137expressionpeakedat24hoursfollowingFigure1CetuximabinducesCD137upregulationonhumanNKcellsfollowingincubationwithEGFR-positivetumorcells.
PeripheralbloodfromhealthydonorswasanalyzedforCD137expressiononCD3–CD56+NKcellsafter24-hourculturewithEGFR-positivetumorcelllinesSCC6,PC1,andSCC4,andcetuximaborrituximab.
(A)PercentageofNKcellsdividedbyquadranttodelineatesubsetsofCD3–CD56brightandCD3–CD56dimexpressingCD137fromarepresentativehealthydonorafter24-hourculturewiththeEGFR-positivetumorcelllinePC1andcetuximab.
(B)PercentageofCD137expressiononNKcellsfrom3healthydonorsafter24-hourculturewiththeEGFR-positivetumorcelllineSCC6,PC1,orSCC4,andcetuximaborrituximab.
(C)CD16expressiononNKcellsfroma3healthydonorsafter24-hourculturewiththeEGFR-positivetumorcelllineSCC6,PC1,orSCC4,andcetuximaborrituximab.
*P90%purityasdefinedbyCD3–CD56+flowcytometry)andactivation(>50%expressionofCD137).
EGFR-expressingcancercelllinesincludingSCC6(AandD),PC1(BandE),andSCC4(CandF)wereculturedfor18hourswithpreactivated,purifiedNKcellsinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml).
CellswerewashedandincubatedwithannexinVand7-AADtodeterminepercentageofapoptotictumorcellsbyannexinVand7-AADstaining.
Chromium-labeledEGFR-expressingcancercelllineswereculturedfor4hourswithpreactivated,purifiedNKcellsinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs.
Shownispercentagelysisoftargetcellsbychromiumreleaseatvaryingeffector(activatedNKcells)/targetcellratiosculturedwithmediumalone(circles),anti-CD137(upside-downtriangle),cetux-imab(triangle),orcetuximabandanti-CD137(square)antibodies.
*P=0.
002,SCC6(A);*P=0.
007,PC1(B);*P=0.
007,SCC4(C);*P=0.
021,SCC6(D);*P=0.
011,PC1(E);*P=0.
049,SCC4(F).
TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142671wassuperiortoconcurrentcombinationdosingineffectonbothtumorgrowthandsurvival.
ThistherapeuticschemawastestedonbothKRAS-WT(T84)andKRAS-mutant(HCT116G13D)tumorsanddemonstratedasimilartherapeuticbenefittothesequentialcombinationofcetuximabfollowedbyanti-CD137mAb,withnearcompletetumorregressionandallanimalssurviving100daysaftertumorimplantation(Figure4,D–F).
Enhancementoftheantitumoractivityofcetuximabbyanti-CD137agonisticmAbisdependentonNKcellsandCD8+Tcells.
Theefficacyweobservedwithcombinationcetuximabandanti-CD137mAbtherapyinathymicmicestronglyimplicatestheinnateimmunesysteminthemechanismofaction.
Inordertodeterminewhethertheadaptiveimmuneresponsealsoplaysarole,weappliedthecombinationtherapytoasyngeneictumormodelinimmuno-competentBALB/cmice,usingatumorcellline,TUBO,trans-fectedwithhumanEGFR,previouslydeterminedtobesensitivetocetuximabtreatment(Figure5Aandref.
22).
ThiscelllineisHER2/neu-dependentandderivedfromBALB/cmicetransgenicfortheneuoncogene.
Inthismodel,anti-CD137mAbtherapyalonehadnoeffectontumorgrowthandcetuximabmonothera-pysignificantlyslowedtumorgrowth,whilethesequentialcom-binationofcetuximabfollowedbyanti-CD137mAbledtotumorregressionandimprovedsurvival,withallmiceinthisgroupaliveatday100(Figure5,BandC).
DepletionofCD4+Tcellsormacrophageshadnoinfluenceonefficacy(Figure5,DandE).
Ashypothesized,depletionofNKcellsalmostcompletelyabrogatedthetherapeuticbenefit.
Butsurpris-ingly,depletionofCD8+TcellspartiallyreducedtheefficacyoftheFigure3CetuximabmAbinducesCD137expressioninvivo.
nu/numicewereinoculatedwith1*106SCC6tumorcellss.
c.
ontheleftflank,andcetux-imabwasadministeredonday21aftertumorinoculation(A,schema).
(B)RepresentativeCD137expressiononCD3–DX5+cellsisolatedfromthespleenorperipheralblood24hoursfollowingcetuximabtreatmentorisotypecontrolinnon–tumor-bearingmice(n=3micepergroup).
(C)RepresentativeCD3–DX5+cellsisolatedfromthespleen,peripheralblood,ortumoranalyzedforCD137expression24,72,or168hoursfol-lowingcetuximabtreatmentorisotypecontrolintumor-bearingmice(n=3micepergroup).
ForBandC,numbersindicatepercentageofcellsaboveandbelowupperlimitofisotypecontrol.
2672TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June2014Figure4Anti-CD137agonisticmAbenhancesantitumoractivityofcetuximabinvivoagainstKRAS-mutantandKRAS-WTEGFR-expressingtumors.
nu/numicewereinoculatedwith1*106SCC6tumorcells(KRAS-WT)s.
c.
ontheleftflank,andcetuximaband/oranti-CD137mAbwasadminis-teredstartingonday21aftertumorinoculation(A,schema).
