Dendriticstablehost
stablehost 时间:2021-01-03 阅读:()
Three-dimensionalstablelithiummetalanodewithnanoscalelithiumislandsembeddedinionicallyconductivesolidmatrixDingchangLina,JieZhaoa,JieSuna,HongbinYaoa,YayuanLiua,KaiYana,andYiCuia,b,1aDepartmentofMaterialsScienceandEngineering,StanfordUniversity,Stanford,CA94305;andbStanfordInstituteforMaterialsandEnergySciences,SLACNationalAcceleratorLaboratory,MenloPark,CA94025EditedbyThomasE.
Mallouk,ThePennsylvaniaStateUniversity,UniversityPark,PA,andapprovedMarch23,2017(receivedforreviewNovember25,2016)Rechargeablebatteriesbasedonlithium(Li)metalchemistryareattractivefornext-generationelectrochemicalenergystorage.
Nev-ertheless,excessivedendritegrowth,infiniterelativedimensionchange,severesidereactions,andlimitedpoweroutputseverelyimpedetheirpracticalapplications.
Althoughexcitingprogresshasbeenmadetosolvepartsoftheaboveissues,aversatilesolutionisstillabsent.
Here,aLi-ionconductiveframeworkwasdevelopedasastable"host"andefficientsurfaceprotectiontoaddressthemulti-facetedproblems,whichisasignificantstepforwardcomparedwithprevioushostconcepts.
Thiswasfulfilledbyreactingoverstoichiom-etryofLiwithSiO.
Theas-formedLixSi–Li2Omatrixwouldnotonlyenableconstantelectrode-levelvolume,butalsoprotecttheembed-dedLifromdirectexposuretoelectrolyte.
BecauseuniformLinucle-ationanddepositioncanbefulfilledowingtothehigh-densityactiveLidomains,theas-obtainednanocompositeelectrodeexhibitslowpolarization,stablecycling,andhigh-poweroutput(upto10mA/cm2)evenincarbonateelectrolytes.
TheLi–Sprototypecellsfurtherexhibitedhighlyimprovedcapacityretentionunderhigh-powerop-eration(600mAh/gat6.
69mA/cm2).
Theall-aroundimprovementonelectrochemicalperformanceshedslightontheeffectivenessofthedesignprinciplefordevelopingsafeandstableLimetalanodes.
Limetal|3Dcomposite|electrolyteproof|overlithiation|high-poweroutputExtensiveresearchhasbeenconductedondevelopingad-vancedlithium(Li)batteryanodesubstitutes,includingsili-con(1–5),tin(6,7),andLimetal(8–15),forhigh-energyapplicationsinportableelectronicsandelectricaltransportation.
Amongall,Limetalisrecognizedasthemostattractivecandidate,whereitslowestelectrochemicalpotential(Li+/Li=3.
040Vvs.
standardhydrogenelectrode)andhighesttheoreticalspecificcapacity(3,860mAh/g)haveattractedbroadscientificandtechnologicalinterest(16–18).
DespitecontinuouseffortsonLimetal,itspracticalapplicationhasremainedstagnantduetomanymaterialschallenges.
Therootcauseslieintwoaspects:thehighchemicalreactivityofLimetalandsig-nificantdimensionalchangeduringcharge/discharge,whichtogethergenerateotherobservedproblemsincludingsolidelectrolyteinter-phase(SEI)fractureanddendriticdeposition.
SincetheadventofLibatteries,greatprogresshasbeenmadeinunderstandingLimetalplating/strippingbehavior(8,9,12,19,20),preventingLidendriteformationandpenetration(13,21–25),aswellasimprovingCoulombicefficiency(CE)(14,26,27).
Al-thoughfurtherimprovementinionicconductivityandinterfacialcontactisstillrequired,solidelectrolyteswereproventobeeffi-caciousformitigatingdendritepenetration(22,23,28–33).
De-velopingelectrolyteadditivesisalsoeffectiveinstabilizingSEIandhomogenizingLideposition(13,26,27,34),whereasengineeringchemicallyandmechanicallystableinterfaciallayerisalsoaprominentalternativetostabilizeSEI(14).
AlthoughthehighreactivityofLimetalhasreceivedsufficientattention,thestudieswerebasedondenseLifoil,whichimposedgrandchallengesbyintroducinglargedimensionalchange.
Werecentlyproposedthatvolumevariationisoneofthetworootcausesattractingmuchrecentattention.
Incontrasttoacarbonaceousanodewith10%volumechangeduringcycling,"hostless"Lifoilinprincipleexhibitsinfiniterelativevolumechangeduringthestripping/platingcycle(35–37).
Ononehand,theenormousvolumechangegeneratesengineeringchallengesatthecelllevelbyintroducinginternalstress.
Ontheotherhand,SEIisvulnerabletothemovinginterfaceandpronetofracture(19,38,39).
ContinuousSEIformationduringcyclingwouldnotonlydecomposeelectrolyteandconsumeLiionbutalsoaccumulateanionicinsulatinglayerwhichfurtherblockstheiontransport(40).
Moreover,dendriticandmossyLiarisenfromunevenLi-ionfluxmightpenetratethroughtheseparatorandcauseaninternalshortcircuit,triggeringthedangerofthermalrunawayandexplosionhazards(18).
Duringthestrippingprocess,dendriticmorphologycanaggravatetheformationofdeadLiandthusresultinlowCE(9,41).
Moreover,duetothelimitedaccessibleactivesurfaceofplanarLifoil,practicalpoweroperation(5–10mA/cm2ofcurrentdensity)withLimetalanodesisnotpossibleatpresent(16).
Toaddresstheseissues,anewdirectionwasrecentlyde-velopedbasedontheconceptofconstructinga3DstablehosttostoreLimetal,whichwasrealizedwithgrapheneoxide(35),nanofibermatrix(36,37),andhollowcarbonspheres(42).
Theas-obtainedcompositeelectrodesaffordminimizedvolumechangetolessthan20%andthusmorestableSEI.
Inaddition,bycreatingalargesurfaceincontactwithliquidelectrolytes,ionfluxcanbehomogenizedsothathigh-poweranddendrite-freeoperationcanbeachieved.
Withtheremarkableimprovementinperformancedemonstratedbytheseexamples,webelievethataSignificanceLithiummetalanodeholdsgreatpromisesfornext-generationhigh-energylithiumbatterysystems.
Thisworkintroducesanelectrolyte-proofdesignofthree-dimensionallithiummetalanodewheremostofthelithiumdomainsareembeddedinalithium-ionconductivematrix.
Inthisarchitecture,thelithium-ionconductivematrixcanisolatetheembeddedlithiumdo-mainsfromliquidelectrolyteandthuspreventsevereinitialsidereactions,whilethematrixcansimultaneouslytransportlithiumionandmaintaintheelectrochemicalactivityoftheembeddedlithium.
Thedesignprincipleenableshighlystable,high-power,andsafelithiummetalanodeswithminimalsidereactionsandnegligiblevolumevariationduringcycling,whichpavesthewayforviablelithiummetalbatteriesinthefuture.
Authorcontributions:D.
L.
andY.
C.
designedresearch;D.
L.
,J.
Z.
,J.
S.
,H.
Y.
,andY.
L.
per-formedresearch;D.
L.
andJ.
Z.
contributednewreagents/analytictools;D.
L.
,K.
Y.
,andY.
C.
analyzeddata;andD.
L.
andY.
C.
wrotethepaper.
Theauthorsdeclarenoconflictofinterest.
ThisarticleisaPNASDirectSubmission.
1Towhomcorrespondenceshouldbeaddressed.
Email:yicui@stanford.
edu.
Thisarticlecontainssupportinginformationonlineatwww.
pnas.
org/lookup/suppl/doi:10.
1073/pnas.
1619489114/-/DCSupplemental.
www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114PNAS|May2,2017|vol.
114|no.
18|4613–4618CHEMISTRYDownloadedbyguestonDecember22,2020significantstepforwardistoconstructaLi-ionconductivestablehostasmatrixwithembeddedLimetaltoformthe3Danode.
Ratherthanhavinghigh-surfaceLiindirectcontactwithliquidelectrolyte,whichwouldgeneratemoreSEIattheearlystage,embeddingLidomainsinsideaLi-ionconductingmatrixwouldprotectthehighlyreactiveLifromexcesssidereactionsattheearlystage,whilesimultaneouslyguaranteeingtheelectrochemicalac-tivityoftheLi.
Wewouldexpectsuchaconcepttoholdnotonlythemeritsofprevious3Danodes,butalsotoprovideapreengineeredLisurfaceprotectiontoalleviatetheinitialsidereactions.
Herein,wedevelopedafacilechemicalsynthesismethodtore-alizesuchaLi-ionconductivenanocompositeelectrode(LCNE).
ByhavingSiOreactedwithanoverstoichiometricamountofLi,weareabletoobtainaLixSi–Li2OmatrixwithembeddedLidomains,wherethematrixcanconductLiionandprotectthemetallicLi.
Withthestructure,anegligiblevolumechange,reducedinitialsidereactions,andstablehigh-currentoperationupto10mA/cm2canbesimultaneouslyobtainedwheretheperformanceparametersareunprecedented.
Inaddition,ahighlyimprovedratecapabilityinLi-Sprototypecellswasfurtherdemonstrated.
Webelievethedesignprinciplein3DLiarchitecturewillofferexcitingopportunitiesfordevelopingstableLimetalanodeandpavethewayforthenext-generationhigh-energyLibatteries.
ResultsandDiscussionStableElectrodeDimensionandDendriticSuppression.
Fig.
1Asche-maticallyshowstheopen-frameworkarchitectureof3DLiwithstablehostthatwasusedpreviouslybymostoftheworks(35–37),whereLiwascoatedontoascaffoldthatservesasthestablehost.
Underthiscircumstance,Liwouldbeexposeddirectlytotheliquidelectrolytesinacell,whichwouldfurtherbringaboutseveresidereactionsattheinitialstage.
DespitethefactthatSEIcanbesta-bilizedinthelatercyclesbyminimizeddimensionalchangeandmoreuniformdeposition,excessinitialSEIformationisstillundesired.
Abetteralternativetothisdesignisanelectrolyte-proofembeddedLiasshowninFig.
1B.
Inthisdesign,Li-ionconductivematerialsareexploitedtoconstructthehost,whereasmostoftheLidomainsareembeddedinthematrixwithoutdirectcontactwithliquidelectrolytes.
Nevertheless,becauseoftheLi-ionconductioncapabilityofthematrix,theembeddedLidomainsarestillelectrochemicallyactive.
Asaconsequence,themeritsof3DLiwouldstillholdwhereasinitialsidereactionswouldbehighlyreduced.
OursyntheticstrategyontheabovementionedstructureistouseoverstoichiometricreactionofLiwithSiOor"overlithiation"ofSiO,extendedfromstoichiometricprelithiationwithmoltenLi(43).
Inconventionalprelithiationstudies,stoichiometricamountofmoltenLimetalwasusedtochemicallyconvertSi,SiOx,metaloxides,andfluoridesintoLixSi,Li2O,LiF(43–46),andthereisnoextraLimetalinthefinalproducts.
Here,anextraamountofLiwasintentionallyaddedintoSiO,whereporousLixSi+Li2OwasfirstobtainedasamatrixandfollowedbytheintakeofmoltenLitothepores.
Byscreeningmultiplecompounds,itwasfoundthattheover-lithiationbehaviorofmetastableSiOphaseisratherunique.
