Laboratories22pipi.com
22pipi.com 时间:2021-04-11 阅读:()
DISEASESOFAQUATICORGANISMSDisAquatOrgVol.
67:9–14,2005PublishedNovember9INTRODUCTIONThereismuchknownaboutthediseasesofwildlifewhichareeconomicallyorrecreationallyimportant.
However,thereismuchlessknownaboutthedis-easesofspeciesthatdonotfallintooneofthesetwocategories.
VirusesofthefamilyIridoviridaehavebeenincreasinglyrecognizedasacauseofamphib-ianmortalityevents(Alford&Richards1999,Greenetal.
2002).
ThefamilyIridoviridaeiscomposedoflargeDNAviruseswithicosahedralcapsids(Bollingeretal.
1999,Chinchar&Mao2000,Chinchar2002).
Withinthisvirusfamilythereare5recognizedgen-era,andRanavirusisoneofthegenerathatispatho-genictoamphibians,fishandreptiles(Daszaketal.
1999).
RanavirusinfectionshavebeenfoundwithinawiderangeofamphibianandfishpopulationsinAustralia(Speare&Smith1992),Europe(Hyattetal.
2000),Asia(Marschangetal.
1999),SouthAmerica(Speareetal.
1991,Zupanovicetal.
1998a)andNorthAmerica(Bollingeretal.
1999,Greenetal.
2002).
Themanifestationofranavirusdiseasewithinlowervertebratespeciesworldwiderangesfrominapparenttofatal,andvariesdependingonthevirusandthehostspecies(Chinchar&Mao2000).
Thisdiseaseinfrogshasbeenobservedtocausemortalityratesapproach-ing100%intadpoleandmetamorphicindividualsinseverallocations(Greenetal.
2002).
TheUnitedStatesGeologicalSurvey(USGS)hasinvestigatedmassmor-talityeventsinvolvingamphibiansandfoundthatbetween1996and2001,31of64occurrencesofamphibianillnessanddeathintheUSAcouldbeattributedtoranaviralinfection,withanadditional9%beingattributedtoranaviralinfectioncombinedwithanotheretiology(Greenetal.
2002).
ItisbecomingInter-Research2005·www.
int-res.
com*Email:amy.
greer@asu.
eduFiveamphibianmortalityeventsassociatedwithranavirusinfectioninsouthcentralOntario,CanadaAmyL.
Greer1,*,MichaelBerrill2,PaulJ.
Wilson2,31WatershedEcosystemsGraduateProgram,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B8,Canada2DepartmentofBiology,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B5,Canada3NaturalResourcesDNAProfilingandForensicCentre,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B5,CanadaABSTRACT:Usingfield,molecularandhistologicalmethods,anepizootic,systemicdiseasecausingdeathwithinwoodfrogRanasylvaticatadpolesandleopardfrogRanapipiensmetamorphsat3dif-ferentlocationswithinSouthernOntario,Canada,hasbeeninvestigated.
Ourresultsdemonstratedthattheprobablecauseofthisdiseasewasaranavirus.
Affectedamphibianswerefoundtoexhibitnecrosiswithinthehematopoieticcells.
LivertissuesampleswerefoundpositiveforthevirusbyPCRamplificationoftheranavirus(Family:Iridoviridae)majorcapsidprotein(MCP).
Positivesampleswereconfirmedbysequenceanalysis.
Clinicallynormal,laboratory-raisedwoodfrogeggbroodswerealsofoundtotestweaklypositiveforranavirus.
Thepopulationeffectsofdiseaseontheseamphibiancommunitieshavenotyetbeenconclusivelyassociatedwithpopulationdeclines,butwar-rantmorefocusedconsideration.
KEYWORDS:Frog·Ranasylvatica·Ranapipiens·Ranavirus·Amphibian·Polymerasechainreaction·CanadaResaleorrepublicationnotpermittedwithoutwrittenconsentofthepublisherDisAquatOrg67:9–14,2005apparentthattheinfectionanddeathofdiversegroupsofamphibianstestingpositiveforranavirusisaseriousandpotentiallydevastatingoccurrence.
Wereporthereontherecurringpresenceofaranavirusin3amphibianpopulationsinSouthernOntario,Canada.
Theobjectivesforthisworkweretodeterminewhetheraranaviruswasassociatedwith5amphibianmortalityeventsinvestigatedinsouthcentralOntario,Canada.
Wealsoexaminedpotentialtransmissionroutesfortheviruswithinthesepopulations.
MATERIALSANDMETHODSFieldsites.
Tadpolesandrecentmetamorphswerecollectedfrom5differentsiteswithinSouthernOntario.
Fieldsitescanbeclassifiedinto2categories,namely(1)siteswherediseasewasobservedandsub-sequentlyinvestigated,and(2)siteswherenodiseasewasobserved.
DiseasesitesincludedtheJamesOliverEcologicalResearchStation('OliverPond';44°31'N,78°32'W),TheKortrightCentreforConservation('KortrightPond';43°57'N,79°42'W)andGannon'sNarrows(44°38'N,78°41'W).
SitesthatshowednosignsofdiseaseandwereusedascontrolsitesincludedthePoplarPond(44°31'N,78°32'W)andtheTrentUniversityArcheologyPond(44°21'N,78°17'W).
Tobeconsideredadiseasedsite,≥10ind.
hadtomeetthecasedefinition.
Thismeantthatsickand/ordeadindividualshadtoexhibit4of7clinicalsigns.
Clinicalsignsincludedrednessinthevent,abdomen,orlegs,swollenvent,edemaofthebodycavity,cutaneouslesions,anorexiaandlossofescapebehaviour.
TheOliverPondlocationisuniquebecausetherehavebeen3consecutiveyearsofsimilarmassmortalityeventshere.
InMay2002,RanasylvaticaeggmasseswerecollectedfromtheOliverPondwithin12hofbeinglaidandrearedinthelaboratoryin5gallon(19l)aquariacontainingagedriverwater.
Collectionofanimals.
Liveanimalsweretrans-portedbacktothelaboratory.
Individualsfoundatthefieldsitethatwerealreadydeadweretransportedbacktothelaboratoryonice,andindividualsthatwerealiveanddemonstratingsignsofdiseaseweretrans-portedaliveandtheneuthanizedbyadministeringanoverdoseoftricainemethanesulfonate(SyndelLabora-tories).
Liversweredissectedfromallindividualsanddatawerecollectedonoverallbodyconditioninclud-ingfatbodies,organcolourationandobservablelesions.
Equipmentwasdecontaminatedusingacon-centratedbleachsolutionbeforemovingbetweenfieldsites(Green&Barry2001).
Animalhusbandry.
Laboratory-raisedtadpoleswerefedcookedspinachandgoldfishflakesdaily.
Allaquariawereconstantlyaerated.
Individualswereeuthanizedatstage42byadministeringanoverdoseoftricainemethanesulfonate(SyndelLaboratories).
Histopathology.
Livertissuesampleswerepro-cessedforhistologyusingstandardhistologicaltech-niques.
Eachsamplewasseriallysectionedat7m,andstainedwithhematoxylinandeosin.
Sectionswereexaminedunderalightmicroscopeforpatho-logicalfindings(Bollingeretal.
1999,Marschangetal.
1999).
Moleculardiagnostics.
DNAwasisolatedfromfrozenandpreservedlivertissuebiopsysamplesfrombothhealthyanddiseasedindividuals.
Tissueswerelysedin500lof1*lysisbufferand25lofProteinaseK(14.
4mgml–1,>60.
0U100l–1).
Sampleswereincu-batedinabeakerof65°Cwaterina37°Cincubator.
Af-ter1h,asecond25lofProteinaseKwasaddedandsampleswereincubatedat37°Covernight.