(BandC)Followingtumorinoculationonday0,micereceivedeitherratIgGcontrolonday21(circles),cetuximabonday21(squares),anti-CD137antibodyonday22(diamonds),cetuximabonday21andanti-CD137antibodyonday21(upside-downtriangles),orcetuximabonday21andanti-CD137antibodyonday22(triangles),witheachinjectionrepeatedweeklyforatotalof4injections.
Mice(10pergroup)werethenmonitoredfortumorgrowth(B)andoverallsurvival(C).
nu/numicewereinoculatedwith1*106T84(KRAS-WT)orHCT116(KRAS-mutant)tumorcellss.
c.
ontheleftflank,andcetuximaband/oranti-CD137mAbwasadministeredstartingonday21aftertumorinoculation(D,schema).
(EandF)Aftertumorinoculation,miceinoculatedwithHCT116thenreceivedeitherratIgGcontrolonday21(blackcircles)orcetuximabonday21andanti-CD137antibodyonday22(bluecircles),witheachinjectionrepeatedweeklyforatotalof4injections.
MiceinoculatedwithT84thenreceivedeitherratIgGcontrolonday21(blackdiamonds)orcetuximabonday21andanti-CD137antibodyonday22(reddiamonds),witheachinjectionrepeatedweeklyforatotalof4injections.
Mice(10pergroup)werethenmonitoredfortumorgrowth(E)andoverallsurvival(F).
*P50%expressionofCD137).
TheactivatedNKcellswerethenculturedwithadditionalEGFR-expressingcancercelllines(SCC6,PC1,orSCC4)ataratioof1:1.
After18hours,thesupernatantswereremovedandassayedforIFN-γbyELISA(HumanInterferon-γBDOptEIAELISASetandBDOptEIAReagentSetB;BDBiosciences—Pharmingen).
CellsfromthesecultureswereincubatedwithFITC–annexinVand7-AAD(bothBDBiosciences)andanalyzedbyflowcytometry.
ForwardandsidescatterwereusedtogateouttheNKcells,andfluorescencewasusedtodeter-minethepercentageofapoptotictumorcellsbyannexinVand7-AADstaining.
NKcellcytotoxicitywasadditionallymeasuredbyachromiumWehavestudiedpatientswithHNcarcinomareceivingthera-pywithcetuximab.
WefoundNKcellsincirculationandinthetumorsthathaveupregulatedCD137.
WehavealsofoundthatFcγRIIIpolymorphismscorrelatewiththemagnitudeofCD137upregulation,withincreasedCD137associatedwiththehigh-affinityalleles.
WeobservedasignificantincreaseinNKcellCD137expressionfollowingcetuximaboccurringwithin24hours;insomepatients,itwaselevatedaslongas1week.
Theincreasedexpressionintheperipheralbloodandwithinthetumorwashet-erogeneous,withasubsetofpatientsdemonstratingminimalupregulation.
ThoughtheongoingandplannedphaseIandI/IIclinicaltrialsdonotlimitenrollmentbasedonCD137expres-sion,wespeculatethatpatientswithminimalupregulationofCD137maynotbenefitfromanti-CD137mAbtherapy.
Toiden-tifypredictorsofCD137upregulation,theFcγRIIIpolymorphismofallpatientswastypedandstratifiedbypresenceofasinglehigh-affinityallele.
PatientswithHNcancerwitheitherV/VorV/FdemonstratedgreaterCD137upregulationcomparedwithpatientswith2low-affinityalleles.
Withcontinuedenrollmentinthisphase0study,wewillidentifyclinicalandpathologicchar-acteristicsthatpredicttheheterogeneityinCD137upregulation.
Bydoingso,wehopetocreateanomogrampredictiveofCD137expression,whichwithvalidationinongoingclinicaltrialsandcorrelationwithclinicalresponsetoanti-CD137mAbtherapy,mayprovideapredictivebiomarkerforpatientselectionandindi-viduallytailoredtreatmentschedules.
Bychangingtheparadigmofimprovingantibodytechnology—notbycreatingsuperiortumor-targetingmAbs,ratherbytarget-ingthesecondaryimmuneresponse—wehaveidentifiedathera-peuticstrategythatisselectivetothetumormicroenvironmentandefficaciousinmultiplemodels,includingthoseresistanttotreatment.
Theapproachisdependentuponsequentialtarget-ingoffirst,thetumor,bycetuximab,andsecond,theactivatedNKcells,byanti-CD137agonisticmAbs.
ThisstrategyinitiatesamorerobustinnateimmuneresponsewithaugmentedADCCaswellasadaptiveimmunitythroughlikelyenhancedcrosstalkbetweenNKcells,DCs,andtumorantigen–specificCD8+Tcells.
ByinvestigatingpredictorsofCD137expression,suchasFcγRIIIpolymorphism,inpatientsreceivingcetuximabmonotherapy,wehopetoidentifybiomarkersthatwillbetestedalongwiththecen-tralhypothesisofthisnewstrategyinaplannedcombinationtrialwithananti-CD137agonisticantibody.
MethodsCelllinesandculture.
ThehumanEGFR-expressingcancercelllinesSCC4,SCC6,andPC1wereobtainedfromJohnSunwooatStanfordUniversity.
SCC4isanupperaerodigestivetractsquamouscellcarcinomawithmuta-tionsinCDKN2A,NF1,andTP53andWTKRAS,EGFR,MET,NRAS,andPTEN.