ViolentreactionwasobservedwhentheoverstoichiometricamountofmoltenLiandsubmicrometer-sizedSiOpowder(SIAppendix,Fig.
S1)weremixedatelevatedtemperatureof300°C.
ItwasnotedthatnanoporousLixSi–Li2Ocompositewasobtainedintheformofamatrixundervigorousstirring,whereLixSiandLi2OcanserveastheLi-ionconductivecomponents(47),withtheextraamountofLiembeddedwithinthenanopores.
Asthereactioniscomplete,drysilverypowders(SIAppendix,Fig.
S2)wereobtainedevenabovethemeltingtemperatureofLi,indicatingfullinfusionofmetallicLiintothenanoporesofthematrix.
Thesizeofthepowderparticlesisontheorderoftensorhundredsofmicrometers,muchlargerthanthedimension(100nm)previouslydemonstratedintheopen-frameworkarchitectures(36,37).
ThissignificantlyreducestheexposureofLiandtheinitialformationofSEIontheFig.
1.
Syntheticproceduresandstripping/platingbehaviorofas-obtainedelectrode.
(A)Schematicshowingtheopen-frameworkconfigurationofLimetalanodewithstablehost,wherethescaffoldiscoatedbymetallicLi.
Inthiscase,Limetalfacesdi-rectlywiththeliquidelectrolyte,whichbringsaboutexcessSEIformationattheearlystage.
(B)Schematicillustrationoftheelectrolyte-proofconfigurationwherethemajorityofLiisembeddedinaLi-ionconductivescaffold.
Afterimmersingintoelectro-lytes,SEIonlyformsontheoutersurfacewhiletheembeddedLidomainsremainintact.
(C–H)Digitalphotoimages(C–E)andcorrespondingSEMimages(F–H)showingthepristine(CandF)LCNEafterstripping8mAh/cm2(DandG)andLCNEafterplating8mAh/cm2ofLiback(EandH).
(I–K)Cross-sectionSEMimagesofpristineLCNE(I),LCNEafterstripping8mAh/cm2ofLi(J),andelectrodeafterstrippingandplatingback8mAh/cm2ofLi(K).
Thecurrentdensitywassetat2mA/cm2foralloftheabovecharacterizations.
4614|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020particlesurfaces.
Subsequently,theLCNEswereobtainedbyap-plyingamechanicalpressonthepowderstoaffordthepelletelectrodes(SIAppendix,Fig.
S3).
ThethicknessoftheLCNEscanbevariedbycontrollingtheamountofpowderforacertainpressingarea.
(SIAppendix,Fig.
S4).
Fig.
1Cshowsthedigitalphotoofanas-obtainedLCNE,whichexhibitssilverycolorwithaflatsurface.
Thecorrespondingmagnifiedscanningelectronmicroscopy(SEM)imageindicatesuniformsurfacecoveragebynanosizeddomainsofmetallicLi(Fig.
1F),whichaccountsfortheobservedcolor.
Tostudytheinternalporousstructureofthematrix,8mAh/cm2ofLi(50%ofLimetalcapacity)wasstrippedfromtheelectrodethroughgalvanostaticcharging.
AfterListripping,thetopsurfaceoftheelectrodeexhibitedobvioussilvery-to-blackcolorchange(Fig.
1D),wheretheblackcolormaybeoriginatedfromLixSiaswellastheopticalantireflectioneffectofthenanoporousstructure.
FromtheSEMimage(Fig.
1G)ofthedelithiatedelectrode,submicrometer-andnanosizedpores,whichwereinitiallyoccu-piedbyLi,wereclearlyvisualized.
Thedomainsizeofthematrixishundredsofnanometers.
Afterward,whenthe8mAh/cm2ofLiwasplatedback,thesilverycolorrecoveredasobservedfromFig.
1E.
ThecorrespondingsurfacecharacterizationfurthershowedthatalmostalloftheporeswererefilledbyLiwithoutobviousLidendrites(Fig.
1H).
ElectrodethicknessvariationwasalsocharacterizedbyexsituSEMstudytoexaminetheelectrode-leveldimensionstabilityafterListripping/plating.
ThethicknessofLCNEsunderthreeconditions,includingpristine(Fig.
1I),half-stripped(8mAh/cm2,Fig.
1J),andafteronestripping/platingcycle(Fig.
1K),wasmeasuredandcompared.
Toguaranteethateachconditionhadthesameinitialthickness,theelectrodesofthethreeconditionscamefromthesameoriginalpiece,whereanLCNEwasequallycutintoindividualsegmentsforeachcondition.
AsshowninFig.
1I–K,thethreeconditionsexhibitedalmostidenticalthicknessof138μm,whichdemonstratesanear-constantelectrodedimen-sionduringstripping/platingprocesseswiththeLi-ionconductivematrixasthehost.
TocomparethesurfacemorphologyofLifoilandLCNEsaftercycling,SEMcharacterizationwasconductedafter20gal-vanostaticcyclesatacurrentdensityof1mA/cm2andacapacityof1mAh/cm2.
Thelow-magnificationandmagnifiedSEMim-agesoftheLifoilsurfaceaftercyclingareshowninFig.
2AandB,respectively.
Fromthelow-magnificationimage,highrough-nesscanbeobserved,withexcessivegrowthofdendriticLionthewholesurface.
ThiswasfurtherconfirmedbythemagnifiedSEMimage,wherelooselystackedLidendritesandnanofila-mentswereobserved.
Incontrast,thesurfaceofLCNEafter20cycles(Fig.
2C)remainedconsistentlyflatoveralargearea.
Thehigh-resolutionimagefurtherindicatesmoreuniformLideposition(Fig.
2D).
SuchstarkcontraststronglysupportsthattheLCNEiscapableofmaintainingstablestructureandsup-pressingdendriteevolutionthroughcontinuouscycling.
TheaboveexperimentsallsupporttheuniformLidepositionwithinthematrix,whichcanbeattributedtotheLi-ioncon-ductivenatureandthushighlyincreasedactivesurfaceofthematrix.
ForLifoil,onlytheverytopsurfacecanhaveaccesstoLiion.
Asaresult,thelocalionfluxwouldbetooconcentratedtoenableuniformLideposition.
Incontrast,withLi-ionconductivematrix,Liioncanalsobetransportedintothebulkwithoutex-cessiveexposuretoliquid,whichactivatesmanymoreLinucle-ationsitesandreducessidereactions.
Inaddition,withahighlyincreasednumberofLinucleiforfurtherdeposition,unevendepositionandthusdendriticgrowthcanalsobesuppressed.
Itisnotedthatthecompositesstillretainhighspecificcapacity.
AsshowninSIAppendix,Fig.
S5A,whentheelectrodewasdeli-thiatedto1Vvs.
Li+/Li,2,250mAh/gcouldbeextracted(basedonthetotalelectrodeweight),whereLimetalcontributed1,300mAh/g.
ThephasesatdifferentdelithiationstageswereconfirmedbyX-raydiffractioninSIAppendix,Fig.
S5B,wherenoLisignalcouldbeobservedatstageB(0.
15-Vcutoff).
Aftertheelectrodewasfullydelithiatedto1V,thestructureofthewholeelectrodecouldstillbemaintained(SIAppendix,Fig.
S6),whichdemonstratedthedimensionalstabilityandinterconnectednatureofthematrix.
FewerInitialSideReactionsandStableLiStripping/Plating.
TostudytheelectrochemicalbehavioroftheLCNEs,symmetriccellswithLCNEsasbothelectrodesweretestedincarbonateelectrolyte.
Electrochemicalimpedancespectroscopy(EIS)wasfirstexploitedpriortobatterycyclingtoevaluatetheinitialSEIformation.
ItisnotedthatthesemicircleobtainedathighfrequencyinthiscellconfigurationcanbeattributedtotheSEIandcharge-transferresistance.
OnceSEIkeepsaccumulatingovertime,augmentationintheresistancecanbeexpectedaccordingly.
Fig.
3AshowstheNyquistplotsofanLCNEsymmetriccellinthefirst12h.
Itwasobservedthattheresistancewasalmostconstantthroughthefirst12h,indicatingthepromptpassivationofsurfacebySEIattheinitialstagewithoutexcesssidereactions.
Incontrast,theresistanceofasymmetriccellbasedonthepreviouslyreportedlayeredLi-reducedgrapheneoxide(rGO)increasedgraduallyinthesametimeframe(Fig.
3B),whichcanbeattributedtothehighexposureofLiandthusmoreinitialsidereactions.
Inaddition,itwasfoundthattheresistanceoftheLCNEsymmetriccellissignificantlylowerthanthatofthelayeredLi-rGOcounterpart,whichindicatesthatLCNEaffordsanevenlargeractiveLisurface.
ThesefactsstronglysupportthattheLi-ionconductivematrixcanefficientlyisolatetheembeddedLifromliquidelectrolyteandstillmaintainitselectrochemicalactivity.
ThefirstgalvanostaticcycleofbothLCNEsandLifoilelectrodeswasfurtheranalyzed.
AsshowninFig.
3C,largeoverpotentialwasobservedforLifoilcellsduringtheinitialcharging(I,400mV),followedbyagradualdropinoverpotential(gradualdeclineto100mV)inthelaterstage.
Duringdischarging,overpotential"bump"wasinitiallyshown(II),followedbyaplateauwitharelativelylowoverpotentialandlaterdischargeoverpotentialaugmentationFig.
2.
CharacterizationonmorphologyofLidepositedondifferentLimetalelectrodesafter20cycles.
(AandB)Low-magnification(A)andmagnified(B)SEMimagesshowingtheLidepositionbehavioronLifoil(B)after20cycles.
(CandD)Low-magnification(C)andmagnified(D)SEMimagesshowingtheLidepositionbehavioronLCNEafter20cycles.
Thecurrentdensitywasfixedat1mA/cm2forbothListrippingandplatingprocesses.
Thestripping/platingcapacitywas1mAh/cm2.
Linetal.
PNAS|May2,2017|vol.
114|no.
18|4615CHEMISTRYDownloadedbyguestonDecember22,2020(III).
Fig.
3DschematicallyshowsthemechanismsoftheListripping/platingbehaviorscorrespondingtotheregionsI,II,andIIIinFig.
3C.
Theoriginsoftheenergybarriersateachstageweremarkedasyellow(strippingbarrier)andred(nucleationbarrier)dotsontheelectrodesurface.
Fortheas-assembledcells,therelativelythicknativeoxidelayerandspontaneouslyformedSEIcanblockiontransport,whichwouldnotonlyhindertheListrippingfromthebottomelectrode,butalsohampertheLide-positiononthetop,bothcontributingtothemuchhigherover-potential(regionI).
Atthedischargestage,thefreshlydepositedLionthetopelectrodeisrathereasytostripduetothethinas-formedSEIlayer.
However,theconcavesurfaceofthebottomelectrodecoveredbythickSEIrequiresahigh-energybarrierforLinucleation,contributingtothehigheroverpotentialinregionII.
Afterward,newlygrownLiwouldfacilitatefurtherdeposition,whichreducestheoverpotentialasshownbetweenregionsIIandIII.
OncetheLifreshlydepositedfromthelastchargingontothetopelectrodeisexhausted,strippingfromoriginaltopLifoilbe-neaththethickSEIisrequired.
Thisimposeshigherenergybarrierandleadstothefinaloverpotentialaugmentation(III).
Incontrast,theLCNEsymmetriccellsshowedflatplateausaswellasmuchloweroverpotentialatthecharging/dischargingstages,withoutanobvious"bump"duringcycling.
