preparedwww.55fang.com
www.55fang.com 时间:2021-04-08 阅读:()
2010WILEY-VCHVerlagGmbH&Co.
KGaA,Weinheim4814www.
advmat.
dewww.
MaterialsViews.
comCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818ByYuhuaXue,HongxiaWang,YanZhao,LimingDai,LianfangFeng,XungaiWang,andTongLin*MagneticLiquidMarbles:A"Precise"MiniatureReactor[*]Dr.
Y.
Xue,Dr.
H.
Wang,Dr.
Y.
Zhao,Prof.
X.
Wang,Prof.
T.
LinCentreforMaterialandFibreInnovationDeakinUniversityGeelong,VIC3217(Australia)E-mail:tong.
lin@deakin.
edu.
auProf.
L.
DaiDepartmentofChemicalEngineeringCaseWesternReserveUniversityCleveland,Ohio44106(USA)Prof.
L.
FengStateKeyLaboratoryofChemicalEngineeringDepartmentofChemicalandBiologicalEngineeringZhejiangUniversityHangzhou310027(P.
R.
China)DOI:10.
1002/adma.
201001898Miniaturizedchemicalprocesseshavemanyadvantages,suchasreduceduseofchemicalreagentsandsolvents,preciselycontrolledreactionconditions,muchshortenedreactiontime,andtheabilitytointegrateintoadigitaldevice.
[1–4]Theyareveryusefulforhigh-throughputanalysesandpuricationsinchemicalandbiologicalprocesses,suchasdrugdiscovery,[5]DNAanalysis,[6,7]proteincrystallization,[8]andthesynthesisofmoleculesorparticles.
[9–11]Indeed,considerableeffortshavebeenmadetominiaturizechemicalprocessesusingvariousprinciples.
Forexample,oil/wateremulsionshavebeenusedtoperformchemicalreactionsinparallelinalargenumberofemulsiedtinydroplets.
[10]Microuidic"lab-on-a-chip"deviceshavebeendesignedtomanipulatechemicalprocesseswithinmicro-channelseitherincontinuousuidordisconnecteduidsegmentsseparatedbyanimmiscibleuidorgas.
[10,12]Althoughthechannel-basedmicro-reactorsshowgreatpoten-tialinhigh-throughputreactionandintegratabilitytoexternalanalysisfacilities,thereaction-specicdevicepreparations,thechannel-associatedcross-contaminations,andtheneedforexternalpumpstoactuatetheuidmotionmakethemneitheruniversalnorminiature.
Also,microuidicsishardtousetohandleasingledroplet.
Recently,freeaqueousdropletsinanimmiscibleuid(channel-freemicrouidics)[9]hasalsobeenproposedasanalternativestrategy,butcross-contaminationstillremainsaconcern.
Inlivingsystems,cellsarewell-knownasthebasicstructuralandfunctionalunits[13]thatmediatevariousbiochemicalreac-tions(e.
g.
,catabolismandanabolism)forsustainingthegrowthanddivisionofcellsandhencelivingsystems.
Althoughtheirstructurelooksverysimple,cellscommunicatewitheachotherinahighlyintelligentway,throughoutthewholelivingsystem.
Inthisregard,howtomimiclivingcells,bothinstructureandfunction,istheultimatechallengetowardthedevelopmentofarticialminiaturereactors.
Liquidmarbles,liquiddropsencapsulatedwithsolidpowderattheliquid–gasinterface,haverecentlybeenreportedasanewapproachtomanipulatingliquid,[14]whichcouldbeusedasminiaturereactors.
Theyarenon-wettingtoanysolidsur-facesandcouldbemanipulatedbyexternalforces,suchasgravity,[14]electrical,[15]andmagnetic,[16]dependingontheircomponents.
[14–19]However,mostliquidmarblesreportedsofarhavebeenbasedonaqueousliquid.
Althoughafewpapers[19]havereportedtheencapsulationoforganicliquidsintoliquidmarbles,theliquidswereconnedtothosehavingahighsur-facetension.
[19]Theformationofstableliquidmarblesfromawiderangeoforganicsolvents,especiallythosethathavealowsurfacetension,isimportantforchemicalreactions,butstillremainsachallenge.
Inthisstudy,wedemonstratetheincredibleabilityofauor-inateddecylpolyhedraloligomericsilsesquioxane(FD-POSS,structureshowninFigure1a)anditscombinationwithhydro-phobicmagneticnanoparticlestoactasencapsulatingagentstopreparestableliquidmarblesfrombothaqueoussolutionandorganicliquidswithasurfacetensionaslowas20.