SCC6isanupperaerodigestivetractsquamouscellcarcinomawithmutationsinCDKN2AandWTNF1,TP53,KRAS,EGFR,ET,NRAS,andPTEN.
PC1isapancreaticadenocarcinomawithmutationsinCDKN2A,KRAS(G12D),andTP53andWTEGFR,NRAS,MET,NF1,andPTEN.
HumanEGFR-positivecancercelllinesT84(KRAS-WT),HCT116(KRASG13Dmutant),andA20(alymphomacelllineusedasanegativecontrol)wereobtainedfromATCC.
T84isaCRCwithmutationsinAPCandTP53andWTNF1,KRAS,NRAS,MET,PTEN,andEGFR.
HCT116isaCRCwithmutationsinKRAS(G13D),APC,andTP53andWTNF1,NRAS,MET,PTEN,andEGFR.
TheTUBO-EGFRmurinecancercelllinewasobtainedfromYang-XinFuattheUniversityofChicago.
TheSCC4,SCC6,PC1,andT84celllineswereculturedina1:1mixtureofDMEM:F12supplemented2680TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June2014precipitateredcells.
Leukocyte-containingsupernatantwasremovedandcentrifuged,andtheremainingredcellswerelysedwithACKLysisBuffer(QualityBiological).
Splenocytesandsubcutaneoustumorsfrom3micepertreatmentgroupwereresected24,72,or168hoursfollowingcetux-imabtreatmentorisotypecontrolintumor-bearingmiceandmechani-callydigestedintoasingle-cellsuspension.
CellswerestainedwithmAbstoevaluateCD137expressiononNKcellsbyCD3–DX5+expressionbyflowcytometry.
SubcutaneousTUBOandTUBO-EGFRtumorsfrom5micepertreatmentgroupwereresectedonday24aftertumorinoculationandanalyzedforNKcell,Tcell,andDCinfiltrationbyflowcytometryofasingle-cellsuspensionfollowingmechanicaldigestion.
Thetumor-infil-tratingDCpopulationwasmarkedbyCD11c+andhighsurfaceexpres-sionofMHCclassIandclassIIproteinswithexpressionofcostimula-torymoleculesCD80(B7.
1)andCD86(B7.
2).
PBMCscollectedonday24aftertumorinoculationfrom5micepergroupinthe2tumormodelwerepurifiedasdescribedandthencoculturedwithmedium,1*106irradiatedA20,TUBO,orTUBO-EGFRcellsfor24hourswith0.
5mganti-mouseCD28mAb(BDBiosciences—Pharmingen)andinthepresenceofmonen-sin(Golgistop;BDBiosciences)forthelast5hoursat37°Cand5%CO2.
IntracellularIFN-γexpressionwasassessedwithBDCytofix/CytopermPlusKitperinstructions.
DepletionofCD4+andCD8+Tcells,NKcells,andmacrophages.
Anti-CD4mAb,anti-CD8mAb,anti-asialoGM1mAb(WakoChemicals),andlipo-somalclodronate(Weissmanlaboratory,StanfordUniversity)wereusedtodepleteCD4+Tcell,CD8+Tcell,NKcell,andmacrophageactivityinvivo,respectively.
AsciticfluidwasharvestedfromSCIDmicebearinghybrid-omasGK1.
5-andGK2.
43-producinganti-CD4(ratIgG2b)andanti-CD8(ratIgG2b)mAbs.
TheascitesweredilutedinsterilePBS.
Depletinganti-CD4mAb,anti-CD8mAb,andanti-asialoGM1mAbwereinjectedi.
p.
onday–1andday0oftumorinoculation,andevery5daysthereafteruntilday45atadoseof500μgperinjectionofanti-CD4mAboranti-CD8mAband50μlperinjectionofanti-asialoGM1mAb.
Liposomalclodronatewasinjectedretroorbitallyonday–2atadoseof200μlandonday0oftumorinoculation,followedbyevery4daysuntilday44atadoseof100μlperinjection.
Thedepletionconditionswerevalidatedbyflowcytometryofbloodshowingmorethan95%depletionofeachcellsubset.
Antibodiesforflowcytometry.
mAbstohumanantigenswereusedforstain-ingofhumanPBMCsincludingthefollowing:CD16FITC,CD4FITC,CD8FITC,CD137ligandPE,CD56PE,CD3PerCP,anti-EGFRAPC,CD137APC,APC-H7,FITC–annexinV,and7-AAD(allfromBD).
HIV-tetramerPEandEGFR-tetramer(YLN)PEwereprovidedbytheNIHTet-ramerCoreFacility(Atlanta,Georgia,USA)aswellasMarkDavis(Stan-fordUniversity).
ThefollowingmAbstomouseantigenswereused:CD8FITC(BD),CD4FITC(BD),CD137ligandPE(eBioscience),CD137PE(eBioscience),CD3PerCP(BD),DX5APC(eBioscience),CD11c-PE(BD),MHCIAPC(BD),MHCIIAPC(BD),B7.
1APC(BD),B7.
2FITC(BD),andF4/80APC(eBioscience).
StainedcellswerecollectedonaFACSCaliburoraLSRII3-lasercytometer(BD),anddatawereanalyzedusingCytobank(http://www.
cytobank.
org)(61).
AnalysisofFcγRIIIapolymorphisms.