ExtractionsoftotalcellularDNAwerecompletedusingthestandardQIAampDNeasyProtocolforAnimalTissue(Qiagen).
Amplificationwasconductedusingprimersspecificfora500bpfragmentoftheranavirusmajorcapsidprotein(MCP)(Maoetal.
1997)in10lPCRreactions.
Thermocyclingconditionsweresimilarforallsamples(94°C5min,94°C30s,55°C30sand72°C30s,cycled35timesfollowedbyanextensionof72°C2min)withtheexceptionoftheannealingtemperaturethatwasoptimizedat60°Cforleopardfrogsamples.
Bothpositivecontrols(plasmidDNAwithRanavirusMCPinsertobtainedfromV.
G.
Chinchar,Univ.
ofMissis-sippiMedicalCenter,Jackson,MS,USA)andnegativecontrolsweretestedalongsideallunknownsamples.
Allamplifiedproductwasvisualizedbyelectrophoresison1.
5%agarosegels.
Sequencesoftheamplifiedpro-ductwerecomparedtopreviouslypublishedsequencesusingtheNCBIBlastSearchnucleotidedatabase.
ClustalmultiplealignmentsforviralsampleswereconductedusingBioEditsequencealignmenteditorsoftwareandthenrealignedwithpublishedFrogVirus3(ranavirus)sequences.
RESULTSFieldobservationsSince1999,massmortalityeventsinvolvingtadpolesandnewmetamorphshavebeeninvestigatedwithinOntario.
Intotal,5mortalityeventshavebeeninvesti-gated.
Theseeventsinvolved2differentspeciesofamphibiansin3differentlocationswithinOntario,andoneoftheselocationshasexperiencedareoccurringepidemicstate.
Ineachlocationmorethan50individ-ualsmeetingthecasedefinitionwereidentified.
AttheOliverPond,Ranasylvaticaexperiencedmassmortal-10Greeretal.
:Amphibianmortalityandranavirusityeventsinvolvingmorethan100individualsinJuneof1999,2000and2001.
Morethan50RanasylvaticawerefounddeadattheKortrightPondinJune2001,andover100RanapipiensmetamorphswerefounddeadatGannon'sNarrowsinAugust2002.
Necropsyobservationsincludededemaofthebodycavity,poorfatbodycomposition,paleandmottledcolourationoftheliverandsystemichemorrhage(Fig.
1).
Cutaneouslesionsandulcerswerenotobserved.
Inallmassmor-talityevents,only1speciesofamphibianwasinvolveddespitethepresenceofadiverseamphibiancommu-nity.
In4eventsonlytadpoleswereinfected,andtheseindividualsrangedfromGosnerstages40to45(Gos-ner1960).
Recentmetamorphswereafflictedbyonly1mortalityevent,andnomorbidordeadadultswereobservedatanyofthelocations.
All5ofthediseaseoutbreaksinOntariohadonsetswithinashorttime-framebetweenJuneandAugust.
NomassmortalityeventswereinvestigatedbetweenSeptemberandMay.
Anaccuratetotalnumberofindividualsthatsuccumbedtoinfectionateachlocationisunknown.
However,during2000and2001nonewwoodfrogmetamorphswereobservedattheOliverPond.
Also,nomortalityeventwasobservedattheOliverPondduring2002after3previousevents.
Histologicalexaminationandpoly-merasechainreactionanalysis(PCR)(Table1)havecontributedtotheiden-tificationofthepresenceofaranaviruswithintheseindividuals.
Sixtypercentoftheamphibianmortalityeventsinvestigatedwereassociatedwiththepresenceofaranavirus(Family:Irido-viradae)andoccurredattheOliverPondin2001,KortrightCentrein2001andGannon'sNarrowsin2002.
Theremaining40%ofcasesaresuspectedtobeassociatedwiththissamevirusbasedonsimilarclinicalsigns,locationandepidemiologicaldatadespitealackofsuitablesamplesfortissuehistologyorPCRidentification.
TheseincludemortalityeventsobservedattheOliverPondin1999and2000.
Thedurationofthesedie-offeventshasrangedfromseveraldaystoseveralweeks.
HistopathologyHistologicalexaminationsoflivertissueyieldedsev-eralfindings.
DiseasefreeleopardfrogmetamorphsfromtheTrentUniversityArcheologyPondwerefoundtoexhibitnormalcellularstructureoftheliver.
Livertissueintheseindividualsdidnotsuggestanytypeofpathologyandwassubsequentlyusedascontroltisse(Fig.
2).
Livertissuesectionsfromleopardfrogmeta-morphscollectedfromadie-offeventatGannon'sNar-rowswerefoundtoexhibitpathologicalchangesandsystemicnecrosisofthehematopoietictissues.
In-flammatorycellswereobservedintheportaltracts,aswasballooningdegenerationwithinthehepatocytes(Fig.
3).
AllleopardfrogliversexaminedwerematchedwithcontrolliversbyGosnerstagetocontrolforanyeffectsofmetamorphosis.
11Fig.
1.
Ranasylvatica.
ExampleofasystemichemorrhageLocationYearNo.
ind.
meetingAbnormalInclusionbodies%samplespositivecasedefinitionliverpathologybyPCROliverPond(field)2001>100NDND71KortrightPond(field)2001>50NDND93OliverPond(lab-raised)20020+–60OliverPond(field)20020+–70Gannon'sNarrows(field)2002>100++82ArcheologyPond(field)20020––0Table1.
DiagnosticcriteriaforamphibianmortalityeventsinsouthcentralOntario,Canada.
+:positiveresult;–:negativeresult;ND:nodataavailableDisAquatOrg67:9–14,2005MoleculardiagnosticsLivertissuescollectedfromindividualsmeetingthecasedefinitionfromtheOliverPond2001,KortrightCentre2001,andGannon'sNarrows2002eventswerefoundtobePCRpositivewhencomparedtoaknownpositivecontrol(ranavirusMCPinsertinplasmidDNAobtainedfromV.
G.
Chinchar).
TheranavirusMCP-codingregionwasalsoidentifiedinlivertissueobtainedfromindividualsattheOliverPond2002(bothpond-raisedandlaboratory-raised)thatdidnotmeetthecasedefinition.
Thesesamplesshowednoobservablediseasecharacteristicsbutthepopulationhadahistoryofpreviousmortalityeventsthatdidmeetthecasedefinitionandhadthereforeexperiencedmortalityeventsinthepast.
However,individualsfromtheArcheologyPond,wherethathavebeennoobser-vationsofunusualmorbidityormortality,werefoundtotestnegativebyPCR.
RepresentativesamplesfromOliverPond2001,OliverPond2002,KortrightPond2001,laboratory-raisedOliverPond2002andGannon'sNarrows2002wereconfirmedpositiveforranavirusMCPcodingDNAbysequenceanalysis.
SequenceanalysisrevealedthatallMCPsequences(~500bp)werea98%matchtoranavirus(FrogVirus3)MCPandalsoindicatedthattheMCPsequencesdidnotdifferamongindividualsorlocations.
DISCUSSIONBasedonfield,histologicalandmolecularinvestiga-tions,aranavirusmaybeassociatedwithmorbidityandmortalitywithinOntarioamphibianpopulations.
Theclinicalsignsofdiseaseintheindividualsexam-inedhavesimilaritiestootherdocumentedobserva-tionsfromranavirusinfectionsites(Careyetal.
1999,Chinchar&Mao2000,Chinchar2002).
Thedatageneratedduringthisstudysupportpre-viousfindingsthatlatestagetadpolesandnewmeta-morphsappearmoresusceptibletoranavirus(Greenetal.
2002).