Thisindicatesalower-energybarrierforbothitsnucleationandstrippingprocesses.
WithcontinuousLi-ionconductivematrix,high-surface-activeLicanserveasanucleus,enabling3DLiplating/stripping,whichcontrib-utestotheobservedlow-energybarrier.
EISmeasurementsfurthersupporttheabovestatements,whereLCNEcellsexhibitedconstantlowresistanceinitiallyandafter10cycles(SIAppendix,Fig.
S11).
Incontrast,theLifoilcellsshowedlargeresistance(650Ω/cm2)beforecycling,followedbyhighlyreducedresistance(85Ω/cm2)after10cyclesasaresultofnativeoxidecollapseanddendriticLiformation.
Inaddition,consistentlylowoverpotentialandflatvoltageplateauscanbemaintainedfortheLCNEcellsforprolongedcycles(Fig.
3E).
Incontrast,theLifoilcellsexhibitedgradualincreaseinoverpotentialthroughthecycling,whichcanbeattributedtotheaccumulationofthickSEIandthecontin-uousconsumptionofelectrolytebythesidereactions.
TheLCNEcellalsoexhibitsoutstandingratecapabilityandcyclingstabilityatdifferentcurrentdensity.
AsshowninFig.
4A,wherethecurrentdensityisvariedfrom0.
5to5mA/cm2,lowoverpotentialfrom18mV(at0.
5mA/cm2)to100mV(at5mA/cm2)canbeobtainedforLCNEs,muchlowerthanthoseoftheLifoilcounterparts.
Thecyclingstabilityatcurrentdensityof1–10mA/cm2wasfurtherdemonstrated(Fig.
4B).
At1mA/cm2(Fig.
4B,Top),stablecyclingwithlowoverpotentialof32mVandflatvoltageplateauswasobservedthrough100cycles,whereastheLifoilcellsshowedcontinuousincreaseinoverpotentialfrom65to140mV.
Inprolongedcycles,theLCNEsstillexhibitedstablecyclingwithminimaloverpotentialincrease,whereastheLifoilssufferedinternalshortcircuitafter160cycles(SIAppendix,Fig.
S12).
ThecyclingstabilityoftheLCNEsandtheLifoilelectrodesdivergedevenmoreatfurtherincreasedcurrentden-sitiesof3mA/cm2(SIAppendix,Fig.
S13),5mA/cm2(Fig.
4B,Middle),and10mA/cm2(Fig.
4B,Bottom),whereLCNEcontin-uedtoaffordstablecyclingwithlowoverpotentialbuttheLifoilcounterpartsshowedadramaticoverpotentialincreaseovercycles.
ItisnotedthattheoverpotentialofLifoilelectrodesstartedtodecreaseandfluctuateaftertensofcycles,whichcanbeattributedtotheinternalsoftshortcircuitbyLidendritepropagation.
Whenthecellswerecycledatahigherarealcapacityof3mAh/cm2(SIAppendix,Fig.
S14),theLCNEscanstillmaintaingoodstabilitywithlowoverpotential,whereastheLifoilcounterpartsexhibitedadramaticoverpotentialincreasefollowedbyinternalshortcircuitaftertensofcycles.
ThecyclelifeofLimetalanodespairedwithhigh-areal-capacityLi4Ti5O12(LTO)canbeagoodindicatoroftheCoulombiceffi-ciencyofLianodeswithextraprestoredLi(35,36).
Here,theLCNEswitharealcapacityof12mAh/cm2werepairedwithLTO(3mAh/cm2),whichshowsstablecyclingwithoutsignifi-cantdecayforatleast90cycles,outperformingboththeLifoilandelectrodepositedLionCu(SIAppendix,Fig.
S16).
Thecyclingtestatvariouscurrentdensitieswasfurthercarriedoutinether-basedelectrolyte.
Ether-basedelectrolyteisgenerallyknowntobeamuchbetterelectrolyteforLimetalanodesbecausemoreflexibleSEIlayercanformtoaccommodatethesurfaceFig.
3.
ElectrochemicalcharacteristicsofListrip-ping/platingandtheirmechanisms.
(AandB)Time-dependentNyquistplotsshowingtheimpedanceevolutionofsymmetriccellswithLCNE(A)andLifoil(B)electrodes.
(C)Typicalstripping/platingvoltageprofileoftheLifoil(black)andLCNE(red)ofthefirstgalvanostaticcycle.
(D)Schematicshowstheinitialstripping/platingbarrier(correspondingtobarrierIinC),platingbarrier(correspondingtobarrierIIinC),andfurtherstrippingbarrier(correspondingtobarrierIIIinC).
(E)VoltageprofileofLifoilsym-metriccell(black)andLCNEsymmetriccell(red)atthe1st,2nd,10th,and100thcycle.
Thecurrentden-sityandthearealcapacitywerefixedat1mA/cm2and1mAh/cm2,respectively.
4616|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020fluctuationduringstripping/plating(48).
Asaresult,muchmoreuniformLidepositioncanalreadybeachievedwithLifoil.
Nev-ertheless,theLCNEscanstilloutperformtheLifoilsinanether-basedsystem.
AsshowninSIAppendix,Fig.
S17A,atacurrentdensityof1mA/cm2theLifoilelectrodeshowedhighoverpotentialintheearlycyclesfollowedbyagradualdecrease.
ThisdecreasingphenomenoncanbeattributedtothedendriticLigrowth,whichsignificantlyincreasesthesurfacearea(20).
ForLCNEcells,aconstantlyloweroverpotentialof15mVcanbeobtainedthrough-outthewhole100cycles.
Atahighcurrentdensityof5mA/cm2(SIAppendix,Fig.
S17B),LCNEsalsoexhibitedstablecyclingwhiletheLifoilcounterpartshowedobviousvoltagefluctuation.
ImprovedRateCapabilityofLi–SBatteries.
MetallicLiisprominentforLi–Sbatterysystems(16,49,50).
Withthelow-energybarrierandstableLCNEasanodes,wefurtherdemonstratedtheirelectro-chemicalperformanceinLi–Sbatteries.
Here,12.
5μL/cm25MLi2S8in1:1vol/vol1,3-dioxolane(DOL)/1,2-dimethoxyethane(DME)(2-mg/cm2equivalentmassloadingofS)wasloadedonKOH-treatedelectrospuncarbonnanofibersasthecathodes.
Aratecapabilitytestwithcurrentdensitiesvariedfrom0.
2to2Cwascarriedout(basedontheoreticalcapacityofS1,673mAh/g,1C=3.
34mA/cm2).
Fig.
5showsthenotableratecapabilitydifferencesbetweenLi–ScellswithLCNEsandLifoilsastheanodes.
Attheearlystagewithlowcurrentdensity,ahighcapacityof1,000mAh/gcanberetainedforbothanodes.
However,itisclearlyshowninFig.
5AandBthattheLCNE-ScellsexhibitedloweroverpotentialcomparedwiththeLifoilcounterpart.
Amorepronounceddifferenceappearedwhenthecur-rentdensitywasfurtherincreased.
At2C,whichisequivalentto6.
69mA/cm2,theLifoil-Scelllostitssecondplateau(Li2S6→Li2S)withcutoffvoltageat1.
7V,whichgaverisetoasignificantcapacitydrop.
Incontrast,theLCNE-Scellyieldedmuchbetterkinetics,withasecondplateauof1.
9Vevenatthehighrateof2C.
Fig.
5CclearlyshowsthecapacityretentionatdifferentCratesduringcycling.
FortheLCNE-Scell,1,050mAh/gofspecificcapacitycanbeobtainedat0.
2C,whereasat10-foldhighercurrentdensity(2C),thecapacitystillremainedover600mAh/g.
Incontrast,muchlowerspecificcapacityretentionwasobservedfortheLifoil-Scellespeciallyathighrateasthecapacitydroppedsignificantlyfrom950mAh/gat0.
2Ctolessthan100mAh/gat2C.
ConclusionsThisworkdemonstratedaLi-ionconductivematrixasthe"host"andsurfaceprotectionforLimetalanodestoaddresstheactivesurfaceversussidereactiondilemma,whichwasfulfilledbytheoverlithiationofSiOtoyieldLi-embeddedLixSi–Li2Omatrixasthestableelectrode(LCNE).
Theas-obtainedLCNEproveshighlyreducedinitialsidereactionscomparedwiththepreviouslyreported"open-framework"architecture,whileconstantelectrode-levelFig.
5.
RatecapabilityofLi–Sbatterieswithdiffer-entLielectrodes.
(AandB)VoltageprofileofLi–SbatterieswithLCNE(A)andLifoil(B)asnegativeelectrodes.
MassloadingofSisfixedat2mg/cm2.
Crateisvariousfrom0.
2to2C(6.
69mA/cm2).
(C)CapacityretentionofLi–SbatteriesatdifferentCratewithLCNE(red)andLifoil(black)asnegativeelectrodes.
Fig.
4.
ElectrochemicalperformanceofLCNEsymmetriccell.
(A)VoltageprofileofLifoilsymmetriccell(red)andthatofLCNEsymmetriccell(blue)atdifferentratevariousfrom0.
5to5mA/cm2.
(B)ThevoltageprofilesofLifoilsymmetriccells(red)andLCNEsymmetriccells(blue)atvariouscurrentdensitiesof1mA/cm2(Top),5mA/cm2(Middle),and10mA/cm2(Bottom).
Stripping/platingcapacityisfixedat1mAh/cm2.
Linetal.
PNAS|May2,2017|vol.
114|no.
18|4617CHEMISTRYDownloadedbyguestonDecember22,2020dimensionandfurtherhomogenizedLidepositionwereobtained.
AlthoughLiwaswellprotected,alargeractivesurfacewithevenlowerimpedancewasfulfilledbytheLi-ionconductivenetwork,renderinglowpolarizationandstablecyclingwithflatplateaus.
Thisfurthercontributestotheexcellentcyclingstabilitywithmuchlowerpolarizationatvariouscurrentdensitiesupto10mA/cm2foratleast100cycles,whereasasimilareffectwasalsoattainedinanether-basedelectrolytesystem.
ByintegratingLCNEsintoLi–Sbatteries,highlyimprovedratecapabilityespeciallyathighcurrentdensitycanbeobserved.
ThisworkcreatesadesignprincipleaswellasmaterialsynthesisstrategyforLimetalanodeandopensupexcitingopportunitiesforthenext-generationhigh-energy,power-intensiveLimetalbatteriesundersafeandstableoperation.
MaterialsandMethodsMaterialsandmethods,includingoverlithiationofSiO,LCNEfabrication,charac-terizations,electrochemicalmeasurementsonsymmetriccells,lithiumpolysulphide(Li2S8)solutionpreparation,andLi–SbatteriestestscanbefoundinSIAppendix.
ACKNOWLEDGMENTS.
Y.
C.
acknowledgesthesupportfromtheAssistantSecretaryforEnergyEfficiencyandRenewableEnergy,OfficeofVehicleTechnologiesoftheUSDepartmentofEnergyundertheBatteryMaterialsResearchProgramandBattery500Consortium.
1.
ChanCK,etal.
(2008)High-performancelithiumbatteryanodesusingsiliconnano-wires.
NatNanotechnol3:31–35.
2.
MagasinskiA,etal.
(2010)High-performancelithium-ionanodesusingahierarchicalbottom-upapproach.
NatMater9:353–358.
3.
WuH,etal.
(2012)Stablecyclingofdouble-walledsiliconnanotubebatteryanodesthroughsolid-electrolyteinterphasecontrol.