1dynecm1,andthefeasibilityofusingthemagneticliquidmarblesasuni-versal"smart"miniaturereactors.
FD-POSSwassynthesizedbythereportedmethod[20,21]andaneFD-POSSpowderwaspreparedbyagrindingtechnique.
TheFD-POSS/Fe3O4nanoparticlecompositepowdersofdif-ferentratios(1:10to1:1,w/w)werepreparedbyco-dispersingtheneFD-POSSpowderwithhydrophobicFe3O4nanopar-ticlesinethanol.
AsshowninthephotographsinFigure1b,FD-POSSpowdersarewhiteandhaveacertaintransparencytovisiblelightbutarenotresponsivetoamagneticeld.
Inethanol,FD-POSSandFe3O4nanoparticleshadastrongafnity,andtheytendedtoaggregateintopowderswhichshowedasuperparamagneticproperty(SupportingInfor-mation).
TheopticalmicrographsinFigure1cindicatethatFD-POSSandFD-POSS/Fe3O4compositepowdersarelessthan70μm,andtheFD-POSSandFe3O4nanoparticlesformedarandomlyblendedcompositestructure.
Whenathinbedofthepowderswerecoatedonasolidsurface,thesurfacebecamehighlyrepellenttoliquids.
Water,dimethylsulfoxide(DMSO),toluene,hexadecane,andethanolallformedrounddropletsonthepowdercoatedsurfacewithoverallcontactanglesof171.
1°,166.
2°,155.
7°,154.
4°,and142.
8°,respectively(Figure1d).
Thepowderlayershowedstrongrepellencetootherorganicsolvents(SupportingInformation).
ThecompositepowdersinthestudiedFD-POSS/Fe3O4ratiorangeshowedaverysimilaruidrepellencytothepureFD-POSSgrains.
Liquidmarbleswerepreparedsimplybydroppingasmallvolumeofliquidontothepowdersurfaceandsubsequentlyshakingthedropletgentlysothatthepowderscoveredtheentiredropletsurface.
Theas-preparedpowders,bothFD-POSS4815www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818Severalwaterdropletswereaddedtotheopenedmarble(10μL)toformawater-in-oilbicomponentliquidmarble.
Oil-in-waterbicomponentliquidmarblescanalsobeproducedinasimilarway.
Thesebicomponentliquidmarblesshowtheabilitytopre-ventthecoreliquidfromrapidevaporation.
Todemonstratetheabilitytomediatechemicalreactions,weusedtwodifferentwaystoundertakeachemiluminescencereaction.
[22,23]Onemethodusedtwomagneticliquidmarblestocarrydifferentreactiveagents,oneforhydrogenperoxideandtheotherforbis(2,4,6-trichlorophenyl)oxalateanddye.
AsshowninFigure2f(toppanel),thetwoliquidmarbleswereactuatedunderamagneticeldtomovetogetherandtheythencoalescedintoalargerliquidmarble,initiatingthechemilumi-nescencereactionwhichcanbeprovenbyanimmediatelightemissionfromthecoalescedliquidmarble.
Anothermethodinvolvedusingabicomponentliquidmarbletohosttwocoredroplets,onefortheoxidantandtheotherfortheuorescer,inanimmiscibleuid.
Undergentlemovementactuatedbyamagnetbar,thetwocoredropletsjointedtogethertriggeringthechemiluminescencereaction.
TheimageinFigure2f(bottompanel)clearlyindicatesthelightemittingjustfromthecombinedcoredroplet.
Besidesthechemiluminescencereaction,otherchemicalreactionssuchasphotochemicalpolymerization,nanoparticlesynthesis,andanacid–basereactionhavealsobeentrialedandproventobesuccessful(SupportingInformation).
Oneimportantaspectforminiaturechemicalreactorsistheabilitytointegratewithpuricationandanalysisfacilities.
SincethemagneticliquidmarblescanbeopenedandclosedreversiblyandFD-POSS/Fe3O4composite,canencapsulatenotonlywaterbutalsoorganicsolventstoformstableliquidmarbles.
Figure2ashowsphotosofliquidmarblespreparedfromwater,DMSO,toluene,ethanol,andoctane(3μL).
Manyotherorganicsol-ventshavealsobeentestedsofarandthepowderswereabletoencapsulatealiquidofsurfacetensionaslowas20.
1dynescm1(SupportingInformation).
Theliquidmarblesareverystableandtheycanoatnotonlyonwaterbutalsoonauidoflowsurfacetension(Figure2b).