Althoughthepathologicalfindingsappeartobesignificantlydifferentbetweennon-infectedandinfectedRanapipiensmetamorphs,itisimportanttoconsiderthatmetamorphosismaycausechangestotheliverregardlessofviralstatus.
However,thisseems12Fig.
2.
Ranapipiens.
LivercrosssectionfromanormalmetamorphcollectedfromTrentUniversityArcheologyPond.
Notearrangementsofthehepatocytesandsinusoids(S)containingbloodcells,radiatingthroughoutFig.
3.
Ranapipiens.
LivercrosssectionfromaGannon'sNarrowsmetamorph.
Ballooningdegeneration(hypertrophy)canbeobservedandischaracterizedbyswellingofcells.
Thisswellingcausedsinusoidstobecomeblocked.
NoopensinusoidscanbeobservedGreeretal.
:Amphibianmortalityandranavirusunlikelysincecontrolliverswerematchedwithexper-imentalliversofthesamestageanddifferenceswerestillobserved.
Metamorphosismayplaceamphibiansatagreaterriskofclinicaldiseasefrompossiblerana-virusinfectionbecauseofthepotentialfortransitoryimmunedeficiencytooccurduringthistimeframe.
Metamorphosisisarguablyoneofthemorestressfuleventsinthelifecycleofananuran(Barkeretal.
1997,Rollins-Smith1998,Calleryetal.
2001).
Ithasbeensuggestedthatdiminishedresilienceoftheimmunesystemduringthetransitionbetweentadpoleandadultmaybeacontributingfactortothesusceptibilityoftheseamphibianstopathogensattheclimaxofmetamorphosis(Moreyetal.
1996);duringmetamor-phosistheremaybeawindowofdecreasedimmunefunction,atwhichtimetheseamphibiansmaybemorevulnerabletoinfectionsresultingfromapathogenicvirus.
Viralpositiveswereobservedinlab-rearedclini-callynormalindividualscollectedaseggsfromloca-tionswithahistoryofpreviousmortalityevents.
Indi-vidualsinthiscategoryofinfectionshowednosignsofmorbidityormortality.
Theonlylinkingfactortoranaviruswasthehistoricalexposureoftheparentfrogs.
Thissuggeststhatinanurans,thepossibilityofverticaltransmissionoftheviruscannotberuledout,norcantheselectionforresistanceamongadultfrogspreviouslyexposed.
Therearenopublishedstudiesthatevaluatethepossibilityofverticaltransmissionofranavirus.
However,therearereportsofotherDNAviruses,suchasgillassociatedvirusandchannelcat-fishvirus,thathavedemonstratedverticaltransmis-sionofvirusfromadultstoeggs(Wiseetal.
1988,Brauer1995,Cowleyetal.
2002).
Itispossiblethatverticaltransmissionisoccurringinanuransinfectedwithranavirusbasedondatacollectedfromthisresearch.
TherewasnomorbidityormortalityobservedattheOliverPondin2002despite3annualreoccurrences.
Literaturesourcesindicatethatinsomeamphibianpopulations,ranavirusinfectionbecomesanannuallyreoccurringproblem(Careyetal.
1999).
Theobserva-tionthattheOliverPonddidnothaveanoutbreakin2002issignificant.
Firstly,itispossiblethatasmallnumberoftadpolesexposedtothepathogenduringdie-offeventsin1999,2000or2001didnotperishduetoinherentresistance.
Theseanimalswouldthenbetheanimalsobservedbreedingthefollowingyear(Whittingtonetal.
1997,Zupanovicetal.
1998a,b).
ThepresentresultsfortheOliverPondareconsistentwithwhatwouldbeexpectedafterseveralyearsofstrongselectionforresistance.
Afterseveralyearsofthisselection,theresultingprogenymaynolongerbepronetothediseasealthoughtheymaycarrythevirusatlowlevels.
Secondly,theOliverPondexperiencedavarietyofdifferentweatherconditionsduringthespringof2002.
Therefore,itispossiblethatthediseasewasnotobservedin2002becausethepopulationwasnotunderthesameamountofstressaspreviousyears.
TherewereconsiderabledifferencesinthewaterleveloftheOliverPondfrom1999to2002,withverylowwaterlevelsobservedduringthesummerof1999to2001andveryhighwaterlevelsobservedin2002.
Dur-ingtimesoflowwaterlevels,thepopulationdensityisincreasedwhichincreasestheprobabilitythatsuscep-tibleindividualswillencounterenough(1)infectiousagentinthewateror(2)infectiousindividualstosuc-cumbtoinfection.
Duringtimesofhighwaterandlowpopulationdensities,itwouldbeexpectedthattheprobabilityofsusceptibleindividualsencounteringinfectedindividualswouldbelowand,asaresult,massmortalitieswouldnotoccurwithinthepopula-tion.
Thesetheoriesregardingtheobservedlackofdis-easeattheOliverPondin2002allowustomakepre-dictionsaboutwhatmaybeobservedinyearstocome.
Ifthediseasewasnotobservedbecauseofselectionforresistance(strongherdimmunity)in2002,thediseasemaynotoccuragainattheOliverPond,ormaybereducedinitsprevalenceevenifthepondexperiencesabioticstress.
Thisselectionforimmunitysuggeststhatthediseaseitselfmaynotbeamajorconservationissue,ashasbeenpreviouslysuggested.
Alternatively,ifthepopulationisnotstronglyselectedforimmunityandtheoutbreaksaretrulyunderthecontrolofstressfactors,moreoutbreaksmaybeobservedattheOliverPondinthefuture.
Continuedmonitoringatlocationswithahistoryofranavirusinfectionwillallowtheeval-uationoflong-termdataonthefrequencyandreoccur-renceofranavirusinfectionsofamphibians.
TheidentificationofthisviruswithinSouthernOntarioamphibianpopulationsisasignificantanduniqueobservation.
Thedatafromthese5outbreaksstronglysuggestthatthesemaybethefirstdocu-mentedcasesofranavirusinamphibianpopulationsinOntario,andonlytheseconddocumentedoccurrenceinCanada.
Thisresearchisastartingpointforthecol-lectionoflongtermdatasetsthatwillservetofurthercharacterizethepresenceofranaviruswithinOntario.
Inthefuture,theeffectofdiseaseoncommunitystabil-ityinthefaceofconstantlychangingenvironmentalfactorswillbeofgreatimportancetoourunderstand-ingandabilitytoconserveamphibianspecies.
Acknowledgements.
FundingforA.
L.
GwasprovidedbyanOntarioGraduateScholarshipinScienceandTechnology(OGSST).
Fig.
1photographedbyT.
Howe.
ThankstoV.
G.
ChincharattheUniversiityofMississippiMedicalCenter,Jackson,MS,USA,andtheNaturalResourcesDNAProfilingandForensicCentreatTrentUniversity.
13DisAquatOrg67:9–14,2005LITERATURECITEDAlfordR,RichardsSJ(1999)Globalamphibiandeclines:aprobleminappliedecology.
AnnuRevEcolSyst30:133–165BarkerKS,DavisAT,LiB,Rollins-SmithLA(1997)Invitrostudiesofspontaneousandcorticosteroidinducedapop-tosisoflymphocytepopulationsfrommetamorphosingFrogs/RU486inhibition.
BrainBehavImmun11:19–131BollingerT,MaoJ,SchockD,BrighamRM,ChincharV(1999)Pathology,isolationandpreliminarymolecularcharacterizationofanovelIridovirusfromtigersalaman-dersinSaskatchewan.
JWildlDis35:413–429BrauerF(1995)Modelsfordiseaseswithverticaltransmissionandnonlinearpopulationdynamics.