GenomicDNAwaspreparedfromperipheralbloodmononuclearcellsusingaDNAextractionkit(QIAGEN).
GenotypingofFcγRIIIa158V/Fpolymorphismwasperformedbyapoly-merasechainreactionfollowedbyallele-specificrestrictionenzymediges-tion(62).
AllgenotypingofFcγRIIIapolymorphismwasconfirmedbydirectsequencingoftheregionofinterest.
Statistics.
Prismsoftware(GraphPad)wasusedtoanalyzetumorgrowthandcomparisonofcellpopulationstodeterminestatisticalsignificanceofdifferences(datapresentedasmean±SEM)betweengroupsbyapplyinga2-tailed,unpairedStudent'sttestor2-wayANOVAwithBonferroni'scor-rectionformultiplecomparisons.
Kaplan-Meierplotswereusedtoanalyzereleaseassay(60):targettumorcellswerelabeledwith150μCi51Crper1*106cellsfor2hours.
Activated,purified,andnonpurifiedNKcellswerepreparedasdescribedaboveandwerethenaddedatvariableeffec-tor/targetcellratiosfrom1:1to50:1.
Percentageoflysiswasdeterminedafter4hoursofcultureinthepresenceofanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml).
Allassayswereperformedintriplicatewith3independentNKcellsamples.
InvitroDCfunctionalassays.
AutologousiDCswerepreparedasprevi-ouslydescribed(52).
iDCpopulationswerecheckedforCD1aandCD80expressionandlackofCD14expression.
ActivatedandpurifiedNKcellswerepreparedasdescribedabove.
ActivationofNKcellswasconfirmedbyflowcytometry(>50%expressionofCD137).
TheactivatedNKcellswerethenculturedwithautologousiDCsandEGFR-expressingcancercelllines(SCC6,PC1,orSCC4)ataratioof1:1:1for18hoursinmediumaloneorwithanti-CD137mAb(BMS-663513,10μg/ml)alone,cetuximab(10μg/ml)alone,orcetuximabplusanti-CD137mAbs(bothat10μg/ml);supernatantwasharvestedandanalyzedbyELISAforIFN-γ,IL-12p40,andTNF-α(humanIFN-γ,IL-12p40,andTNR-αBDOptEIAELISASetsandBDOptEIAReagentSetB;BDPharmingen).
Allassayswereperformedintriplicatewith3independentNK:iDCcellsamples.
Tumortransplantationandantibodytherapy.
SCC6cancercellswereimplanteds.
c.
into5-to6-week-oldfemaleathymicnu/numiceatadoseof1*106cellsin50μlofPBS(BDBiosciences).
mAbwereadministeredbyi.
p.
injections.
Cetuximabwasgiveni.
p.
at200μgperinjection,andanti-CD137mAbwasgiveni.
p.
at150μgperinjectionatvarioustimesaftertumorinoculation.
TodeterminetheefficacyagainstKRAS-WTandmutanttumors,nu/numicewereinoculatedwith1*106T84(KRAS-WT)tumorcellsor1*106HCT116(KRASG13Dmutant)tumorcellss.
c.
ontheleftflank.
Todeterminetheimmunecellsubsetsnecessaryforefficacy,BALB/cmicewereinoculatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflank.
Cetuximabandanti-CD137mAbwereadministeredaftertumorinoculationasabove.
Tocharacterizetheadaptiveimmuneresponsefollowingcombinationtherapy,BALB/cmicewereinoculatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflankand1*106TUBO-EGFRtumorcellss.
c.
ontherightflank.
Cetuximabandanti-CD137mAbwereadministeredaftertumorinoculationasabove.
Tumorgrowthwasmea-suredbycalipertwiceaweekandexpressedastheproductoflengthbywidthinsquarecentimeters.
Miceweresacrificedwhentumorsizereached4cm2orwhentumorsitesulcerated.
Allinvivomodelswerepilotedwith5micepergroupandrepeatedwith10micepergroup,exceptinthe2-tumormodel,inwhich15micewereinoculatedand5pergroupsacri-ficedonday24fortumor-infiltratinglymphocyteanalysisandperipheralbloodcollectionforassessmentofCD8+Tcellfunctionexvivo.
Adoptiveimmunitytransferexperiments(Winnassay).
BALB/cmicewereinoc-ulatedwith1*106TUBO-EGFRtumorcellss.
c.
ontheleftflankandtreat-edwitheithercetuximabmonotherapyondays14,21,28,and35orsequen-tialcetuximabandanti-CD137mAbcombinationwithcetuximabdosedasinthemonotherapygroupandanti-CD137mAbonthesubsequentday.
Allmiceweresacrificed50daysafterbeinginoculatedwithtumorcells.
TotalorCD8-selectedsuspensionswerepreparedbyCD8a+TcellIsolationKit(MiltenyiBiotecGmbH)andpurificationassessedbyflowcytometricanalysisandconfirmedasmorethan95%.
Totalorinvitro–purifiedCD8+splenocyteswereadmixedwithTUBOorTUBO-EGFRtumorcells(100:1ratio)andcoinjectedintonaiveBALB/cmice.
Theresultswereexpressedaspercentageofanimalsthatweretumor-freeovertime.
DetectionofCD137-expressingcellsinvivo,tumor-infiltratingimmunecellsinvivo,andIFN-γproductionassayexvivo.