NatNanotechnol7:310–315.
4.
LiuN,etal.
(2014)Apomegranate-inspirednanoscaledesignforlarge-volume-changelithiumbatteryanodes.
NatNanotechnol9:187–192.
5.
LinD,etal.
(2015)Ahightapdensitysecondarysiliconparticleanodefabricatedbyscalablemechanicalpressingforlithium-ionbatteries.
EnergyEnvironSci8:2371–2376.
6.
YangJ,WinterM,BesenhardJO(1996)SmallparticlesizemultiphaseLi-alloyanodesforlithium-ionbatteries.
SolidStateIon90:281–287.
7.
WhiteheadAH,ElliottJM,OwenJR(1999)NanostructuredtinforuseasanegativeelectrodematerialinLi-ionbatteries.
JPowerSources81–82:33–38.
8.
PeledE(1979)Theelectrochemicalbehaviorofalkaliandalkalineearthmetalsinnonaqueousbatterysystems—thesolidelectrolyteinterphasemodel.
JElectrochemSoc126:2047–2051.
9.
YamakiJ-i,etal.
(1998)Aconsiderationofthemorphologyofelectrochemicallyde-positedlithiuminanorganicelectrolyte.
JPowerSources74:219–227.
10.
BhattacharyyaR,etal.
(2010)InsituNMRobservationoftheformationofmetalliclithiummicrostructuresinlithiumbatteries.
NatMater9:504–510.
11.
ChandrashekarS,etal.
(2012)7LiMRIofLibatteriesrevealslocationofmicrostruc-turallithium.
NatMater11:311–315.
12.
HarryKJ,HallinanDT,ParkinsonDY,MacDowellAA,BalsaraNP(2014)Detectionofsubsurfacestructuresunderneathdendritesformedoncycledlithiummetalelec-trodes.
NatMater13:69–73.
13.
DingF,etal.
(2013)Dendrite-freelithiumdepositionviaself-healingelectrostaticshieldmechanism.
JAmChemSoc135:4450–4456.
14.
ZhengG,etal.
(2014)Interconnectedhollowcarbonnanospheresforstablelithiummetalanodes.
NatNanotechnol9:618–623.
15.
LuY,TuZ,ArcherLA(2014)Stablelithiumelectrodepositioninliquidandnano-poroussolidelectrolytes.
NatMater13:961–969.
16.
BrucePG,FreunbergerSA,HardwickLJ,TarasconJM(2011)Li-O2andLi-Sbatterieswithhighenergystorage.
NatMater11:19–29.
17.
LinD,LiuY,CuiY(2017)Revivingthelithiummetalanodeforhigh-energybatteries.
NatNanotechnol12:194–206.
18.
XuW,etal.
(2014)Lithiummetalanodesforrechargeablebatteries.
EnergyEnvironSci7:513–537.
19.
AurbachD,ZinigradE,CohenY,TellerH(2002)Ashortreviewoffailuremechanismsoflithiummetalandlithiatedgraphiteanodesinliquidelectrolytesolutions.
SolidStateIon148:405–416.
20.
BiekerG,WinterM,BiekerP(2015)ElectrochemicalinsituinvestigationsofSEIanddendriteformationonthelithiummetalanode.
PhysChemChemPhys17:8670–8679.
21.
JiX,etal.
(2012)Spatiallyheterogeneouscarbon-fiberpapersassurfacedendrite-freecurrentcollectorsforlithiumdeposition.
NanoToday7:10–20.
22.
StoneGM,etal.
(2012)Resolutionofthemodulusversusadhesiondilemmainsolidpolymerelectrolytesforrechargeablelithiummetalbatteries.
JElectrochemSoc159:A222–A227.
23.
BouchetR,etal.
(2013)Single-ionBABtriblockcopolymersashighlyefficientelec-trolytesforlithium-metalbatteries.
NatMater12:452–457.
24.
YangC-P,YinY-X,ZhangS-F,LiN-W,GuoY-G(2015)Accommodatinglithiuminto3Dcurrentcollectorswithasubmicronskeletontowardslong-lifelithiummetalanodes.
NatCommun6:8058.
25.
LinD,ZhuoD,LiuY,CuiY(2016)All-integratedbifunctionalseparatorforLidendritedetectionvianovelsolutionsynthesisofathermostablepolyimideseparator.
JAmChemSoc138:11044–11050.
26.
QianJ,etal.
(2015)Highrateandstablecyclingoflithiummetalanode.
NatCommun6:6362.
27.
LiW,etal.
(2015)Thesynergeticeffectoflithiumpolysulfideandlithiumnitratetopreventlithiumdendritegrowth.
NatCommun6:7436.
28.
BatesJB,DudneyNJ,NeudeckerB,UedaA,EvansCD(2000)Thin-filmlithiumandlithium-ionbatteries.
SolidStateIon135:33–45.
29.
MuruganR,ThangaduraiV,WeppnerW(2007)Fastlithiumionconductioningarnet-typeLi7La3Zr2O12.
AngewChemIntEdEngl46:7778–7781.
30.
KamayaN,etal.
(2011)Alithiumsuperionicconductor.
NatMater10:682–686.
31.
BuschmannH,etal.
(2011)Structureanddynamicsofthefastlithiumionconductor"Li7La3Zr2O12".
PhysChemChemPhys13:19378–19392.
32.
LinD,etal.
(2016)HighionicconductivityofcompositesolidpolymerelectrolyteviainsitusynthesisofmonodispersedSiO2nanospheresinpoly(ethyleneoxide).
NanoLett16:459–465.
33.
ChoudhuryS,MangalR,AgrawalA,ArcherLA(2015)Ahighlyreversibleroom-temperaturelithiummetalbatterybasedoncrosslinkedhairynanoparticles.
NatCommun6:10101.
34.
CrowtherO,WestAC(2008)Effectofelectrolytecompositiononlithiumdendritegrowth.
JElectrochemSoc155:A806–A811.
35.
LinD,etal.
(2016)Layeredreducedgrapheneoxidewithnanoscaleinterlayergapsasastablehostforlithiummetalanodes.
NatNanotechnol11:626–632.
36.
LiuY,etal.
(2016)Lithium-coatedpolymericmatrixasaminimumvolume-changeanddendrite-freelithiummetalanode.
NatCommun7:10992.
37.
LiangZ,etal.
(2016)Compositelithiummetalanodebymeltinfusionoflithiumintoa3Dconductingscaffoldwithlithiophiliccoating.
ProcNatlAcadSciUSA113:2862–2867.
38.
XuK(2004)Nonaqueousliquidelectrolytesforlithium-basedrechargeablebatteries.
ChemRev104:4303–4417.
39.
XuK(2014)ElectrolytesandinterphasesinLi-ionbatteriesandbeyond.
ChemRev114:11503–11618.
40.
OtaH,SakataY,WangX,SasaharaJ,YasukawaE(2004)CharacterizationofLithiumelectrodeinlithiumimides/ethylenecarbonateandcyclicetherelectrolytes:II.
Surfacechemistry.
JElectrochemSoc151:A437–A446.
41.
AurbachD(2000)Reviewofselectedelectrode–solutioninteractionswhichdeterminetheperformanceofLiandLiionbatteries.
JPowerSources89:206–218.
42.
YanK,etal.
(2016)Selectivedepositionandstableencapsulationoflithiumthroughheterogeneousseededgrowth.
NatEnergy1:16010.
43.
ZhaoJ,etal.
(2014)Dry-air-stablelithiumsilicide-lithiumoxidecore-shellnano-particlesashigh-capacityprelithiationreagents.
NatCommun5:5088.
44.
SunY,etal.
(2016)High-capacitybatterycathodeprelithiationtooffsetinitiallithiumloss.
NatEnergy1:15008.
45.
SunY,etal.
(2016)Insituchemicalsynthesisoflithiumfluoride/metalnanocompositeforhighcapacityprelithiationofcathodes.
NanoLett16:1497–1501.
46.
ZhaoJ,etal.
(2016)MetallurgicallylithiatedSiOxanodewithhighcapacityandambientaircompatibility.
ProcNatlAcadSciUSA113:7408–7413.
47.
ChouC-Y,KimH,HwangGS(2011)Acomparativefirst-principlesstudyofthestructure,energetics,andpropertiesofLi–M(M=Si,Ge,Sn)Alloys.
JPhysChemC115:20018–20026.
48.
AurbachD,YoungmanO,GoferY,MeitavA(1990)Theelectrochemicalbehaviourof1,3-dioxolane–LiClO4solutions.
I.
Uncontaminatedsolutions.
ElectrochimActa35:625–638.
49.
JiX,LeeKT,NazarLF(2009)Ahighlyorderednanostructuredcarbon-sulphurcathodeforlithium-sulphurbatteries.
NatMater8:500–506.
50.
BuscheMR,etal.
(2014)Systematicalelectrochemicalstudyontheparasiticshuttle-effectinlithium-sulfur-cellsatdifferenttemperaturesanddifferentrates.
JPowerSources259:289–299.
4618|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020
Mallouk,ThePennsylvaniaStateUniversity,UniversityPark,PA,andapprovedMarch23,2017(receivedforreviewNovember25,2016)Rechargeablebatteriesbasedonlithium(Li)metalchemistryareattractivefornext-generationelectrochemicalenergystorage.
Nev-ertheless,excessivedendritegrowth,infiniterelativedimensionchange,severesidereactions,andlimitedpoweroutputseverelyimpedetheirpracticalapplications.
Althoughexcitingprogresshasbeenmadetosolvepartsoftheaboveissues,aversatilesolutionisstillabsent.
Here,aLi-ionconductiveframeworkwasdevelopedasastable"host"andefficientsurfaceprotectiontoaddressthemulti-facetedproblems,whichisasignificantstepforwardcomparedwithprevioushostconcepts.
Thiswasfulfilledbyreactingoverstoichiom-etryofLiwithSiO.
Theas-formedLixSi–Li2Omatrixwouldnotonlyenableconstantelectrode-levelvolume,butalsoprotecttheembed-dedLifromdirectexposuretoelectrolyte.
BecauseuniformLinucle-ationanddepositioncanbefulfilledowingtothehigh-densityactiveLidomains,theas-obtainednanocompositeelectrodeexhibitslowpolarization,stablecycling,andhigh-poweroutput(upto10mA/cm2)evenincarbonateelectrolytes.
TheLi–Sprototypecellsfurtherexhibitedhighlyimprovedcapacityretentionunderhigh-powerop-eration(600mAh/gat6.
69mA/cm2).
Theall-aroundimprovementonelectrochemicalperformanceshedslightontheeffectivenessofthedesignprinciplefordevelopingsafeandstableLimetalanodes.
Limetal|3Dcomposite|electrolyteproof|overlithiation|high-poweroutputExtensiveresearchhasbeenconductedondevelopingad-vancedlithium(Li)batteryanodesubstitutes,includingsili-con(1–5),tin(6,7),andLimetal(8–15),forhigh-energyapplicationsinportableelectronicsandelectricaltransportation.
Amongall,Limetalisrecognizedasthemostattractivecandidate,whereitslowestelectrochemicalpotential(Li+/Li=3.
040Vvs.
standardhydrogenelectrode)andhighesttheoreticalspecificcapacity(3,860mAh/g)haveattractedbroadscientificandtechnologicalinterest(16–18).
DespitecontinuouseffortsonLimetal,itspracticalapplicationhasremainedstagnantduetomanymaterialschallenges.