MathBiosci128:13–24CalleryE,FangH,ElinsonRP(2001)Frogswithoutpolliwogs:evolutionofanurandirectdevelopment.
BioEssays23:233–241CareyC,CohenN,Rollins-SmithL(1999)Amphibiande-clines:animmunologicalperspective.
DevCompImmunol23:459–472ChincharVG(2002)Ranaviruses(familyIridoviridae):emerg-ingcold-bloodedkillers.
ArchVirol147:447–470ChincharVG,MaoJ(2000)Moleculardiagnosticsofirido-virusinfectionsincoldbloodedanimals.
SeminarsAvianExoticPetMed9:27–35CowleyJA,HallMR,CadoganLC,SpannKM,WalkerPJ(2002)Verticaltransmissionofgill-associatedvirus(GAV)intheblacktigerprawnPenaeusmonodon.
DisAquatOrg50:95–104DaszakP,BergerL,CunninghamAA,HyattAD,GreenD,SpeareR(1999)Emerginginfectiousdiseasesandamphibianpopulationdeclines.
EmergInfectDis5:1–23Gosner(1960)Asimplifiedtableforstaginganuranembryosandlarvaewithnotesonidentification.
Herpetologica16:183–190GreenDE,ConverseKA,SchraderAK(2002)Epizootiologyofsixty-fouramphibianmorbidityandmortalityeventsintheUSA,1996–2001.
AnnNYAcadSci969:323–339HyattAD,GouldAR,ZupanovicZ,CunninghamAA,Hengst-bergerS,WhittingtonRJ,KattenbeltJ,CouparBEH(2000)Comparativestudiesofpiscineandamphibianirido-viruses.
ArchVirol145:301–331MaoJ,HendrickRP,ChincharVG(1997)Molecularcharac-terization,sequenceanalysisandtaxonomicpositionofnewlyisolatedfishiridoviruses.
Virology229:212–220MarschangRE,BecherP,PosthausH,WildP,ThielHJ,Muller-DobliesU,KalettaEF,BacciariniLN(1999)Iso-lationandcharacterizationofaniridovirusfromHer-mann'stortoises(Testudohermanni).
ArchVirol144:1909–1922MoreySR,WithamCW,BauderET,BelkD,FerrenWR,Orn-duffR(1996)Pooldurationinfluencesageandbodymassatmetamorphosisinthewesternspadefoottoad:implica-tionsforvernalpoolconservation.
Ecology,Conservation,andManagementofVernalPoolEcosystems,ProcCalifNativePlantSoc,Sacramento,CA,p86–91Rollins-SmithLA(1998)Metamorphosisandtheamphibianimmunesystem.
ImmunolRev166:221–230SpeareR,SmithJR(1992)AniridoviruslikeagentisolatedfromtheornateburrowingfrogLimnodynastesornatusinnorthernAustralia.
DisAquatOrg14:51–57SpeareR,FreelandWJ,BoltonSJ(1991)ApossibleiridovirusinerythrocytesofBufomarinusinCostaRica.
JWildlDis27:457–462WhittingtonRJ,KearnsC,SpeareR(1997)Detectionofanti-bodiesagainstiridovirusesintheserumoftheamphibianBufomarinus.
JVirolMethods68:105–108WiseJA,HarrellSF,BuschRL,BoyleJA(1988)Verticaltrans-missionofchannelcatfishvirus.
AmJVetRes49:1506–1509ZupanovicZ,LopezG,HyattAD,GreenB,BartranG,ParkesH,WhittingtonRJ,SpeareR(1998a)GianttoadsBufomar-inusinAustraliaandVenezuelahaveantibodiesagainst'ranaviruses'.
DisAquatOrg32:1–8ZupanovicZ,LopezG,HyattAD,ShiellBJ,RobinsonAJ(1998b)Animprovedenzymelinkedimmunosorbentassayfordetectionofanti-ranavirusantibodiesintheserumofthegianttoad.
DevCompImmunol22:573–58514Editorialresponsibility:PeernelZwart,Utrecht,TheNetherlandsSubmitted:October13,2004;Accepted:June17,2005Proofsreceivedfromauthor(s):November1,2005
67:9–14,2005PublishedNovember9INTRODUCTIONThereismuchknownaboutthediseasesofwildlifewhichareeconomicallyorrecreationallyimportant.
However,thereismuchlessknownaboutthedis-easesofspeciesthatdonotfallintooneofthesetwocategories.
VirusesofthefamilyIridoviridaehavebeenincreasinglyrecognizedasacauseofamphib-ianmortalityevents(Alford&Richards1999,Greenetal.
2002).
ThefamilyIridoviridaeiscomposedoflargeDNAviruseswithicosahedralcapsids(Bollingeretal.
1999,Chinchar&Mao2000,Chinchar2002).
Withinthisvirusfamilythereare5recognizedgen-era,andRanavirusisoneofthegenerathatispatho-genictoamphibians,fishandreptiles(Daszaketal.
1999).
RanavirusinfectionshavebeenfoundwithinawiderangeofamphibianandfishpopulationsinAustralia(Speare&Smith1992),Europe(Hyattetal.
2000),Asia(Marschangetal.
1999),SouthAmerica(Speareetal.
1991,Zupanovicetal.
1998a)andNorthAmerica(Bollingeretal.
1999,Greenetal.
2002).
Themanifestationofranavirusdiseasewithinlowervertebratespeciesworldwiderangesfrominapparenttofatal,andvariesdependingonthevirusandthehostspecies(Chinchar&Mao2000).
Thisdiseaseinfrogshasbeenobservedtocausemortalityratesapproach-ing100%intadpoleandmetamorphicindividualsinseverallocations(Greenetal.
2002).
TheUnitedStatesGeologicalSurvey(USGS)hasinvestigatedmassmor-talityeventsinvolvingamphibiansandfoundthatbetween1996and2001,31of64occurrencesofamphibianillnessanddeathintheUSAcouldbeattributedtoranaviralinfection,withanadditional9%beingattributedtoranaviralinfectioncombinedwithanotheretiology(Greenetal.
2002).
ItisbecomingInter-Research2005·www.
int-res.
com*Email:amy.
greer@asu.
eduFiveamphibianmortalityeventsassociatedwithranavirusinfectioninsouthcentralOntario,CanadaAmyL.
Greer1,*,MichaelBerrill2,PaulJ.
Wilson2,31WatershedEcosystemsGraduateProgram,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B8,Canada2DepartmentofBiology,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B5,Canada3NaturalResourcesDNAProfilingandForensicCentre,TrentUniversity,1600WestBankDr.
,Peterborough,OntarioK9J7B5,CanadaABSTRACT:Usingfield,molecularandhistologicalmethods,anepizootic,systemicdiseasecausingdeathwithinwoodfrogRanasylvaticatadpolesandleopardfrogRanapipiensmetamorphsat3dif-ferentlocationswithinSouthernOntario,Canada,hasbeeninvestigated.
Ourresultsdemonstratedthattheprobablecauseofthisdiseasewasaranavirus.
Affectedamphibianswerefoundtoexhibitnecrosiswithinthehematopoieticcells.
LivertissuesampleswerefoundpositiveforthevirusbyPCRamplificationoftheranavirus(Family:Iridoviridae)majorcapsidprotein(MCP).
Positivesampleswereconfirmedbysequenceanalysis.
Clinicallynormal,laboratory-raisedwoodfrogeggbroodswerealsofoundtotestweaklypositiveforranavirus.
Thepopulationeffectsofdiseaseontheseamphibiancommunitieshavenotyetbeenconclusivelyassociatedwithpopulationdeclines,butwar-rantmorefocusedconsideration.