Bloodwascollectedfromthetailvein,anticoagulatedwith2mMEDTAinPBS,thendiluted1:1with2%DextranT500(Pharmacosmos)inPBSandincubatedat37°Cfor45minutestoTheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June20142681ardHughesMedicalInstituteResearchTrainingFellowship.
R.
HouotissupportedbyfellowshipsfromtheFoundationdeFranceandtheAssociationpourlaRecherchesurleCancer.
H.
E.
KohrtisaDamonRunyonClinicalInvestigatorandissupportedbyfellowshipsfromtheAmericanSocietyofHematologyandtheAmericanSocietyofClinicalOncologyandDepartmentofDefenseBreastCancerpostdoctoralfellowshipaward(26687).
K.
WeiskopfissupportedbytheStanfordUniversityMedicalSci-entistTrainingProgram.
R.
LevyisanAmericanCancerSocietyClinicalResearchProfessor.
ReceivedforpublicationSeptember3,2013,andacceptedinrevisedformMarch6,2014.
Addresscorrespondenceto:HolbrookKohrt,DivisionofOncol-ogy,269CampusDrive,CCSR1105,StanfordUniversityMedi-calCenter,Stanford,California94305-5151,USA.
Phone:650.
725.
6452;Fax:650.
736.
1454;E-mail:Kohrt@stanford.
edu.
survival.
Comparisonsofsurvivalcurvesweremadeusingthelog-ranktest.
CorrelationcoefficientswerecalculatedforcomparisonofpercentageofEGFR+CD8+TcellsandpercentageofCD137+NKcells(datapresentedasmean±SEM).
P<0.
05wasconsideredsignificant.
Fortumorburdens,comparisonsofmeansweredonebyANOVA.
Studyapproval.
HumanperipheralbloodandprimarybreasttumorsampleswereobtainedunderareviewedprotocolapprovedbyStanfordUniversity'sAdministrativePanelsonHumanSubjects(NCT01114256)andfollowinginformedconsentfrompatientsinaccordancewiththeDec-larationofHelsinki.
AllanimalexperimentswereapprovedbytheStanfordAdministrativePanelonLaboratoryAnimalCareandconductedinaccor-dancewithNIHguidelines.
AcknowledgmentsThisworkwassupportedbytheCaliforniaBreastCancerResearchProgram,EUREKA(R01CA153248-01),theReliableCancerTherapyFund,andNadia'sGiftFoundationattheStan-fordCancerCenter.
M.
J.
GoldsteinissupportedbytheHow-1.
AmericanCancerSociety.
CancerFactsandFigures2013.
Atlanta,Georgia,USA:AmericanCancerSociety;2013.
2.
LeeCM,TannockIF.
Thedistributionofthethera-peuticmonoclonalantibodiescetuximabandtrastuzumabwithinsolidtumors.
BMCCancer.
2010;10:255.
3.
DeRoockW,DeVriendtV,NormannoN,Ciardi-elloF,TejparS.
KRAS,BRAF,PIK3CA,andPTENmutations:implicationsfortargetedtherapiesinmetastaticcolorectalcancer.
LancetOncol.
2011;12(6):594–603.
4.
BaselgaJ,etal.
PhaseIImulticenterstudyoftheantiepidermalgrowthfactorreceptormonoclo-nalantibodycetuximabincombinationwithplatinum-basedchemotherapyinpatientswithplatinum-refractorymetastaticand/orrecurrentsquamouscellcarcinomaoftheheadandneck.
JClinOncol.
2005;23(24):5568–5577.
5.
VermorkenJB,etal.
Open-label,uncontrolled,mul-ticenterphaseIIstudytoevaluatetheefficacyandtoxicityofcetuximabasasingleagentinpatientswithrecurrentand/ormetastaticsquamouscellcar-cinomaoftheheadandneckwhofailedtorespondtoplatinum-basedtherapy.
JClinOncol.
2007;25(16):2171–2177.
6.
LiuL,CaoY,TanA,LiaoC,GaoF.
Cetuximab-basedtherapyversusnon-cetuximabtherapyforadvancedcancer:ameta-analysisof17randomizedcontrolledtrials.
CancerChemotherPharmacol.
2010;65(5):849–861.
7.
LiX,LuY,LiangK,PanT,MendelsohnJ,FanZ.
Requirementofhypoxia-induciblefactor-1alphadown-regulationinmediatingtheantitumoractiv-ityoftheanti-epidermalgrowthfactorreceptormonoclonalantibodycetuximab.
MolCancerTher.
2008;7(5):1207–1217.
8.
HuangSM,LiJ,HarariPM.
Molecularinhibitionofangiogenesisandmetastaticpotentialinhumansquamouscellcarcinomasafterepidermalgrowthfactorreceptorblockade.
MolCancerTher.
2002;1(7):507–514.
9.
ClynesRA,TowersTL,PrestaLG,RavetchJV.
InhibitoryFcreceptorsmodulateinvivocytoxicityagainsttumortargets.
NatMed.
2000;6(4):443–446.
10.
KuraiJ,etal.
Antibody-dependentcellularcytotoxicitymediatedbycetuximabagainstlungcan-cercelllines.
ClinCancerRes.
2007;13(5):1552–1561.
11.
SteinerP,etal.
Tumorgrowthinhibitionwithcetuximabandchemotherapyinnon-smallcelllungcancerxenograftsexpressingwild-typeandmutatedepidermalgrowthfactorreceptor.
ClinCancerRes.