Therootcauseslieintwoaspects:thehighchemicalreactivityofLimetalandsig-nificantdimensionalchangeduringcharge/discharge,whichtogethergenerateotherobservedproblemsincludingsolidelectrolyteinter-phase(SEI)fractureanddendriticdeposition.
SincetheadventofLibatteries,greatprogresshasbeenmadeinunderstandingLimetalplating/strippingbehavior(8,9,12,19,20),preventingLidendriteformationandpenetration(13,21–25),aswellasimprovingCoulombicefficiency(CE)(14,26,27).
Al-thoughfurtherimprovementinionicconductivityandinterfacialcontactisstillrequired,solidelectrolyteswereproventobeeffi-caciousformitigatingdendritepenetration(22,23,28–33).
De-velopingelectrolyteadditivesisalsoeffectiveinstabilizingSEIandhomogenizingLideposition(13,26,27,34),whereasengineeringchemicallyandmechanicallystableinterfaciallayerisalsoaprominentalternativetostabilizeSEI(14).
AlthoughthehighreactivityofLimetalhasreceivedsufficientattention,thestudieswerebasedondenseLifoil,whichimposedgrandchallengesbyintroducinglargedimensionalchange.
Werecentlyproposedthatvolumevariationisoneofthetworootcausesattractingmuchrecentattention.
Incontrasttoacarbonaceousanodewith10%volumechangeduringcycling,"hostless"Lifoilinprincipleexhibitsinfiniterelativevolumechangeduringthestripping/platingcycle(35–37).
Ononehand,theenormousvolumechangegeneratesengineeringchallengesatthecelllevelbyintroducinginternalstress.
Ontheotherhand,SEIisvulnerabletothemovinginterfaceandpronetofracture(19,38,39).
ContinuousSEIformationduringcyclingwouldnotonlydecomposeelectrolyteandconsumeLiionbutalsoaccumulateanionicinsulatinglayerwhichfurtherblockstheiontransport(40).
Moreover,dendriticandmossyLiarisenfromunevenLi-ionfluxmightpenetratethroughtheseparatorandcauseaninternalshortcircuit,triggeringthedangerofthermalrunawayandexplosionhazards(18).
Duringthestrippingprocess,dendriticmorphologycanaggravatetheformationofdeadLiandthusresultinlowCE(9,41).
Moreover,duetothelimitedaccessibleactivesurfaceofplanarLifoil,practicalpoweroperation(5–10mA/cm2ofcurrentdensity)withLimetalanodesisnotpossibleatpresent(16).
Toaddresstheseissues,anewdirectionwasrecentlyde-velopedbasedontheconceptofconstructinga3DstablehosttostoreLimetal,whichwasrealizedwithgrapheneoxide(35),nanofibermatrix(36,37),andhollowcarbonspheres(42).
Theas-obtainedcompositeelectrodesaffordminimizedvolumechangetolessthan20%andthusmorestableSEI.
Inaddition,bycreatingalargesurfaceincontactwithliquidelectrolytes,ionfluxcanbehomogenizedsothathigh-poweranddendrite-freeoperationcanbeachieved.
Withtheremarkableimprovementinperformancedemonstratedbytheseexamples,webelievethataSignificanceLithiummetalanodeholdsgreatpromisesfornext-generationhigh-energylithiumbatterysystems.
Thisworkintroducesanelectrolyte-proofdesignofthree-dimensionallithiummetalanodewheremostofthelithiumdomainsareembeddedinalithium-ionconductivematrix.
Inthisarchitecture,thelithium-ionconductivematrixcanisolatetheembeddedlithiumdo-mainsfromliquidelectrolyteandthuspreventsevereinitialsidereactions,whilethematrixcansimultaneouslytransportlithiumionandmaintaintheelectrochemicalactivityoftheembeddedlithium.
Thedesignprincipleenableshighlystable,high-power,andsafelithiummetalanodeswithminimalsidereactionsandnegligiblevolumevariationduringcycling,whichpavesthewayforviablelithiummetalbatteriesinthefuture.
Authorcontributions:D.
L.
andY.
C.
designedresearch;D.
L.
,J.
Z.
,J.
S.
,H.
Y.
,andY.
L.
per-formedresearch;D.
L.
andJ.
Z.
contributednewreagents/analytictools;D.
L.
,K.
Y.
,andY.
C.
analyzeddata;andD.
L.
andY.
C.
wrotethepaper.
Theauthorsdeclarenoconflictofinterest.
ThisarticleisaPNASDirectSubmission.
1Towhomcorrespondenceshouldbeaddressed.
Email:yicui@stanford.
edu.
Thisarticlecontainssupportinginformationonlineatwww.
pnas.
org/lookup/suppl/doi:10.
1073/pnas.
1619489114/-/DCSupplemental.
www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114PNAS|May2,2017|vol.
114|no.
18|4613–4618CHEMISTRYDownloadedbyguestonDecember22,2020significantstepforwardistoconstructaLi-ionconductivestablehostasmatrixwithembeddedLimetaltoformthe3Danode.
Ratherthanhavinghigh-surfaceLiindirectcontactwithliquidelectrolyte,whichwouldgeneratemoreSEIattheearlystage,embeddingLidomainsinsideaLi-ionconductingmatrixwouldprotectthehighlyreactiveLifromexcesssidereactionsattheearlystage,whilesimultaneouslyguaranteeingtheelectrochemicalac-tivityoftheLi.
Wewouldexpectsuchaconcepttoholdnotonlythemeritsofprevious3Danodes,butalsotoprovideapreengineeredLisurfaceprotectiontoalleviatetheinitialsidereactions.
Herein,wedevelopedafacilechemicalsynthesismethodtore-alizesuchaLi-ionconductivenanocompositeelectrode(LCNE).
ByhavingSiOreactedwithanoverstoichiometricamountofLi,weareabletoobtainaLixSi–Li2OmatrixwithembeddedLidomains,wherethematrixcanconductLiionandprotectthemetallicLi.
Withthestructure,anegligiblevolumechange,reducedinitialsidereactions,andstablehigh-currentoperationupto10mA/cm2canbesimultaneouslyobtainedwheretheperformanceparametersareunprecedented.
Inaddition,ahighlyimprovedratecapabilityinLi-Sprototypecellswasfurtherdemonstrated.
Webelievethedesignprinciplein3DLiarchitecturewillofferexcitingopportunitiesfordevelopingstableLimetalanodeandpavethewayforthenext-generationhigh-energyLibatteries.
ResultsandDiscussionStableElectrodeDimensionandDendriticSuppression.
Fig.
1Asche-maticallyshowstheopen-frameworkarchitectureof3DLiwithstablehostthatwasusedpreviouslybymostoftheworks(35–37),whereLiwascoatedontoascaffoldthatservesasthestablehost.
Underthiscircumstance,Liwouldbeexposeddirectlytotheliquidelectrolytesinacell,whichwouldfurtherbringaboutseveresidereactionsattheinitialstage.
DespitethefactthatSEIcanbesta-bilizedinthelatercyclesbyminimizeddimensionalchangeandmoreuniformdeposition,excessinitialSEIformationisstillundesired.
Abetteralternativetothisdesignisanelectrolyte-proofembeddedLiasshowninFig.
1B.
Inthisdesign,Li-ionconductivematerialsareexploitedtoconstructthehost,whereasmostoftheLidomainsareembeddedinthematrixwithoutdirectcontactwithliquidelectrolytes.
Nevertheless,becauseoftheLi-ionconductioncapabilityofthematrix,theembeddedLidomainsarestillelectrochemicallyactive.
Asaconsequence,themeritsof3DLiwouldstillholdwhereasinitialsidereactionswouldbehighlyreduced.
OursyntheticstrategyontheabovementionedstructureistouseoverstoichiometricreactionofLiwithSiOor"overlithiation"ofSiO,extendedfromstoichiometricprelithiationwithmoltenLi(43).
Inconventionalprelithiationstudies,stoichiometricamountofmoltenLimetalwasusedtochemicallyconvertSi,SiOx,metaloxides,andfluoridesintoLixSi,Li2O,LiF(43–46),andthereisnoextraLimetalinthefinalproducts.
Here,anextraamountofLiwasintentionallyaddedintoSiO,whereporousLixSi+Li2OwasfirstobtainedasamatrixandfollowedbytheintakeofmoltenLitothepores.
Byscreeningmultiplecompounds,itwasfoundthattheover-lithiationbehaviorofmetastableSiOphaseisratherunique.
ViolentreactionwasobservedwhentheoverstoichiometricamountofmoltenLiandsubmicrometer-sizedSiOpowder(SIAppendix,Fig.
S1)weremixedatelevatedtemperatureof300°C.
ItwasnotedthatnanoporousLixSi–Li2Ocompositewasobtainedintheformofamatrixundervigorousstirring,whereLixSiandLi2OcanserveastheLi-ionconductivecomponents(47),withtheextraamountofLiembeddedwithinthenanopores.
Asthereactioniscomplete,drysilverypowders(SIAppendix,Fig.
S2)wereobtainedevenabovethemeltingtemperatureofLi,indicatingfullinfusionofmetallicLiintothenanoporesofthematrix.
Thesizeofthepowderparticlesisontheorderoftensorhundredsofmicrometers,muchlargerthanthedimension(100nm)previouslydemonstratedintheopen-frameworkarchitectures(36,37).
ThissignificantlyreducestheexposureofLiandtheinitialformationofSEIontheFig.
1.
Syntheticproceduresandstripping/platingbehaviorofas-obtainedelectrode.
(A)Schematicshowingtheopen-frameworkconfigurationofLimetalanodewithstablehost,wherethescaffoldiscoatedbymetallicLi.
Inthiscase,Limetalfacesdi-rectlywiththeliquidelectrolyte,whichbringsaboutexcessSEIformationattheearlystage.
(B)Schematicillustrationoftheelectrolyte-proofconfigurationwherethemajorityofLiisembeddedinaLi-ionconductivescaffold.
Afterimmersingintoelectro-lytes,SEIonlyformsontheoutersurfacewhiletheembeddedLidomainsremainintact.
(C–H)Digitalphotoimages(C–E)andcorrespondingSEMimages(F–H)showingthepristine(CandF)LCNEafterstripping8mAh/cm2(DandG)andLCNEafterplating8mAh/cm2ofLiback(EandH).
(I–K)Cross-sectionSEMimagesofpristineLCNE(I),LCNEafterstripping8mAh/cm2ofLi(J),andelectrodeafterstrippingandplatingback8mAh/cm2ofLi(K).
Thecurrentdensitywassetat2mA/cm2foralloftheabovecharacterizations.
4614|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020particlesurfaces.
Subsequently,theLCNEswereobtainedbyap-plyingamechanicalpressonthepowderstoaffordthepelletelectrodes(SIAppendix,Fig.
S3).
ThethicknessoftheLCNEscanbevariedbycontrollingtheamountofpowderforacertainpressingarea.
(SIAppendix,Fig.
S4).
Fig.
1Cshowsthedigitalphotoofanas-obtainedLCNE,whichexhibitssilverycolorwithaflatsurface.
Thecorrespondingmagnifiedscanningelectronmicroscopy(SEM)imageindicatesuniformsurfacecoveragebynanosizeddomainsofmetallicLi(Fig.
1F),whichaccountsfortheobservedcolor.
Tostudytheinternalporousstructureofthematrix,8mAh/cm2ofLi(50%ofLimetalcapacity)wasstrippedfromtheelectrodethroughgalvanostaticcharging.
AfterListripping,thetopsurfaceoftheelectrodeexhibitedobvioussilvery-to-blackcolorchange(Fig.