KEYWORDS:Frog·Ranasylvatica·Ranapipiens·Ranavirus·Amphibian·Polymerasechainreaction·CanadaResaleorrepublicationnotpermittedwithoutwrittenconsentofthepublisherDisAquatOrg67:9–14,2005apparentthattheinfectionanddeathofdiversegroupsofamphibianstestingpositiveforranavirusisaseriousandpotentiallydevastatingoccurrence.
Wereporthereontherecurringpresenceofaranavirusin3amphibianpopulationsinSouthernOntario,Canada.
Theobjectivesforthisworkweretodeterminewhetheraranaviruswasassociatedwith5amphibianmortalityeventsinvestigatedinsouthcentralOntario,Canada.
Wealsoexaminedpotentialtransmissionroutesfortheviruswithinthesepopulations.
MATERIALSANDMETHODSFieldsites.
Tadpolesandrecentmetamorphswerecollectedfrom5differentsiteswithinSouthernOntario.
Fieldsitescanbeclassifiedinto2categories,namely(1)siteswherediseasewasobservedandsub-sequentlyinvestigated,and(2)siteswherenodiseasewasobserved.
DiseasesitesincludedtheJamesOliverEcologicalResearchStation('OliverPond';44°31'N,78°32'W),TheKortrightCentreforConservation('KortrightPond';43°57'N,79°42'W)andGannon'sNarrows(44°38'N,78°41'W).
SitesthatshowednosignsofdiseaseandwereusedascontrolsitesincludedthePoplarPond(44°31'N,78°32'W)andtheTrentUniversityArcheologyPond(44°21'N,78°17'W).
Tobeconsideredadiseasedsite,≥10ind.
hadtomeetthecasedefinition.
Thismeantthatsickand/ordeadindividualshadtoexhibit4of7clinicalsigns.
Clinicalsignsincludedrednessinthevent,abdomen,orlegs,swollenvent,edemaofthebodycavity,cutaneouslesions,anorexiaandlossofescapebehaviour.
TheOliverPondlocationisuniquebecausetherehavebeen3consecutiveyearsofsimilarmassmortalityeventshere.
InMay2002,RanasylvaticaeggmasseswerecollectedfromtheOliverPondwithin12hofbeinglaidandrearedinthelaboratoryin5gallon(19l)aquariacontainingagedriverwater.
Collectionofanimals.
Liveanimalsweretrans-portedbacktothelaboratory.
Individualsfoundatthefieldsitethatwerealreadydeadweretransportedbacktothelaboratoryonice,andindividualsthatwerealiveanddemonstratingsignsofdiseaseweretrans-portedaliveandtheneuthanizedbyadministeringanoverdoseoftricainemethanesulfonate(SyndelLabora-tories).
Liversweredissectedfromallindividualsanddatawerecollectedonoverallbodyconditioninclud-ingfatbodies,organcolourationandobservablelesions.
Equipmentwasdecontaminatedusingacon-centratedbleachsolutionbeforemovingbetweenfieldsites(Green&Barry2001).
Animalhusbandry.
Laboratory-raisedtadpoleswerefedcookedspinachandgoldfishflakesdaily.
Allaquariawereconstantlyaerated.
Individualswereeuthanizedatstage42byadministeringanoverdoseoftricainemethanesulfonate(SyndelLaboratories).
Histopathology.
Livertissuesampleswerepro-cessedforhistologyusingstandardhistologicaltech-niques.
Eachsamplewasseriallysectionedat7m,andstainedwithhematoxylinandeosin.
Sectionswereexaminedunderalightmicroscopeforpatho-logicalfindings(Bollingeretal.
1999,Marschangetal.
1999).
Moleculardiagnostics.
DNAwasisolatedfromfrozenandpreservedlivertissuebiopsysamplesfrombothhealthyanddiseasedindividuals.
Tissueswerelysedin500lof1*lysisbufferand25lofProteinaseK(14.
4mgml–1,>60.
0U100l–1).
Sampleswereincu-batedinabeakerof65°Cwaterina37°Cincubator.
Af-ter1h,asecond25lofProteinaseKwasaddedandsampleswereincubatedat37°Covernight.
ExtractionsoftotalcellularDNAwerecompletedusingthestandardQIAampDNeasyProtocolforAnimalTissue(Qiagen).
Amplificationwasconductedusingprimersspecificfora500bpfragmentoftheranavirusmajorcapsidprotein(MCP)(Maoetal.
1997)in10lPCRreactions.
Thermocyclingconditionsweresimilarforallsamples(94°C5min,94°C30s,55°C30sand72°C30s,cycled35timesfollowedbyanextensionof72°C2min)withtheexceptionoftheannealingtemperaturethatwasoptimizedat60°Cforleopardfrogsamples.
Bothpositivecontrols(plasmidDNAwithRanavirusMCPinsertobtainedfromV.
G.
Chinchar,Univ.
ofMissis-sippiMedicalCenter,Jackson,MS,USA)andnegativecontrolsweretestedalongsideallunknownsamples.
Allamplifiedproductwasvisualizedbyelectrophoresison1.
5%agarosegels.
Sequencesoftheamplifiedpro-ductwerecomparedtopreviouslypublishedsequencesusingtheNCBIBlastSearchnucleotidedatabase.
ClustalmultiplealignmentsforviralsampleswereconductedusingBioEditsequencealignmenteditorsoftwareandthenrealignedwithpublishedFrogVirus3(ranavirus)sequences.
RESULTSFieldobservationsSince1999,massmortalityeventsinvolvingtadpolesandnewmetamorphshavebeeninvestigatedwithinOntario.
Intotal,5mortalityeventshavebeeninvesti-gated.
Theseeventsinvolved2differentspeciesofamphibiansin3differentlocationswithinOntario,andoneoftheselocationshasexperiencedareoccurringepidemicstate.
Ineachlocationmorethan50individ-ualsmeetingthecasedefinitionwereidentified.
AttheOliverPond,Ranasylvaticaexperiencedmassmortal-10Greeretal.
:Amphibianmortalityandranavirusityeventsinvolvingmorethan100individualsinJuneof1999,2000and2001.
Morethan50RanasylvaticawerefounddeadattheKortrightPondinJune2001,andover100RanapipiensmetamorphswerefounddeadatGannon'sNarrowsinAugust2002.
Necropsyobservationsincludededemaofthebodycavity,poorfatbodycomposition,paleandmottledcolourationoftheliverandsystemichemorrhage(Fig.
1).
Cutaneouslesionsandulcerswerenotobserved.
Inallmassmor-talityevents,only1speciesofamphibianwasinvolveddespitethepresenceofadiverseamphibiancommu-nity.
In4eventsonlytadpoleswereinfected,andtheseindividualsrangedfromGosnerstages40to45(Gos-ner1960).
Recentmetamorphswereafflictedbyonly1mortalityevent,andnomorbidordeadadultswereobservedatanyofthelocations.
All5ofthediseaseoutbreaksinOntariohadonsetswithinashorttime-framebetweenJuneandAugust.
NomassmortalityeventswereinvestigatedbetweenSeptemberandMay.
Anaccuratetotalnumberofindividualsthatsuccumbedtoinfectionateachlocationisunknown.
However,during2000and2001nonewwoodfrogmetamorphswereobservedattheOliverPond.
Also,nomortalityeventwasobservedattheOliverPondduring2002after3previousevents.
Histologicalexaminationandpoly-merasechainreactionanalysis(PCR)(Table1)havecontributedtotheiden-tificationofthepresenceofaranaviruswithintheseindividuals.
Sixtypercentoftheamphibianmortalityeventsinvestigatedwereassociatedwiththepresenceofaranavirus(Family:Irido-viradae)andoccurredattheOliverPondin2001,KortrightCentrein2001andGannon'sNarrowsin2002.