2007;13(5):1540–1551.
12.
RabenD,etal.
Theeffectsofcetuximabaloneandincombinationwithradiationand/orchemotherapyinlungcancer.
ClinCancerRes.
2005;11(2pt1):795–805.
13.
WilsonNS,etal.
AnFcγreceptor-dependentmechanismdrivesantibody-mediatedtarget-receptorsignalingincancercells.
CancerCell.
2011;19(1):101–113.
14.
MarechalR,etal.
PutativecontributionofCD56positivecellsincetuximabtreatmentefficacyinfirst-linemetastaticcolorectalcancerpatients.
BMCCancer.
2010;10:340.
15.
Lopez-AlbaiteroA,etal.
RoleofpolymorphicFcγreceptorIIIaandEGFRexpressionlevelincetuximabmediated,NKcelldependentinvitrocytotoxicityofheadandnecksquamouscellcarcinomacells.
CancerImmunolImmunother.
2009;58(11):1853–1864.
16.
BibeauF,etal.
ImpactofFc{gamma}RIIa-Fc{gamma}RIIIapolymorphismsandKRASmuta-tionsontheclinicaloutcomeofpatientswithmet-astaticcolorectalcancertreatedwithcetuximabplusirinotecan.
JClinOncol.
2009;27(7):1122–1129.
17.
FerrisRL,JaffeeEM,FerroneS.
Tumorantigen-tar-geted,monoclonalantibody-basedimmunothera-py:clinicalresponse,cellularimmunity,andimmu-noescape.
JClinOncol.
2010;28(28):4390–4399.
18.
QiuLX,etal.
PredictiveandprognosticvalueofKRASmutationsinmetastaticcolorectalcancerpatientstreatedwithcetuximab:ameta-analysisof22studies.
EurJCancer.
2010;46(15):2781–2787.
19.
RodriguezJ,etal.
FcγreceptorpolymorphismsaspredictivemarkersofCetuximabefficacyinepider-malgrowthfactorreceptordownstream-mutatedmetastaticcolorectalcancer.
EurJCancer.
2012;48(12):1774–1780.
20.
LeeSC,SrivastavaRM,Lopez-AlbaiteroA,FerroneS,FerrisRL.
Naturalkiller(NK):dendriticcell(DC)crosstalkinducedbytherapeuticmonoclonalanti-bodytriggerstumorantigen-specificTcellimmu-nity.
ImmunolRes.
2011;50(2–3):248–254.
21.
LinW,etal.
Fc-dependentexpressionofCD137onhumanNKcells:insightsinto"agonistic"effectsofanti-CD137monoclonalantibodies.
Blood.
2008;112(3):699–707.
22.
YangX,ZhangX,MortensonED,Radkevich-BrownO,WangY,FuYX.
Cetuximab-mediatedtumorregressiondependsoninnateandadaptiveimmuneresponses.
MolTher.
2013;21(1):91–100.
23.
SchulerPJ,etal.
EGFR-specificTcellfrequenciescorrelatewithEGFRexpressioninheadandnecksquamouscellcarcinoma.
JTranslMed.
2011;9:168.
24.
SrivastavaRM,etal.
Cetuximab-activatednatu-ralkilleranddendriticcellscollaboratetotriggertumorantigen-specificT-cellimmunityinheadandneckcancerpatients.
ClinCancerRes.
2013;19(7):1858–1872.
25.
MendelsohnJ,BaselgaJ.
Statusofepidermalgrowthfactorreceptorantagonistsinthebiol-ogyandtreatmentofcancer.
JClinOncol.
2003;21(14):2787–2799.
26.
DassonvilleO,etal.
Expressionofepidermalgrowthfactorreceptorandsurvivalinupperaerodigestivetractcancer.
JClinOncol.
1993;11(10):1873–1878.
27.
KohrtHE,etal.
CD137stimulationenhancestheantilymphomaactivityofanti-CD20antibodies.
Blood.
2011;117(8):2423–2432.
28.
KohrtHE,etal.
StimulationofnaturalkillercellswithaCD137-specificantibodyenhancestrastu-zumabefficacyinxenotransplantmodelsofbreastcancer.
JClinInvest.
2012;122(3):1066–1075.
29.
MeleroI,Hervas-StubbsS,GlennieM,PardollDM,ChenL.
Immunostimulatorymonoclonalantibodiesforcancertherapy.
NatRevCancer.
2007;7(2):95–106.
30.
WilcoxRA,etal.
Cuttingedge:Expressionoffunc-tionalCD137receptorbydendriticcells.
JImmunol.
2002;168(9):4262–4267.
31.
NarazakiH,ZhuY,LuoL,ZhuG,ChenL.
CD137agonistantibodypreventscancerrecurrence:contri-butionofCD137onbothhematopoieticandnon-hematopoieticcells.
Blood.
2010;115(10):1941–1948.
32.
BaesslerT,etal.
CD137ligandmediatesoppositeeffectsinhumanandmouseNKcellsandimpairsNKcellreactivityagainsthumanacutemyeloidleukemiacells.
Blood.
2010;115(15):3058–3069.
33.
MeleroI,JohnstonJV,ShuffordWW,MittlerRS,ChenL.
NK1.
1cellsexpress4-1BB(CDw137)costimulatorymoleculeandarerequiredfortumorimmunityelicitedbyanti-4-1BBmonoclonalanti-bodies.
CellImmunol.