ThesuperparamagneticpropertyoftheFe3O4nanoparticlesimpartstheFD-POSS/Fe3O4powder-encapsulatedliquidmar-bleswithamagnetresponsiveability.
Asaresult,theliquidmarblescanbeactuatedtomoveindifferentdirections,oropenedandclosedreversibly,underamagneticeld.
Figure2cshowsmagnetopenedliquidmarblesfromdifferentliquids.
Theopenedliquidmarbleallowsittobecoveredwithanothertypeofhydrophobicmaterial.
Todemonstratethis,weusedpureFD-POSSpowdertocovertheopenedliquidsurfaceofamagneticliquidmarble,andthecoveredpartshowedadifferentcolortothemagneticpowdercoveredsurface.
Consequently,themotionofliquidcanbetracedeasily.
Figure2dillustratesthemotionofawhiteFD-POSS-stainedFD-POSS/Fe3O4hexa-decanemarbleunderamagneticeld.
Itcanbeclearlyseenthatthemarblerotatesonasolidsurface(toppanel),butshiftssidewaysonaliquidsurface(bottompanel).
Themagnetopenedliquidmarblealsoallowstheliquidtobetakenfromthemarbleorthemarbletobereplenishedwithotherliquids.
Figure2edemonstratestheadditionofcolorfulwaterdropletsintoamagnetopenedhexadecanemarble.
Figure1.
a)StructureofFD-POSS.
b)ProcesstomaketheFD-POSS/Fe3O4nanoparticlecomposite.
Theinsertimagetotherightshowsthesuspen-sionsofDP-POSSandtheFD-POSS/Fe3O4nanoparticlecompositepowdersinsolventandtheFD-POSS/Fe3O4underamagneticeld.
c)OpticalmicroscopyimagesofFD-POSSandFD-POSS/Fe3O4powders(FD-POSS:Fe3O4=1:1,w/w).
d)Water,dimethylsulfoxide(DMSO),toluene,hexade-cane,andethanoldropletsontheFD-POSS/Fe3O4powder-coatedsiliconsurface.
4816www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818thatpushestheliquidoutofthepowderlayer.
Whetheraliquidmarblecanremainstableonasolidsurfaceornotisdeterminedbytheabilityofthenon-wettingpowderlayertopreventtheliquidbreakthrough(i.
e.
,breakthroughpressure,Pbreakthrough)andtheactualliquidpressuregeneratedbytheliquiddroplet(Pdroplet).
IfPbreakthroughPdroplet,theactualpressuregeneratedbytheliquiddropletisnotenoughtoforcethroughthepowderlayerhencetheliquidmarbleisstable.
WhenPdropletisclosetoPbreakthrough,itiseasyfortheliquidtobreakthepowderlayer,whichleadstobreakageoftheliquidmarble.
Basedonthecapillaryforceandtheliquidpressurethatforcesliquidtosagintothepowderypores(Figure3b,c),theratiobetweenthebreakthroughpressureandthemaximumliquidpressureformedbythedroplet(Pmaxdroplet=Dgl,whereρisthedensityoftheliquid,gistheconstantduetogravity,andl=γlv/ρgiscapillarylengthoftheliquid),PbreakthroughPmaxdroplet/,canbeestimatedas:PbreakthroughPmaxdroplet=A=H.
TH+T/(1)byamagneticeld,thereactionliquidcanberemovedeasilybyacapillarydeviceforfurtheranalysesordirectlytestedbyanopticalspectrometer(e.
g.
,UVanduorescentspectrom-eters).
Figure2galsoshowsthedirectpuricationofareac-tionproductthroughanarrowchromatographicaluminasheetpluggedintotheopenedliquidmarble.
Theabilityofahydrophobicpowdertorepelliquiduidismainlydeterminedbythesurfacechemistryandphysicaltopologyofthepowdermaterial,thesurfacetensionofaliquid,andthecontactmodebetweentheliquidandpowder.
TheFD-POSSmoleculecompriseseightuorinateddecylgroupswhichinclude136uorineatomssurroundingthePOSScage,makingthemoleculeextremelylowinsurfacefreeenergy.
Theroughsurfaceattributabletotheparticulatestructurefacilitatestheformationofanon-wettingpowderysurface.
[24–26]Inthestaticstate,theencapsulatingpowdersbetweenthedropletandthesolidsupport(Figure3a)receivethelargestliquidpressure.
Becauseofthenon-wettingfeature,theliquidonapowderyporouslayerreceivesanegativecapillarypressureFigure2.
a)DigitalgraphicimagesofliquidmarblesproducedfromdifferentliquidsandFD-POSSpowder(dropletsize3μL).