Theremaining40%ofcasesaresuspectedtobeassociatedwiththissamevirusbasedonsimilarclinicalsigns,locationandepidemiologicaldatadespitealackofsuitablesamplesfortissuehistologyorPCRidentification.
TheseincludemortalityeventsobservedattheOliverPondin1999and2000.
Thedurationofthesedie-offeventshasrangedfromseveraldaystoseveralweeks.
HistopathologyHistologicalexaminationsoflivertissueyieldedsev-eralfindings.
DiseasefreeleopardfrogmetamorphsfromtheTrentUniversityArcheologyPondwerefoundtoexhibitnormalcellularstructureoftheliver.
Livertissueintheseindividualsdidnotsuggestanytypeofpathologyandwassubsequentlyusedascontroltisse(Fig.
2).
Livertissuesectionsfromleopardfrogmeta-morphscollectedfromadie-offeventatGannon'sNar-rowswerefoundtoexhibitpathologicalchangesandsystemicnecrosisofthehematopoietictissues.
In-flammatorycellswereobservedintheportaltracts,aswasballooningdegenerationwithinthehepatocytes(Fig.
3).
AllleopardfrogliversexaminedwerematchedwithcontrolliversbyGosnerstagetocontrolforanyeffectsofmetamorphosis.
11Fig.
1.
Ranasylvatica.
ExampleofasystemichemorrhageLocationYearNo.
ind.
meetingAbnormalInclusionbodies%samplespositivecasedefinitionliverpathologybyPCROliverPond(field)2001>100NDND71KortrightPond(field)2001>50NDND93OliverPond(lab-raised)20020+–60OliverPond(field)20020+–70Gannon'sNarrows(field)2002>100++82ArcheologyPond(field)20020––0Table1.
DiagnosticcriteriaforamphibianmortalityeventsinsouthcentralOntario,Canada.
+:positiveresult;–:negativeresult;ND:nodataavailableDisAquatOrg67:9–14,2005MoleculardiagnosticsLivertissuescollectedfromindividualsmeetingthecasedefinitionfromtheOliverPond2001,KortrightCentre2001,andGannon'sNarrows2002eventswerefoundtobePCRpositivewhencomparedtoaknownpositivecontrol(ranavirusMCPinsertinplasmidDNAobtainedfromV.
G.
Chinchar).
TheranavirusMCP-codingregionwasalsoidentifiedinlivertissueobtainedfromindividualsattheOliverPond2002(bothpond-raisedandlaboratory-raised)thatdidnotmeetthecasedefinition.
Thesesamplesshowednoobservablediseasecharacteristicsbutthepopulationhadahistoryofpreviousmortalityeventsthatdidmeetthecasedefinitionandhadthereforeexperiencedmortalityeventsinthepast.
However,individualsfromtheArcheologyPond,wherethathavebeennoobser-vationsofunusualmorbidityormortality,werefoundtotestnegativebyPCR.
RepresentativesamplesfromOliverPond2001,OliverPond2002,KortrightPond2001,laboratory-raisedOliverPond2002andGannon'sNarrows2002wereconfirmedpositiveforranavirusMCPcodingDNAbysequenceanalysis.
SequenceanalysisrevealedthatallMCPsequences(~500bp)werea98%matchtoranavirus(FrogVirus3)MCPandalsoindicatedthattheMCPsequencesdidnotdifferamongindividualsorlocations.
DISCUSSIONBasedonfield,histologicalandmolecularinvestiga-tions,aranavirusmaybeassociatedwithmorbidityandmortalitywithinOntarioamphibianpopulations.
Theclinicalsignsofdiseaseintheindividualsexam-inedhavesimilaritiestootherdocumentedobserva-tionsfromranavirusinfectionsites(Careyetal.
1999,Chinchar&Mao2000,Chinchar2002).
Thedatageneratedduringthisstudysupportpre-viousfindingsthatlatestagetadpolesandnewmeta-morphsappearmoresusceptibletoranavirus(Greenetal.
2002).
Althoughthepathologicalfindingsappeartobesignificantlydifferentbetweennon-infectedandinfectedRanapipiensmetamorphs,itisimportanttoconsiderthatmetamorphosismaycausechangestotheliverregardlessofviralstatus.
However,thisseems12Fig.
2.
Ranapipiens.
LivercrosssectionfromanormalmetamorphcollectedfromTrentUniversityArcheologyPond.
Notearrangementsofthehepatocytesandsinusoids(S)containingbloodcells,radiatingthroughoutFig.
3.
Ranapipiens.
LivercrosssectionfromaGannon'sNarrowsmetamorph.
Ballooningdegeneration(hypertrophy)canbeobservedandischaracterizedbyswellingofcells.
Thisswellingcausedsinusoidstobecomeblocked.
NoopensinusoidscanbeobservedGreeretal.
:Amphibianmortalityandranavirusunlikelysincecontrolliverswerematchedwithexper-imentalliversofthesamestageanddifferenceswerestillobserved.
Metamorphosismayplaceamphibiansatagreaterriskofclinicaldiseasefrompossiblerana-virusinfectionbecauseofthepotentialfortransitoryimmunedeficiencytooccurduringthistimeframe.
Metamorphosisisarguablyoneofthemorestressfuleventsinthelifecycleofananuran(Barkeretal.
1997,Rollins-Smith1998,Calleryetal.
2001).
Ithasbeensuggestedthatdiminishedresilienceoftheimmunesystemduringthetransitionbetweentadpoleandadultmaybeacontributingfactortothesusceptibilityoftheseamphibianstopathogensattheclimaxofmetamorphosis(Moreyetal.
1996);duringmetamor-phosistheremaybeawindowofdecreasedimmunefunction,atwhichtimetheseamphibiansmaybemorevulnerabletoinfectionsresultingfromapathogenicvirus.
Viralpositiveswereobservedinlab-rearedclini-callynormalindividualscollectedaseggsfromloca-tionswithahistoryofpreviousmortalityevents.
Indi-vidualsinthiscategoryofinfectionshowednosignsofmorbidityormortality.
Theonlylinkingfactortoranaviruswasthehistoricalexposureoftheparentfrogs.
Thissuggeststhatinanurans,thepossibilityofverticaltransmissionoftheviruscannotberuledout,norcantheselectionforresistanceamongadultfrogspreviouslyexposed.
Therearenopublishedstudiesthatevaluatethepossibilityofverticaltransmissionofranavirus.
However,therearereportsofotherDNAviruses,suchasgillassociatedvirusandchannelcat-fishvirus,thathavedemonstratedverticaltransmis-sionofvirusfromadultstoeggs(Wiseetal.
1988,Brauer1995,Cowleyetal.
2002).
Itispossiblethatverticaltransmissionisoccurringinanuransinfectedwithranavirusbasedondatacollectedfromthisresearch.
TherewasnomorbidityormortalityobservedattheOliverPondin2002despite3annualreoccurrences.
Literaturesourcesindicatethatinsomeamphibianpopulations,ranavirusinfectionbecomesanannuallyreoccurringproblem(Careyetal.
1999).
Theobserva-tionthattheOliverPonddidnothaveanoutbreakin2002issignificant.
Firstly,itispossiblethatasmallnumberoftadpolesexposedtothepathogenduringdie-offeventsin1999,2000or2001didnotperishduetoinherentresistance.
Theseanimalswouldthenbetheanimalsobservedbreedingthefollowingyear(Whittingtonetal.
1997,Zupanovicetal.
1998a,b).
ThepresentresultsfortheOliverPondareconsistentwithwhatwouldbeexpectedafterseveralyearsofstrongselectionforresistance.
Afterseveralyearsofthisselection,theresultingprogenymaynolongerbepronetothediseasealthoughtheymaycarrythevirusatlowlevels.