1D),wheretheblackcolormaybeoriginatedfromLixSiaswellastheopticalantireflectioneffectofthenanoporousstructure.
FromtheSEMimage(Fig.
1G)ofthedelithiatedelectrode,submicrometer-andnanosizedpores,whichwereinitiallyoccu-piedbyLi,wereclearlyvisualized.
Thedomainsizeofthematrixishundredsofnanometers.
Afterward,whenthe8mAh/cm2ofLiwasplatedback,thesilverycolorrecoveredasobservedfromFig.
1E.
ThecorrespondingsurfacecharacterizationfurthershowedthatalmostalloftheporeswererefilledbyLiwithoutobviousLidendrites(Fig.
1H).
ElectrodethicknessvariationwasalsocharacterizedbyexsituSEMstudytoexaminetheelectrode-leveldimensionstabilityafterListripping/plating.
ThethicknessofLCNEsunderthreeconditions,includingpristine(Fig.
1I),half-stripped(8mAh/cm2,Fig.
1J),andafteronestripping/platingcycle(Fig.
1K),wasmeasuredandcompared.
Toguaranteethateachconditionhadthesameinitialthickness,theelectrodesofthethreeconditionscamefromthesameoriginalpiece,whereanLCNEwasequallycutintoindividualsegmentsforeachcondition.
AsshowninFig.
1I–K,thethreeconditionsexhibitedalmostidenticalthicknessof138μm,whichdemonstratesanear-constantelectrodedimen-sionduringstripping/platingprocesseswiththeLi-ionconductivematrixasthehost.
TocomparethesurfacemorphologyofLifoilandLCNEsaftercycling,SEMcharacterizationwasconductedafter20gal-vanostaticcyclesatacurrentdensityof1mA/cm2andacapacityof1mAh/cm2.
Thelow-magnificationandmagnifiedSEMim-agesoftheLifoilsurfaceaftercyclingareshowninFig.
2AandB,respectively.
Fromthelow-magnificationimage,highrough-nesscanbeobserved,withexcessivegrowthofdendriticLionthewholesurface.
ThiswasfurtherconfirmedbythemagnifiedSEMimage,wherelooselystackedLidendritesandnanofila-mentswereobserved.
Incontrast,thesurfaceofLCNEafter20cycles(Fig.
2C)remainedconsistentlyflatoveralargearea.
Thehigh-resolutionimagefurtherindicatesmoreuniformLideposition(Fig.
2D).
SuchstarkcontraststronglysupportsthattheLCNEiscapableofmaintainingstablestructureandsup-pressingdendriteevolutionthroughcontinuouscycling.
TheaboveexperimentsallsupporttheuniformLidepositionwithinthematrix,whichcanbeattributedtotheLi-ioncon-ductivenatureandthushighlyincreasedactivesurfaceofthematrix.
ForLifoil,onlytheverytopsurfacecanhaveaccesstoLiion.
Asaresult,thelocalionfluxwouldbetooconcentratedtoenableuniformLideposition.
Incontrast,withLi-ionconductivematrix,Liioncanalsobetransportedintothebulkwithoutex-cessiveexposuretoliquid,whichactivatesmanymoreLinucle-ationsitesandreducessidereactions.
Inaddition,withahighlyincreasednumberofLinucleiforfurtherdeposition,unevendepositionandthusdendriticgrowthcanalsobesuppressed.
Itisnotedthatthecompositesstillretainhighspecificcapacity.
AsshowninSIAppendix,Fig.
S5A,whentheelectrodewasdeli-thiatedto1Vvs.
Li+/Li,2,250mAh/gcouldbeextracted(basedonthetotalelectrodeweight),whereLimetalcontributed1,300mAh/g.
ThephasesatdifferentdelithiationstageswereconfirmedbyX-raydiffractioninSIAppendix,Fig.
S5B,wherenoLisignalcouldbeobservedatstageB(0.
15-Vcutoff).
Aftertheelectrodewasfullydelithiatedto1V,thestructureofthewholeelectrodecouldstillbemaintained(SIAppendix,Fig.
S6),whichdemonstratedthedimensionalstabilityandinterconnectednatureofthematrix.
FewerInitialSideReactionsandStableLiStripping/Plating.
TostudytheelectrochemicalbehavioroftheLCNEs,symmetriccellswithLCNEsasbothelectrodesweretestedincarbonateelectrolyte.
Electrochemicalimpedancespectroscopy(EIS)wasfirstexploitedpriortobatterycyclingtoevaluatetheinitialSEIformation.
ItisnotedthatthesemicircleobtainedathighfrequencyinthiscellconfigurationcanbeattributedtotheSEIandcharge-transferresistance.
OnceSEIkeepsaccumulatingovertime,augmentationintheresistancecanbeexpectedaccordingly.
Fig.
3AshowstheNyquistplotsofanLCNEsymmetriccellinthefirst12h.
Itwasobservedthattheresistancewasalmostconstantthroughthefirst12h,indicatingthepromptpassivationofsurfacebySEIattheinitialstagewithoutexcesssidereactions.
Incontrast,theresistanceofasymmetriccellbasedonthepreviouslyreportedlayeredLi-reducedgrapheneoxide(rGO)increasedgraduallyinthesametimeframe(Fig.
3B),whichcanbeattributedtothehighexposureofLiandthusmoreinitialsidereactions.
Inaddition,itwasfoundthattheresistanceoftheLCNEsymmetriccellissignificantlylowerthanthatofthelayeredLi-rGOcounterpart,whichindicatesthatLCNEaffordsanevenlargeractiveLisurface.
ThesefactsstronglysupportthattheLi-ionconductivematrixcanefficientlyisolatetheembeddedLifromliquidelectrolyteandstillmaintainitselectrochemicalactivity.
ThefirstgalvanostaticcycleofbothLCNEsandLifoilelectrodeswasfurtheranalyzed.
AsshowninFig.
3C,largeoverpotentialwasobservedforLifoilcellsduringtheinitialcharging(I,400mV),followedbyagradualdropinoverpotential(gradualdeclineto100mV)inthelaterstage.
Duringdischarging,overpotential"bump"wasinitiallyshown(II),followedbyaplateauwitharelativelylowoverpotentialandlaterdischargeoverpotentialaugmentationFig.
2.
CharacterizationonmorphologyofLidepositedondifferentLimetalelectrodesafter20cycles.
(AandB)Low-magnification(A)andmagnified(B)SEMimagesshowingtheLidepositionbehavioronLifoil(B)after20cycles.
(CandD)Low-magnification(C)andmagnified(D)SEMimagesshowingtheLidepositionbehavioronLCNEafter20cycles.
Thecurrentdensitywasfixedat1mA/cm2forbothListrippingandplatingprocesses.
Thestripping/platingcapacitywas1mAh/cm2.
Linetal.
PNAS|May2,2017|vol.
114|no.
18|4615CHEMISTRYDownloadedbyguestonDecember22,2020(III).
Fig.
3DschematicallyshowsthemechanismsoftheListripping/platingbehaviorscorrespondingtotheregionsI,II,andIIIinFig.
3C.
Theoriginsoftheenergybarriersateachstageweremarkedasyellow(strippingbarrier)andred(nucleationbarrier)dotsontheelectrodesurface.
Fortheas-assembledcells,therelativelythicknativeoxidelayerandspontaneouslyformedSEIcanblockiontransport,whichwouldnotonlyhindertheListrippingfromthebottomelectrode,butalsohampertheLide-positiononthetop,bothcontributingtothemuchhigherover-potential(regionI).
Atthedischargestage,thefreshlydepositedLionthetopelectrodeisrathereasytostripduetothethinas-formedSEIlayer.
However,theconcavesurfaceofthebottomelectrodecoveredbythickSEIrequiresahigh-energybarrierforLinucleation,contributingtothehigheroverpotentialinregionII.
Afterward,newlygrownLiwouldfacilitatefurtherdeposition,whichreducestheoverpotentialasshownbetweenregionsIIandIII.
OncetheLifreshlydepositedfromthelastchargingontothetopelectrodeisexhausted,strippingfromoriginaltopLifoilbe-neaththethickSEIisrequired.
Thisimposeshigherenergybarrierandleadstothefinaloverpotentialaugmentation(III).
Incontrast,theLCNEsymmetriccellsshowedflatplateausaswellasmuchloweroverpotentialatthecharging/dischargingstages,withoutanobvious"bump"duringcycling.
Thisindicatesalower-energybarrierforbothitsnucleationandstrippingprocesses.
WithcontinuousLi-ionconductivematrix,high-surface-activeLicanserveasanucleus,enabling3DLiplating/stripping,whichcontrib-utestotheobservedlow-energybarrier.
EISmeasurementsfurthersupporttheabovestatements,whereLCNEcellsexhibitedconstantlowresistanceinitiallyandafter10cycles(SIAppendix,Fig.
S11).
Incontrast,theLifoilcellsshowedlargeresistance(650Ω/cm2)beforecycling,followedbyhighlyreducedresistance(85Ω/cm2)after10cyclesasaresultofnativeoxidecollapseanddendriticLiformation.
Inaddition,consistentlylowoverpotentialandflatvoltageplateauscanbemaintainedfortheLCNEcellsforprolongedcycles(Fig.
3E).
Incontrast,theLifoilcellsexhibitedgradualincreaseinoverpotentialthroughthecycling,whichcanbeattributedtotheaccumulationofthickSEIandthecontin-uousconsumptionofelectrolytebythesidereactions.
TheLCNEcellalsoexhibitsoutstandingratecapabilityandcyclingstabilityatdifferentcurrentdensity.
AsshowninFig.
4A,wherethecurrentdensityisvariedfrom0.
5to5mA/cm2,lowoverpotentialfrom18mV(at0.
5mA/cm2)to100mV(at5mA/cm2)canbeobtainedforLCNEs,muchlowerthanthoseoftheLifoilcounterparts.
Thecyclingstabilityatcurrentdensityof1–10mA/cm2wasfurtherdemonstrated(Fig.
4B).
At1mA/cm2(Fig.
4B,Top),stablecyclingwithlowoverpotentialof32mVandflatvoltageplateauswasobservedthrough100cycles,whereastheLifoilcellsshowedcontinuousincreaseinoverpotentialfrom65to140mV.
Inprolongedcycles,theLCNEsstillexhibitedstablecyclingwithminimaloverpotentialincrease,whereastheLifoilssufferedinternalshortcircuitafter160cycles(SIAppendix,Fig.
S12).
ThecyclingstabilityoftheLCNEsandtheLifoilelectrodesdivergedevenmoreatfurtherincreasedcurrentden-sitiesof3mA/cm2(SIAppendix,Fig.
S13),5mA/cm2(Fig.
4B,Middle),and10mA/cm2(Fig.
4B,Bottom),whereLCNEcontin-uedtoaffordstablecyclingwithlowoverpotentialbuttheLifoilcounterpartsshowedadramaticoverpotentialincreaseovercycles.
ItisnotedthattheoverpotentialofLifoilelectrodesstartedtodecreaseandfluctuateaftertensofcycles,whichcanbeattributedtotheinternalsoftshortcircuitbyLidendritepropagation.
Whenthecellswerecycledatahigherarealcapacityof3mAh/cm2(SIAppendix,Fig.
S14),theLCNEscanstillmaintaingoodstabilitywithlowoverpotential,whereastheLifoilcounterpartsexhibitedadramaticoverpotentialincreasefollowedbyinternalshortcircuitaftertensofcycles.