Foreasyobservation,asmallamountofdyewasaddedtotheliquidandtheexistenceofthedyehasnoinuenceonthestabilityoftheliquidmarbles.
b)Liquidmarblesoatingonwaterandhexadecanesurfaces(dropletsize3μL).
c)Magnetopenedliquidmarbles(dropletsize7μL).
d)Magnet-drivenmotionsofaFD-POSS-stainedFD-POSS/Fe3O4hexadecanemarbleonaglassslide(top)andwatersurface(bottom)(dropletsize7μL).
e)Magnetopenedhexa-decanemarbleswithdifferentnumbersofcoloredwaterdropletsadded(overalldropletsize10μL).
f)Achemiluminescencereactionthatoccursasaresultofcoalescingoftwomagneticliquidmarblesthatcontaindifferentreagents(top),andthesamechemiluminescencereactionhappeningwithinasingleliquidmarble(bottom)(dropletsize10μL).
g)Chromatographicanalysisofliquidintheopenedliquidmarble(dropletsize10μL).
Scalebar:1mm.
4817www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818liquids.
Magneticmanipulationenablestheliquidinthemarbletocommunicatewiththeoutsideenvironmentondemand,whichprovidesasimplewaytointegratewithportablepurica-tionandanalysisfacilities.
Allthesefeaturescouldleadtonewuniversal"lab-on-chip"reactorsystemswithexcellentintegrationabilitytoincorporatesamplefabrication,actuation,purication,andanalysisintoasingleelectronicchipfordiverseapplications.
ExperimentalSectionPreparationofFD-POSS/Fe3O4NanoparticleCompositePowders:(1H,1H,2H,2H-heptadecauorodecyl)8Si8O12(FD-POSS),synthesizedaccordingtotheliteraturemethod,[20]andhydrophobicFe3O4nanoparticles,preparedbyaliteraturemethod,[16]weredispersedin20mLofethanolandcontinuouslystirredfor24h.
Themixturewasthenisolatedfromthesolutionwithabarmagnetanddriedundervacuumat60°C.
PreparationandMagneticManipulationofLiquidMarbles:LiquidmarbleswerepreparedbydroppingliquiddropletsontoabedofFD-POSSorFD-POSS/Fe3O4nanoparticlegrains.
Upongentleshakingofthedroplet,thesolidpowdersbecamestucktothedropletsurfaceformingapowder-encapsulatedliquiddroplet(i.
e.
,liquidmarble).
Magneticmanipulationoftheliquidmarbleswasperformedusinganeodymiumcylindermagnet(diameter10mmandlength12mm).
Thedirectedmovementoftheliquidmarbleswasrecordedusingadigitalmicroscope(Dino-LiteAM313).
SupportingInformationSupportingInformationisavailablefromtheWileyOnlineLibraryorfromtheauthor.
H=2(1cosθ)lcapD(2√3D2ππ)(2)T=Blcapsin(2Rmin)R(2√3DB)(3)HandTaretherobustnessheightandangledeterminedbytheliquid–gassurfacetension(γlv),theparticleradius(R),theinterparticledistance(2D),andtheapparentcontactangleofliquidonthepowdermaterial(θ)(fordetailsseetheSupportingInformation).
ThedependencyofPbreakthroughPmaxdroplet/onthepar-ticleradiusandthehalfinter-particledistanceforthepowderarrangementisgiveninFigure3d(thecalculationontheotherparticlearrangementisgivenintheSupportingInformation).
ItisinterestingtonotethatamaximumPbreakthroughPmaxdroplet/ratiooccurswhenthepowderradiusisabout35μmandtheinter-powderdistanceissmall.
Basedontheactualpowderdimen-sionandsurfaceproperties,thePbreakthroughPmaxdroplet/Dforthreestudiedliquidsystemswascomputed(Figure3e).
Whenthisinter-powderdistanceislessthan160μm(D<80μm),thePbreakthroughPmaxdroplet/islargerthanunityeveniftheliquidsurfacetensionisverylow,suggestingtheformationofaverystableliquidmarble.
Insummary,wehavedemonstratedthepotentialofusinguorinatedPOSSandmagneticnanoparticlesasauniversalencapsulatingagenttoprepareliquidmarblesfromvariousFigure3.
a)Idealizedliquidmarblewithasinglelayerofpowderscoveringthedropletsurface.
b)Fluidsaggingintotwoparticlesandthedimen-sionsforcalculationofthebreakthroughpressure.
c)Onepowderarrangementusedforcalculationofbreakthroughpressure.
d)DependenciesofPbreakthroughPmax/dropletratioonRandDforoctane(θ=55°).
e)PbreakthroughPmax/dropletDdependencycalculatedbasedontheactualpowderdimensionforwater(θ=120°),hexadecane(θ=80°),andoctane.