1998;190(2):167–172.
34.
WilcoxRA,TamadaK,StromeSE,ChenL.
Signal-ingthroughNKcell-associatedCD137promotesbothhelperfunctionforCD8+cytolyticTcellsandresponsivenesstoIL-2butnotcytolyticactivity.
JImmunol.
2002;169(8):4230–4236.
35.
ManiarA,etal.
Human{gamma}{delta}Tlympho-cytesinducerobustNKcellmediatedantitumorcytotoxicitythroughCD137engagement.
Blood.
2010;116(10):1726–1733.
36.
GongW,etal.
ImmobilizedMHCclassIchain-relatedproteinAsynergizeswithIL-15andsoluble4-1BBligandtoexpandNKcellswithhighcytotoxicityexvivo.
CellMolImmunol.
2010;7(6):477–484.
37.
KohrtHE,etal.
CD137stimulationenhancestheanti-lymphomaactivityofanti-CD20antibodies.
Blood.
2010;117(8):2423–2432.
38.
CooperMA,FehnigerTA,CaligiuriMA.
Thebiol-ogyofhumannaturalkiller-cellsubsets.
TrendsImmunol.
2001;22(11):633–640.
2682TheJournalofClinicalInvestigationhttp://www.
jci.
orgVolume124Number6June201439.
BowlesJA,WeinerGJ.
CD16polymorphismsandNKactivationinducedbymonoclonalantibody-coatedtargetcells.
JImmunolMethods.
2005;304(1–2):88–99.
40.
JuelkeK,etal.
CD62LexpressionidentifiesauniquesubsetofpolyfunctionalCD56dimNKcells.
Blood.
2010;116(8):1299–1307.
41.
ManiA,etal.
AphaseIItrialoftrastuzumabincombinationwithlow-doseinterleukin-2(IL-2)inpatients(PTS)withmetastaticbreastcancer(MBC)whohavepreviouslyfailedtrastuzumab.
BreastCan-cerResTreat.
2009;117(1):83–89.
42.
AcquavellaN,etal.
Toxicityandactivityofatwicedailyhigh-dosebolusinterleukin2regimeninpatientswithmetastaticmelanomaandmetastaticrenalcellcancer.
JImmunother.
2008;31(6):569–576.
43.
GluckWL,etal.
PhaseIstudiesofinterleukin(IL)-2andrituximabinB-cellnon-hodgkin'slymphoma:IL-2mediatednaturalkillercellexpansioncorrela-tionswithclinicalresponse.
ClinCancerRes.
2004;10(7):2253–2264.
44.
AnsellSM,etal.
Phase1studyofinterleukin-12incombinationwithrituximabinpatientswithB-cellnon-Hodgkinlymphoma.
Blood.
2002;99(1):67–74.
45.
HouotR,etal.
TherapeuticeffectofCD137immu-nomodulationinlymphomaanditsenhancementbyTregdepletion.
Blood.
2009;114(16):3431–3438.
46.
KohrtHE,etal.
CD137stimulationenhancestheanti-lymphomaactivityofanti-CD20antibodies.
Blood.
2011;117(8):2423–2432.
47.
GenovesiEV,PetteyCL,CollinsJJ.
UseofadoptivetransferandWinnassayproceduresinthefurtheranalysisofantiviralacquiredimmunityinmiceprotectedagainstFriendleukemiavirus-induceddiseasebypassiveserumtherapy.
CancerRes.
1984;44(4):1489–1498.
48.
JiangD,SchwarzH.
RegulationofgranulocyteandmacrophagepopulationsofmurinebonemarrowcellsbyG-CSFandCD137protein.
PLoSOne.
2010;5(12):e15565.
49.
ZhangX,etal.
CD137promotesproliferationandsurvivalofhumanBcells.
JImmunol.
2010;184(2):787–795.
50.
MurilloO,etal.
InvivodepletionofDCimpairstheanti-tumoreffectofagonisticanti-CD137mAb.
EurJImmunol.
2009;39(9):2424–2436.
51.
SharmaRK,etal.
4-1BBligandasaneffectivemul-tifunctionalimmunomodulatorandantigendeliv-eryvehicleforthedevelopmentoftherapeuticcan-cervaccines.
CancerRes.
2010;70(10):3945–3954.
52.
PiccioliD,SbranaS,MelandriE,ValianteNM.
Con-tact-dependentstimulationandinhibitionofden-driticcellsbynaturalkillercells.
JExpMed.
2002;195(3):335–341.
53.
WehnerR,DietzeK,BachmannM,SchmitzM.
Thebidirectionalcrosstalkbetweenhumandendriticcellsandnaturalkillercells.
JInnateImmun.
2011;3(3):258–263.
54.
OllionV,etal.
Tumorcell-derivedAgcross-presen-tationinhumanDCinducedbyNKcells:mecha-nismsandspecializationoftheBDCA-3+DCsub-set.
Presentedat:15thInternationalCongressofImmunology;August22–27,2013;Milan,Italy.
AbstractP1.
08.
18.
55.
GerosaF,Baldani-GuerraB,NisiiC,MarchesiniV,CarraG,TrinchieriG.
Reciprocalactivatinginter-actionbetweennaturalkillercellsanddendriticcells.
JExpMed.
2002;195(3):327–333.
56.
MailliardRB,etal.
DendriticcellsmediateNKcellhelpforTh1andCTLresponses:two-signalrequirementfortheinductionofNKcellhelperfunction.