ThecyclelifeofLimetalanodespairedwithhigh-areal-capacityLi4Ti5O12(LTO)canbeagoodindicatoroftheCoulombiceffi-ciencyofLianodeswithextraprestoredLi(35,36).
Here,theLCNEswitharealcapacityof12mAh/cm2werepairedwithLTO(3mAh/cm2),whichshowsstablecyclingwithoutsignifi-cantdecayforatleast90cycles,outperformingboththeLifoilandelectrodepositedLionCu(SIAppendix,Fig.
S16).
Thecyclingtestatvariouscurrentdensitieswasfurthercarriedoutinether-basedelectrolyte.
Ether-basedelectrolyteisgenerallyknowntobeamuchbetterelectrolyteforLimetalanodesbecausemoreflexibleSEIlayercanformtoaccommodatethesurfaceFig.
3.
ElectrochemicalcharacteristicsofListrip-ping/platingandtheirmechanisms.
(AandB)Time-dependentNyquistplotsshowingtheimpedanceevolutionofsymmetriccellswithLCNE(A)andLifoil(B)electrodes.
(C)Typicalstripping/platingvoltageprofileoftheLifoil(black)andLCNE(red)ofthefirstgalvanostaticcycle.
(D)Schematicshowstheinitialstripping/platingbarrier(correspondingtobarrierIinC),platingbarrier(correspondingtobarrierIIinC),andfurtherstrippingbarrier(correspondingtobarrierIIIinC).
(E)VoltageprofileofLifoilsym-metriccell(black)andLCNEsymmetriccell(red)atthe1st,2nd,10th,and100thcycle.
Thecurrentden-sityandthearealcapacitywerefixedat1mA/cm2and1mAh/cm2,respectively.
4616|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020fluctuationduringstripping/plating(48).
Asaresult,muchmoreuniformLidepositioncanalreadybeachievedwithLifoil.
Nev-ertheless,theLCNEscanstilloutperformtheLifoilsinanether-basedsystem.
AsshowninSIAppendix,Fig.
S17A,atacurrentdensityof1mA/cm2theLifoilelectrodeshowedhighoverpotentialintheearlycyclesfollowedbyagradualdecrease.
ThisdecreasingphenomenoncanbeattributedtothedendriticLigrowth,whichsignificantlyincreasesthesurfacearea(20).
ForLCNEcells,aconstantlyloweroverpotentialof15mVcanbeobtainedthrough-outthewhole100cycles.
Atahighcurrentdensityof5mA/cm2(SIAppendix,Fig.
S17B),LCNEsalsoexhibitedstablecyclingwhiletheLifoilcounterpartshowedobviousvoltagefluctuation.
ImprovedRateCapabilityofLi–SBatteries.
MetallicLiisprominentforLi–Sbatterysystems(16,49,50).
Withthelow-energybarrierandstableLCNEasanodes,wefurtherdemonstratedtheirelectro-chemicalperformanceinLi–Sbatteries.
Here,12.
5μL/cm25MLi2S8in1:1vol/vol1,3-dioxolane(DOL)/1,2-dimethoxyethane(DME)(2-mg/cm2equivalentmassloadingofS)wasloadedonKOH-treatedelectrospuncarbonnanofibersasthecathodes.
Aratecapabilitytestwithcurrentdensitiesvariedfrom0.
2to2Cwascarriedout(basedontheoreticalcapacityofS1,673mAh/g,1C=3.
34mA/cm2).
Fig.
5showsthenotableratecapabilitydifferencesbetweenLi–ScellswithLCNEsandLifoilsastheanodes.
Attheearlystagewithlowcurrentdensity,ahighcapacityof1,000mAh/gcanberetainedforbothanodes.
However,itisclearlyshowninFig.
5AandBthattheLCNE-ScellsexhibitedloweroverpotentialcomparedwiththeLifoilcounterpart.
Amorepronounceddifferenceappearedwhenthecur-rentdensitywasfurtherincreased.
At2C,whichisequivalentto6.
69mA/cm2,theLifoil-Scelllostitssecondplateau(Li2S6→Li2S)withcutoffvoltageat1.
7V,whichgaverisetoasignificantcapacitydrop.
Incontrast,theLCNE-Scellyieldedmuchbetterkinetics,withasecondplateauof1.
9Vevenatthehighrateof2C.
Fig.
5CclearlyshowsthecapacityretentionatdifferentCratesduringcycling.
FortheLCNE-Scell,1,050mAh/gofspecificcapacitycanbeobtainedat0.
2C,whereasat10-foldhighercurrentdensity(2C),thecapacitystillremainedover600mAh/g.
Incontrast,muchlowerspecificcapacityretentionwasobservedfortheLifoil-Scellespeciallyathighrateasthecapacitydroppedsignificantlyfrom950mAh/gat0.
2Ctolessthan100mAh/gat2C.
ConclusionsThisworkdemonstratedaLi-ionconductivematrixasthe"host"andsurfaceprotectionforLimetalanodestoaddresstheactivesurfaceversussidereactiondilemma,whichwasfulfilledbytheoverlithiationofSiOtoyieldLi-embeddedLixSi–Li2Omatrixasthestableelectrode(LCNE).
Theas-obtainedLCNEproveshighlyreducedinitialsidereactionscomparedwiththepreviouslyreported"open-framework"architecture,whileconstantelectrode-levelFig.
5.
RatecapabilityofLi–Sbatterieswithdiffer-entLielectrodes.
(AandB)VoltageprofileofLi–SbatterieswithLCNE(A)andLifoil(B)asnegativeelectrodes.
MassloadingofSisfixedat2mg/cm2.
Crateisvariousfrom0.
2to2C(6.
69mA/cm2).
(C)CapacityretentionofLi–SbatteriesatdifferentCratewithLCNE(red)andLifoil(black)asnegativeelectrodes.
Fig.
4.
ElectrochemicalperformanceofLCNEsymmetriccell.
(A)VoltageprofileofLifoilsymmetriccell(red)andthatofLCNEsymmetriccell(blue)atdifferentratevariousfrom0.
5to5mA/cm2.
(B)ThevoltageprofilesofLifoilsymmetriccells(red)andLCNEsymmetriccells(blue)atvariouscurrentdensitiesof1mA/cm2(Top),5mA/cm2(Middle),and10mA/cm2(Bottom).
Stripping/platingcapacityisfixedat1mAh/cm2.
Linetal.
PNAS|May2,2017|vol.
114|no.
18|4617CHEMISTRYDownloadedbyguestonDecember22,2020dimensionandfurtherhomogenizedLidepositionwereobtained.
AlthoughLiwaswellprotected,alargeractivesurfacewithevenlowerimpedancewasfulfilledbytheLi-ionconductivenetwork,renderinglowpolarizationandstablecyclingwithflatplateaus.
Thisfurthercontributestotheexcellentcyclingstabilitywithmuchlowerpolarizationatvariouscurrentdensitiesupto10mA/cm2foratleast100cycles,whereasasimilareffectwasalsoattainedinanether-basedelectrolytesystem.
ByintegratingLCNEsintoLi–Sbatteries,highlyimprovedratecapabilityespeciallyathighcurrentdensitycanbeobserved.
ThisworkcreatesadesignprincipleaswellasmaterialsynthesisstrategyforLimetalanodeandopensupexcitingopportunitiesforthenext-generationhigh-energy,power-intensiveLimetalbatteriesundersafeandstableoperation.
MaterialsandMethodsMaterialsandmethods,includingoverlithiationofSiO,LCNEfabrication,charac-terizations,electrochemicalmeasurementsonsymmetriccells,lithiumpolysulphide(Li2S8)solutionpreparation,andLi–SbatteriestestscanbefoundinSIAppendix.
ACKNOWLEDGMENTS.
Y.
C.
acknowledgesthesupportfromtheAssistantSecretaryforEnergyEfficiencyandRenewableEnergy,OfficeofVehicleTechnologiesoftheUSDepartmentofEnergyundertheBatteryMaterialsResearchProgramandBattery500Consortium.
1.
ChanCK,etal.
(2008)High-performancelithiumbatteryanodesusingsiliconnano-wires.
NatNanotechnol3:31–35.
2.
MagasinskiA,etal.
(2010)High-performancelithium-ionanodesusingahierarchicalbottom-upapproach.
NatMater9:353–358.
3.
WuH,etal.
(2012)Stablecyclingofdouble-walledsiliconnanotubebatteryanodesthroughsolid-electrolyteinterphasecontrol.
NatNanotechnol7:310–315.
4.
LiuN,etal.
(2014)Apomegranate-inspirednanoscaledesignforlarge-volume-changelithiumbatteryanodes.
NatNanotechnol9:187–192.
5.
LinD,etal.
(2015)Ahightapdensitysecondarysiliconparticleanodefabricatedbyscalablemechanicalpressingforlithium-ionbatteries.
EnergyEnvironSci8:2371–2376.
6.
YangJ,WinterM,BesenhardJO(1996)SmallparticlesizemultiphaseLi-alloyanodesforlithium-ionbatteries.
SolidStateIon90:281–287.
7.
WhiteheadAH,ElliottJM,OwenJR(1999)NanostructuredtinforuseasanegativeelectrodematerialinLi-ionbatteries.
JPowerSources81–82:33–38.
8.
PeledE(1979)Theelectrochemicalbehaviorofalkaliandalkalineearthmetalsinnonaqueousbatterysystems—thesolidelectrolyteinterphasemodel.
JElectrochemSoc126:2047–2051.
9.
YamakiJ-i,etal.
(1998)Aconsiderationofthemorphologyofelectrochemicallyde-positedlithiuminanorganicelectrolyte.
JPowerSources74:219–227.
10.
BhattacharyyaR,etal.
(2010)InsituNMRobservationoftheformationofmetalliclithiummicrostructuresinlithiumbatteries.
NatMater9:504–510.
11.
ChandrashekarS,etal.
(2012)7LiMRIofLibatteriesrevealslocationofmicrostruc-turallithium.
NatMater11:311–315.
12.
HarryKJ,HallinanDT,ParkinsonDY,MacDowellAA,BalsaraNP(2014)Detectionofsubsurfacestructuresunderneathdendritesformedoncycledlithiummetalelec-trodes.
NatMater13:69–73.
13.
DingF,etal.
(2013)Dendrite-freelithiumdepositionviaself-healingelectrostaticshieldmechanism.
JAmChemSoc135:4450–4456.
14.
ZhengG,etal.
(2014)Interconnectedhollowcarbonnanospheresforstablelithiummetalanodes.
NatNanotechnol9:618–623.
15.
LuY,TuZ,ArcherLA(2014)Stablelithiumelectrodepositioninliquidandnano-poroussolidelectrolytes.
NatMater13:961–969.
16.
BrucePG,FreunbergerSA,HardwickLJ,TarasconJM(2011)Li-O2andLi-Sbatterieswithhighenergystorage.
NatMater11:19–29.
17.
LinD,LiuY,CuiY(2017)Revivingthelithiummetalanodeforhigh-energybatteries.
NatNanotechnol12:194–206.
18.
XuW,etal.
(2014)Lithiummetalanodesforrechargeablebatteries.
EnergyEnvironSci7:513–537.
19.
AurbachD,ZinigradE,CohenY,TellerH(2002)Ashortreviewoffailuremechanismsoflithiummetalandlithiatedgraphiteanodesinliquidelectrolytesolutions.
SolidStateIon148:405–416.
20.
BiekerG,WinterM,BiekerP(2015)ElectrochemicalinsituinvestigationsofSEIanddendriteformationonthelithiummetalanode.