Secondly,theOliverPondexperiencedavarietyofdifferentweatherconditionsduringthespringof2002.
Therefore,itispossiblethatthediseasewasnotobservedin2002becausethepopulationwasnotunderthesameamountofstressaspreviousyears.
TherewereconsiderabledifferencesinthewaterleveloftheOliverPondfrom1999to2002,withverylowwaterlevelsobservedduringthesummerof1999to2001andveryhighwaterlevelsobservedin2002.
Dur-ingtimesoflowwaterlevels,thepopulationdensityisincreasedwhichincreasestheprobabilitythatsuscep-tibleindividualswillencounterenough(1)infectiousagentinthewateror(2)infectiousindividualstosuc-cumbtoinfection.
Duringtimesofhighwaterandlowpopulationdensities,itwouldbeexpectedthattheprobabilityofsusceptibleindividualsencounteringinfectedindividualswouldbelowand,asaresult,massmortalitieswouldnotoccurwithinthepopula-tion.
Thesetheoriesregardingtheobservedlackofdis-easeattheOliverPondin2002allowustomakepre-dictionsaboutwhatmaybeobservedinyearstocome.
Ifthediseasewasnotobservedbecauseofselectionforresistance(strongherdimmunity)in2002,thediseasemaynotoccuragainattheOliverPond,ormaybereducedinitsprevalenceevenifthepondexperiencesabioticstress.
Thisselectionforimmunitysuggeststhatthediseaseitselfmaynotbeamajorconservationissue,ashasbeenpreviouslysuggested.
Alternatively,ifthepopulationisnotstronglyselectedforimmunityandtheoutbreaksaretrulyunderthecontrolofstressfactors,moreoutbreaksmaybeobservedattheOliverPondinthefuture.
Continuedmonitoringatlocationswithahistoryofranavirusinfectionwillallowtheeval-uationoflong-termdataonthefrequencyandreoccur-renceofranavirusinfectionsofamphibians.
TheidentificationofthisviruswithinSouthernOntarioamphibianpopulationsisasignificantanduniqueobservation.
Thedatafromthese5outbreaksstronglysuggestthatthesemaybethefirstdocu-mentedcasesofranavirusinamphibianpopulationsinOntario,andonlytheseconddocumentedoccurrenceinCanada.
Thisresearchisastartingpointforthecol-lectionoflongtermdatasetsthatwillservetofurthercharacterizethepresenceofranaviruswithinOntario.
Inthefuture,theeffectofdiseaseoncommunitystabil-ityinthefaceofconstantlychangingenvironmentalfactorswillbeofgreatimportancetoourunderstand-ingandabilitytoconserveamphibianspecies.
Acknowledgements.
FundingforA.
L.
GwasprovidedbyanOntarioGraduateScholarshipinScienceandTechnology(OGSST).
Fig.
1photographedbyT.
Howe.
ThankstoV.
G.
ChincharattheUniversiityofMississippiMedicalCenter,Jackson,MS,USA,andtheNaturalResourcesDNAProfilingandForensicCentreatTrentUniversity.
13DisAquatOrg67:9–14,2005LITERATURECITEDAlfordR,RichardsSJ(1999)Globalamphibiandeclines:aprobleminappliedecology.
AnnuRevEcolSyst30:133–165BarkerKS,DavisAT,LiB,Rollins-SmithLA(1997)Invitrostudiesofspontaneousandcorticosteroidinducedapop-tosisoflymphocytepopulationsfrommetamorphosingFrogs/RU486inhibition.
BrainBehavImmun11:19–131BollingerT,MaoJ,SchockD,BrighamRM,ChincharV(1999)Pathology,isolationandpreliminarymolecularcharacterizationofanovelIridovirusfromtigersalaman-dersinSaskatchewan.
JWildlDis35:413–429BrauerF(1995)Modelsfordiseaseswithverticaltransmissionandnonlinearpopulationdynamics.
MathBiosci128:13–24CalleryE,FangH,ElinsonRP(2001)Frogswithoutpolliwogs:evolutionofanurandirectdevelopment.
BioEssays23:233–241CareyC,CohenN,Rollins-SmithL(1999)Amphibiande-clines:animmunologicalperspective.
DevCompImmunol23:459–472ChincharVG(2002)Ranaviruses(familyIridoviridae):emerg-ingcold-bloodedkillers.
ArchVirol147:447–470ChincharVG,MaoJ(2000)Moleculardiagnosticsofirido-virusinfectionsincoldbloodedanimals.
SeminarsAvianExoticPetMed9:27–35CowleyJA,HallMR,CadoganLC,SpannKM,WalkerPJ(2002)Verticaltransmissionofgill-associatedvirus(GAV)intheblacktigerprawnPenaeusmonodon.
DisAquatOrg50:95–104DaszakP,BergerL,CunninghamAA,HyattAD,GreenD,SpeareR(1999)Emerginginfectiousdiseasesandamphibianpopulationdeclines.
EmergInfectDis5:1–23Gosner(1960)Asimplifiedtableforstaginganuranembryosandlarvaewithnotesonidentification.
Herpetologica16:183–190GreenDE,ConverseKA,SchraderAK(2002)Epizootiologyofsixty-fouramphibianmorbidityandmortalityeventsintheUSA,1996–2001.
AnnNYAcadSci969:323–339HyattAD,GouldAR,ZupanovicZ,CunninghamAA,Hengst-bergerS,WhittingtonRJ,KattenbeltJ,CouparBEH(2000)Comparativestudiesofpiscineandamphibianirido-viruses.
ArchVirol145:301–331MaoJ,HendrickRP,ChincharVG(1997)Molecularcharac-terization,sequenceanalysisandtaxonomicpositionofnewlyisolatedfishiridoviruses.
Virology229:212–220MarschangRE,BecherP,PosthausH,WildP,ThielHJ,Muller-DobliesU,KalettaEF,BacciariniLN(1999)Iso-lationandcharacterizationofaniridovirusfromHer-mann'stortoises(Testudohermanni).
ArchVirol144:1909–1922MoreySR,WithamCW,BauderET,BelkD,FerrenWR,Orn-duffR(1996)Pooldurationinfluencesageandbodymassatmetamorphosisinthewesternspadefoottoad:implica-tionsforvernalpoolconservation.
Ecology,Conservation,andManagementofVernalPoolEcosystems,ProcCalifNativePlantSoc,Sacramento,CA,p86–91Rollins-SmithLA(1998)Metamorphosisandtheamphibianimmunesystem.
ImmunolRev166:221–230SpeareR,SmithJR(1992)AniridoviruslikeagentisolatedfromtheornateburrowingfrogLimnodynastesornatusinnorthernAustralia.
DisAquatOrg14:51–57SpeareR,FreelandWJ,BoltonSJ(1991)ApossibleiridovirusinerythrocytesofBufomarinusinCostaRica.
JWildlDis27:457–462WhittingtonRJ,KearnsC,SpeareR(1997)Detectionofanti-bodiesagainstiridovirusesintheserumoftheamphibianBufomarinus.
JVirolMethods68:105–108WiseJA,HarrellSF,BuschRL,BoyleJA(1988)Verticaltrans-missionofchannelcatfishvirus.
AmJVetRes49:1506–1509ZupanovicZ,LopezG,HyattAD,GreenB,BartranG,ParkesH,WhittingtonRJ,SpeareR(1998a)GianttoadsBufomar-inusinAustraliaandVenezuelahaveantibodiesagainst'ranaviruses'.
DisAquatOrg32:1–8ZupanovicZ,LopezG,HyattAD,ShiellBJ,RobinsonAJ(1998b)Animprovedenzymelinkedimmunosorbentassayfordetectionofanti-ranavirusantibodiesintheserumofthegianttoad.