JImmunol.
2003;171(5):2366–2373.
57.
AgaugueS,MarcenaroE,FerrantiB,MorettaL,MorettaA.
HumannaturalkillercellsexposedtoIL-2,IL-12,IL-18,orIL-4differentlymodulateprimingofnaiveTcellsbymonocyte-derivedden-driticcells.
Blood.
2008;112(5):1776–1783.
58.
MatsuoT,NishizukaSS,IshidaK,IwayaT,IkedaM,WakabayashiG.
Analysisoftheanti-tumoreffectofcetuximabusingproteinkineticsandmousexeno-graftmodels.
BMCResNotes.
2011;4:140.
59.
WilcoxRA,etal.
ProvisionofantigenandCD137signalingbreaksimmunologicalignorance,pro-motingregressionofpoorlyimmunogenictumors.
JClinInvest.
2002;109(5):651–659.
60.
KimGG,DonnenbergVS,DonnenbergAD,Good-ingW,WhitesideTL.
Anovelmultiparametricflowcytometry-basedcytotoxicityassaysimultaneouslyimmunophenotypeseffectorcells:comparisonstoa4h51Cr-releaseassay.
JImmunolMethods.
2007;325(1–2):51–66.
61.
KotechaN,etal.
Single-cellprofilingidentifiesaberrantSTAT5activationinmyeloidmalignan-cieswithspecificclinicalandbiologiccorrelates.
CancerCell.
2008;14(4):335–343.
62.
KoeneHR,KleijerM,AlgraJ,RoosD,vondemBorneAE,deHaasM.
FcγRIIIa-158V/Fpolymor-phisminfluencesthebindingofIgGbynaturalkillercellFcγRIIIa,independentlyoftheFcγRIIIa-48L/R/Hphenotype.
Blood.
1997;90(3):1109–1114.
- fectedww.4399.com相关文档
- 厦门ww.4399.com
- 厦门ww.4399.com
- restww.4399.com
- tumorsww.4399.com
- periodontalww.4399.com
- thermofisher.com/contactus
云雀云(larkyun)低至368元/月,广州移动1Gbps带宽VDS(带100G防御),常州联通1Gbps带宽VDS
云雀云(larkyun)当前主要运作国内线路的机器,最大提供1Gbps服务器,有云服务器(VDS)、也有独立服务器,对接国内、国外的效果都是相当靠谱的。此外,还有台湾hinet线路的动态云服务器和静态云服务器。当前,larkyun对广州移动二期正在搞优惠促销!官方网站:https://larkyun.top付款方式:支付宝、微信、USDT广移二期开售8折折扣码:56NZVE0YZN (试用于常州联...
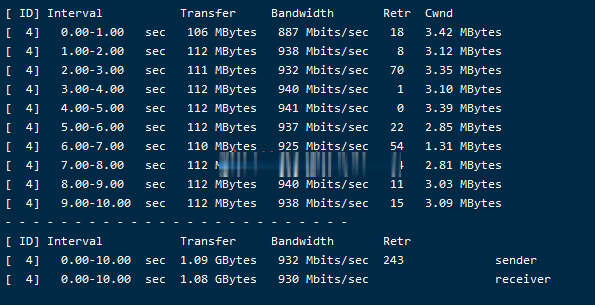
百驰云(19/月),高性能服务器,香港三网CN2 2核2G 10M 国内、香港、美国、日本、VPS、物理机、站群全站7.5折,无理由退换,IP免费换!
百驰云成立于2017年,是一家新国人IDC商家,且正规持证IDC/ISP/CDN,商家主要提供数据中心基础服务、互联网业务解决方案,及专属服务器租用、云服务器、云虚拟主机、专属服务器托管、带宽租用等产品和服务。百驰云提供源自大陆、香港、韩国和美国等地骨干级机房优质资源,包括BGP国际多线网络,CN2点对点直连带宽以及国际顶尖品牌硬件。专注为个人开发者用户,中小型,大型企业用户提供一站式核心网络云端...

腾讯云CVM云服务器大硬盘方案400GB和800GB数据盘方案
最近看到群里的不少网友在搭建大数据内容网站,内容量有百万篇幅,包括图片可能有超过50GB,如果一台服务器有需要多个站点的话,那肯定默认的服务器50GB存储空间是不够用的。如果单独在购买数据盘会成本提高不少。这里我们看到腾讯云促销活动中有2款带大数据盘的套餐还是比较实惠的,一台是400GB数据盘,一台是800GB数据盘,适合他们的大数据网站。 直达链接 - 腾讯云 大数据盘套餐服务器这里我们看到当前...
ww.4399.com为你推荐
-
阿丽克丝·布莱肯瑞吉阿丽克斯布莱肯瑞吉演的美国恐怖故事哪两集bbs.99nets.com送点卷的冒险岛私服5xoy.comhttp://www.5yau.com (舞与伦比),以前是这个地址,后来更新了,很长时间没玩了,谁知道现在的地址? 谢谢,se95se.comwww.sea8.com这个网站是用什么做的 需要多少钱103838.com39052.com这电影网支持网页观看吗?avtt4.comwww.51kao4.com为什么进不去啊?dadi.tv智能网络电视smartTV是什么牌子59ddd.comarmada m300什么装系统www.jizzbo.comwww.toubai.com是什么网站bihaiyinsha碧海银沙中国十大网页?