PhysChemChemPhys17:8670–8679.
21.
JiX,etal.
(2012)Spatiallyheterogeneouscarbon-fiberpapersassurfacedendrite-freecurrentcollectorsforlithiumdeposition.
NanoToday7:10–20.
22.
StoneGM,etal.
(2012)Resolutionofthemodulusversusadhesiondilemmainsolidpolymerelectrolytesforrechargeablelithiummetalbatteries.
JElectrochemSoc159:A222–A227.
23.
BouchetR,etal.
(2013)Single-ionBABtriblockcopolymersashighlyefficientelec-trolytesforlithium-metalbatteries.
NatMater12:452–457.
24.
YangC-P,YinY-X,ZhangS-F,LiN-W,GuoY-G(2015)Accommodatinglithiuminto3Dcurrentcollectorswithasubmicronskeletontowardslong-lifelithiummetalanodes.
NatCommun6:8058.
25.
LinD,ZhuoD,LiuY,CuiY(2016)All-integratedbifunctionalseparatorforLidendritedetectionvianovelsolutionsynthesisofathermostablepolyimideseparator.
JAmChemSoc138:11044–11050.
26.
QianJ,etal.
(2015)Highrateandstablecyclingoflithiummetalanode.
NatCommun6:6362.
27.
LiW,etal.
(2015)Thesynergeticeffectoflithiumpolysulfideandlithiumnitratetopreventlithiumdendritegrowth.
NatCommun6:7436.
28.
BatesJB,DudneyNJ,NeudeckerB,UedaA,EvansCD(2000)Thin-filmlithiumandlithium-ionbatteries.
SolidStateIon135:33–45.
29.
MuruganR,ThangaduraiV,WeppnerW(2007)Fastlithiumionconductioningarnet-typeLi7La3Zr2O12.
AngewChemIntEdEngl46:7778–7781.
30.
KamayaN,etal.
(2011)Alithiumsuperionicconductor.
NatMater10:682–686.
31.
BuschmannH,etal.
(2011)Structureanddynamicsofthefastlithiumionconductor"Li7La3Zr2O12".
PhysChemChemPhys13:19378–19392.
32.
LinD,etal.
(2016)HighionicconductivityofcompositesolidpolymerelectrolyteviainsitusynthesisofmonodispersedSiO2nanospheresinpoly(ethyleneoxide).
NanoLett16:459–465.
33.
ChoudhuryS,MangalR,AgrawalA,ArcherLA(2015)Ahighlyreversibleroom-temperaturelithiummetalbatterybasedoncrosslinkedhairynanoparticles.
NatCommun6:10101.
34.
CrowtherO,WestAC(2008)Effectofelectrolytecompositiononlithiumdendritegrowth.
JElectrochemSoc155:A806–A811.
35.
LinD,etal.
(2016)Layeredreducedgrapheneoxidewithnanoscaleinterlayergapsasastablehostforlithiummetalanodes.
NatNanotechnol11:626–632.
36.
LiuY,etal.
(2016)Lithium-coatedpolymericmatrixasaminimumvolume-changeanddendrite-freelithiummetalanode.
NatCommun7:10992.
37.
LiangZ,etal.
(2016)Compositelithiummetalanodebymeltinfusionoflithiumintoa3Dconductingscaffoldwithlithiophiliccoating.
ProcNatlAcadSciUSA113:2862–2867.
38.
XuK(2004)Nonaqueousliquidelectrolytesforlithium-basedrechargeablebatteries.
ChemRev104:4303–4417.
39.
XuK(2014)ElectrolytesandinterphasesinLi-ionbatteriesandbeyond.
ChemRev114:11503–11618.
40.
OtaH,SakataY,WangX,SasaharaJ,YasukawaE(2004)CharacterizationofLithiumelectrodeinlithiumimides/ethylenecarbonateandcyclicetherelectrolytes:II.
Surfacechemistry.
JElectrochemSoc151:A437–A446.
41.
AurbachD(2000)Reviewofselectedelectrode–solutioninteractionswhichdeterminetheperformanceofLiandLiionbatteries.
JPowerSources89:206–218.
42.
YanK,etal.
(2016)Selectivedepositionandstableencapsulationoflithiumthroughheterogeneousseededgrowth.
NatEnergy1:16010.
43.
ZhaoJ,etal.
(2014)Dry-air-stablelithiumsilicide-lithiumoxidecore-shellnano-particlesashigh-capacityprelithiationreagents.
NatCommun5:5088.
44.
SunY,etal.
(2016)High-capacitybatterycathodeprelithiationtooffsetinitiallithiumloss.
NatEnergy1:15008.
45.
SunY,etal.
(2016)Insituchemicalsynthesisoflithiumfluoride/metalnanocompositeforhighcapacityprelithiationofcathodes.
NanoLett16:1497–1501.
46.
ZhaoJ,etal.
(2016)MetallurgicallylithiatedSiOxanodewithhighcapacityandambientaircompatibility.
ProcNatlAcadSciUSA113:7408–7413.
47.
ChouC-Y,KimH,HwangGS(2011)Acomparativefirst-principlesstudyofthestructure,energetics,andpropertiesofLi–M(M=Si,Ge,Sn)Alloys.
JPhysChemC115:20018–20026.
48.
AurbachD,YoungmanO,GoferY,MeitavA(1990)Theelectrochemicalbehaviourof1,3-dioxolane–LiClO4solutions.
I.
Uncontaminatedsolutions.
ElectrochimActa35:625–638.
49.
JiX,LeeKT,NazarLF(2009)Ahighlyorderednanostructuredcarbon-sulphurcathodeforlithium-sulphurbatteries.
NatMater8:500–506.
50.
BuscheMR,etal.
(2014)Systematicalelectrochemicalstudyontheparasiticshuttle-effectinlithium-sulfur-cellsatdifferenttemperaturesanddifferentrates.
JPowerSources259:289–299.
4618|www.
pnas.
org/cgi/doi/10.
1073/pnas.
1619489114Linetal.
DownloadedbyguestonDecember22,2020
- Dendriticstablehost相关文档
- waterstablehost
- 化学stablehost
- 主机StableHost主机购买图文教程
- 主机stablehost主机50%优惠码 终身5折
- 优惠Stablehost主机注意事项
- 主机StableHost主机介绍
趣米云月付460元,香港CN2云服务器VPS月付低至18元
趣米云早期为做技术起家,为3家IDC提供技术服务2年多,目前商家在售的服务有香港vps、香港独立服务器、香港站群服务器等,线路方面都是目前最优质的CN2,直连大陆,延时非常低,适合做站,目前商家正在做七月优惠活动,VPS低至18元,价格算是比较便宜的了。趣米云vps优惠套餐:KVM虚拟架构,香港沙田机房,线路采用三网(电信,联通,移动)回程电信cn2、cn2 gia优质网络,延迟低,速度快。自行封...
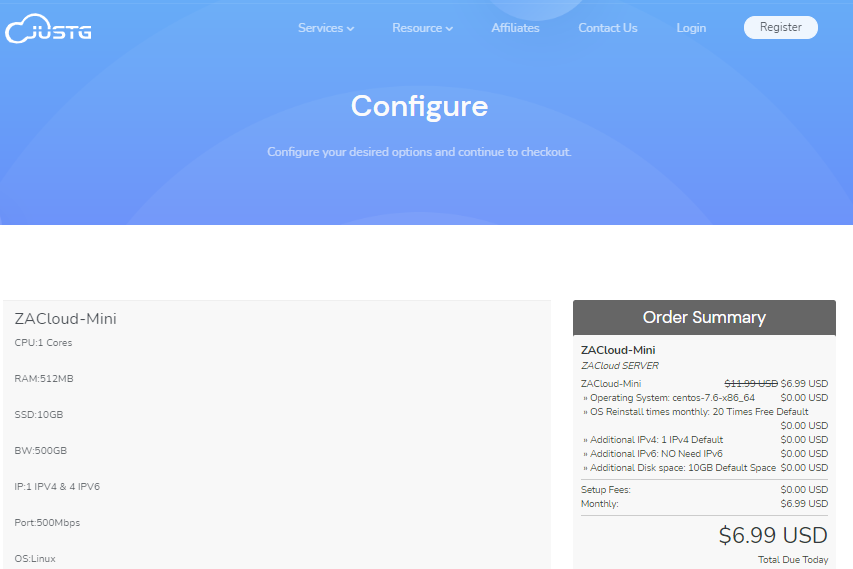
JUSTG(5.99美元/月)最新5折优惠,KVM虚拟虚拟512Mkvm路线
Justg是一家俄罗斯VPS云服务器提供商,主要提供南非地区的VPS服务器产品,CN2高质量线路网络,100Mbps带宽,自带一个IPv4和8个IPv6,线路质量还不错,主要是用户较少,带宽使用率不高,比较空闲,不拥挤,比较适合面向非洲、欧美的用户业务需求,也适合追求速度快又需要冷门的朋友。justg的俄罗斯VPS云服务器位于莫斯科机房,到美国和中国速度都非常不错,到欧洲的平均延迟时间为40毫秒,...
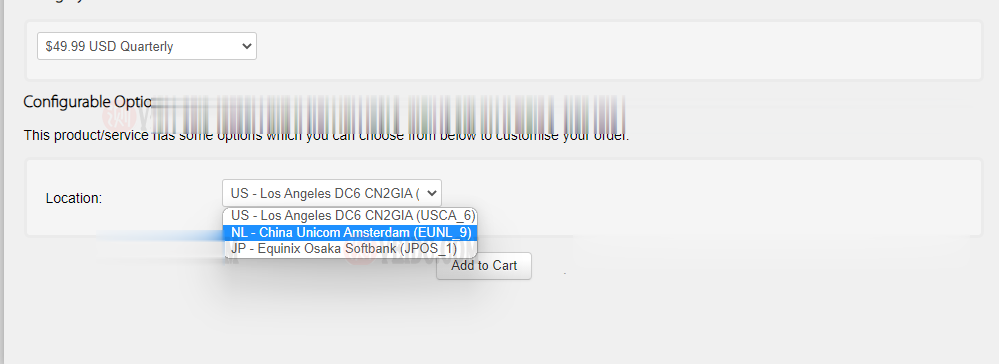
搬瓦工:新增荷兰机房 EUNL_9 测评,联通 AS10099/AS9929 高端优化路线/速度 延迟 路由 丢包测试
搬瓦工最近上线了一个新的荷兰机房,荷兰 EUNL_9 机房,这个 9 的编号感觉也挺随性的,之前的荷兰机房编号是 EUNL_3。这次荷兰新机房 EUNL_9 采用联通 AS9929 高端路线,三网都接入了 AS9929,对于联通用户来说是个好消息,又多了一个选择。对于其他用户可能还是 CN2 GIA 机房更合适一些。其实对于联通用户,这个荷兰机房也是比较远的,相比之下日本软银 JPOS_1 机房可...
stablehost为你推荐
-
美国主机租用租用美国服务器有什么优势?asp主机空间Asp空间是什么空间啊?跟有的网站提供的免费空间有什么区别吗?域名服务什么叫主域名服务器?免费网站空间哪里有永久免费的网站空间?asp网站空间ASP空间是什么?网站空间申请网站空间申请虚拟主机软件常见的虚拟机软件有哪几种?1g虚拟主机我要做一个下载资料类网站,刚买了一个虚拟主机1G的,提供商说一次,只能上传一个小于10M的文件云南虚拟主机用哪家虚拟主机?(美橙互联还是西部数码)虚拟主机测评虚拟主机哪个最好