DevCompImmunol22:573–58514Editorialresponsibility:PeernelZwart,Utrecht,TheNetherlandsSubmitted:October13,2004;Accepted:June17,2005Proofsreceivedfromauthor(s):November1,2005
- Laboratories22pipi.com相关文档
- Balakrishna22pipi.com
- 注册22pipi.com
- Stress22pipi.com
- Rabbit22pipi.com
- selected22pipi.com
- Capacity22pipi.com
丽萨主机122元/每季,原生IP,CN2 GIA网络
萨主机(lisahost)新上了美国cn2 gia国际精品网络 – 精品线路,支持解锁美区Netflix所有资源,HULU, DISNEY, StartZ, HBO MAX,ESPN, Amazon Prime Video等,同时支持Tiktok。套餐原价基础上加价20元可更换23段美国原生ip。支持Tiktok。成功下单后,在线充值相应差价,提交工单更换美国原生IP。!!!注意是加价20换原生I...

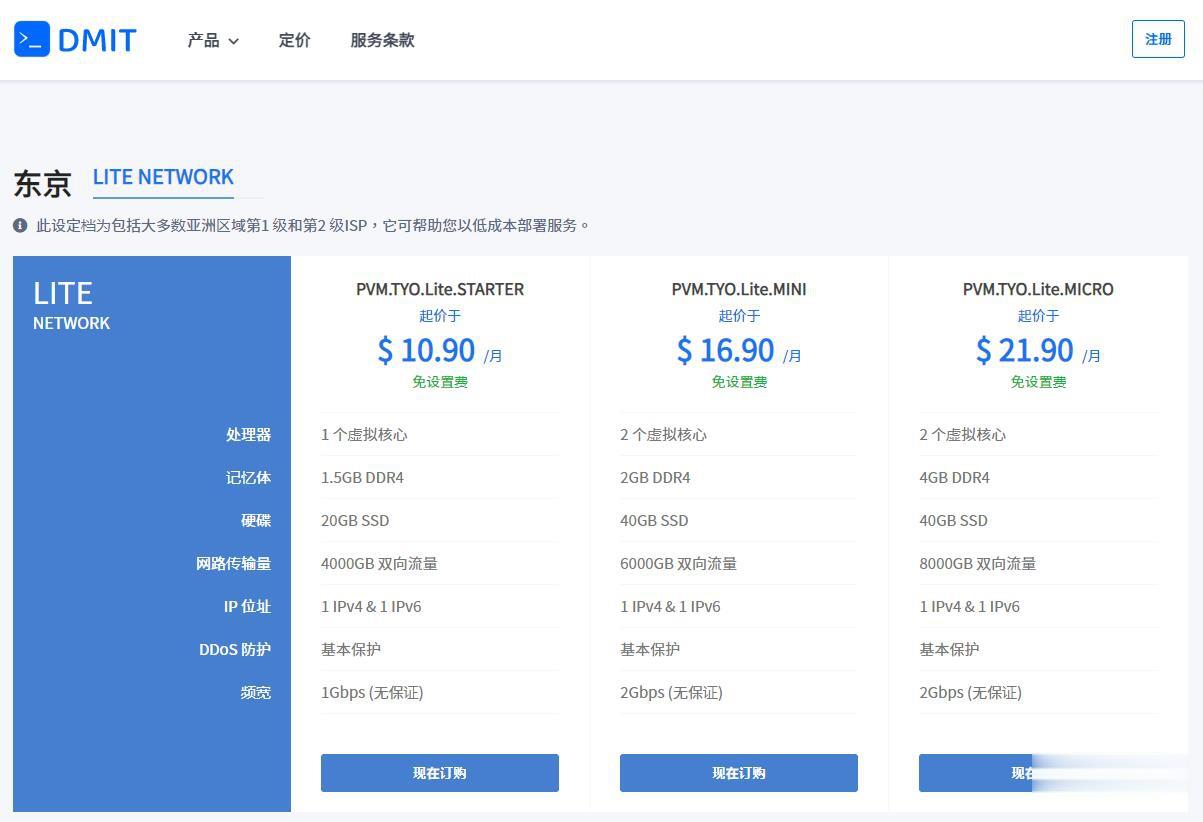
DMIT$10.9/月,日本VPS/三网直连/1核1.5G内存/20GB存储/1Gbps端口
优惠码年付一次性5折优惠码:TYO-Lite-Open-Beta-1y-50OFF永久8折优惠码:TYO-Lite-Open-Beta-Recur-20OFF日本vpsCPU内存SSD流量带宽价格购买1核1.5G20 GB4 TB1Gbps$10.9/月购买2核2 G40 GB6 TB1Gbps$16.9/月购买2核4 G60 GB8 TB1Gbps$21.9/月购买4核4 G80 GB12 TB...
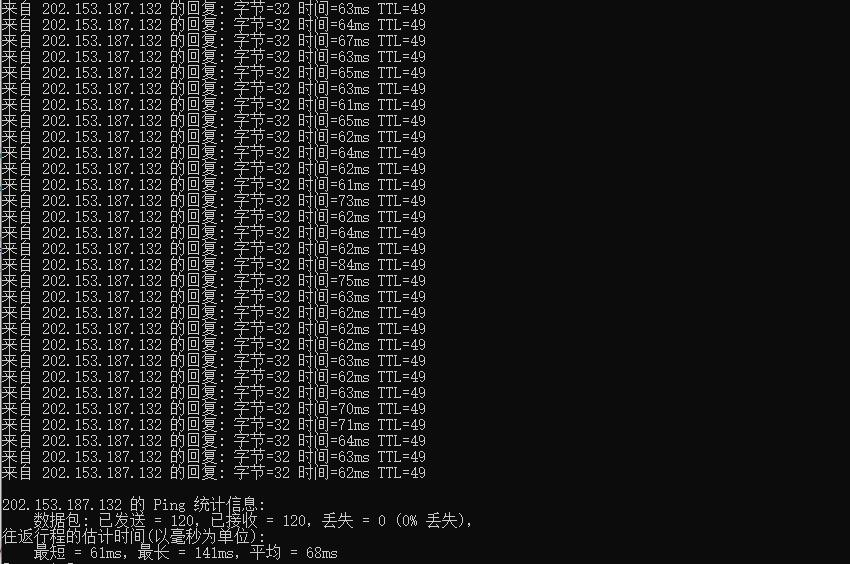
酷番云78元台湾精品CN2 2核 1G 60G SSD硬盘
酷番云怎么样?酷番云就不讲太多了,介绍过很多次,老牌商家完事,最近有不少小伙伴,一直问我台湾VPS,比较难找好的商家,台湾VPS本来就比较少,也介绍了不少商家,线路都不是很好,有些需求支持Windows是比较少的,这里我们就给大家测评下 酷番云的台湾VPS,支持多个版本Linux和Windows操作系统,提供了CN2线路,并且还是原生IP,更惊喜的是提供的是无限流量。有需求的可以试试。可以看到回程...
22pipi.com为你推荐
-
网易网盘关闭入口网易网盘怎么打不开了中老铁路中国有哪些正在修的铁路广东GDP破10万亿广东省2019年各市gdp是多少?甲骨文不满赔偿工作不满半年被辞退,请问赔偿金是怎么算的?百度关键词价格查询百度推广里怎么查指定的关键字参与竞价的价位呢网站检测请问,对网站进行监控检测的工具有哪些?haokandianyingwang有什么好看的电影网站www.kanav001.com长虹V001手机小游戏下载的网址是什么www.zjs.com.cn请问宅急送客服电话号码是多少?www.se222se.com请问http://www.dibao222.com这个网是做什么