4818www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818AcknowledgementsTheauthorsacknowledgethefundingsupportfromDeakinUniversityundertheAlfredDeakinPostdoctoralFellowshipschemeandStateKeyLaboratoryofChemicalEngineering(SKL-ChE-09D05).
Received:May22,2010Publishedonline:September5,2010[1]K.
Jensen,Nature1998,393,735.
[2]A.
J.
deMello,Nature2006,442,394.
[3]S.
-Y.
Teh,R.
Lin,L.
-H.
Hung,A.
P.
Lee,LabChip2008,8,198.
[4]M.
Abdelgawad,A.
R.
Wheeler,Adv.
Mater.
2009,21,920.
[5]P.
S.
Dittrich,A.
Manz,Nat.
Rev.
DrugDiscov.
2006,5,210.
[6]R.
H.
Liu,J.
Yang,R.
Lenigk,J.
Bonanno,P.
Grodzinski,Anal.
Chem.
2004,79,1824.
[7]M.
A.
Burns,B.
N.
Johnson,S.
N.
Brahmasandra,K.
Handique,J.
R.
Webster,M.
Krishnan,T.
S.
Sammarco,P.
M.
Man,D.
Jones,D.
Heldsinger,C.
H.
Mastrangelo,D.
T.
Burke,Science1998,282,484.
[8]B.
Zheng,L.
S.
Roach,R.
F.
Ismagilov,J.
Am.
Chem.
Soc.
2003,125,11170.
[9]O.
D.
Velev,B.
G.
Prevo,K.
H.
Bhatt,Nature2003,426,515.
[10]M.
Joanicot,A.
Ajdari,Science2005,309,887.
[11]H.
Song,D.
L.
Chen,R.
F.
Ismagilov,Angew.
Chem.
Int.
Ed.
2006,45,7336.
[12]A.
Günther,M.
Jhunjhunwala,M.
Thalmann,M.
A.
Schmidt,K.
F.
Jensen,Langmuir2005,21,1547.
[13]B.
Alberts,A.
Johnson,J.
Lewis,M.
Raff,K.
Roberts,P.
Walter,MolecularBiologyoftheCell,GarlandScience,NewYork2002.
[14]P.
Aussillous,D.
Quere,Nature2001,411,924.
[15]P.
Aussillous,D.
Quere,Proc.
R.
Soc.
A2006,462,973.
[16]Y.
Zhao,J.
Fang,H.
Wang,X.
Wang,T.
Lin,Adv.
Mater.
2010,22,707.
[17]A.
V.
Rao,M.
M.
Kulkarni,S.
D.
Bhagat,J.
ColloidInterfaceSci.
2005,285,413.
[18]G.
McHale,D.
L.
Herbertson,S.
J.
Elliott,N.
J.
Shirtcliffe,M.
I.
Newton,Langmuir2007,23,918.
[19]L.
Gao,T.
J.
Mccarthy,Langmuir2007,23,10445.
[20]J.
M.
Mabry,A.
Vij,S.
T.
Iacono,B.
D.
Viers,Angew.
Chem.
Int.
Ed.
2008,47,4137.
[21]A.
Tuteja,w.
Choi,M.
Ma,J.
M.
Mabry,S.
A.
Mazzella,G.
C.
Rutledge,G.
H.
Mckinley,R.
E.
Cohen,Science2007,318,1618.
[22]R.
B.
Thompson,S.
E.
S.
McBee,Langmuir1988,4,106.
[23]R.
E.
Milofsky,J.
W.
Birks,J.
Am.
Chem.
Soc.
1991,113,9715.
[24]X.
Feng,L.
Jiang,Adv.
Mater.
2006,18,3063.
[25]P.
Roach,N.
J.
Shirtcliffe,M.
I.
Newton,SoftMatter2008,4,224.
[26]X.
Zhang,F.
Shi,J.
Niu,Y.
Jiang,Z.
Wang,J.
Chem.
Mater.
2008,18,621.
KGaA,Weinheim4814www.
advmat.
dewww.
MaterialsViews.
comCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818ByYuhuaXue,HongxiaWang,YanZhao,LimingDai,LianfangFeng,XungaiWang,andTongLin*MagneticLiquidMarbles:A"Precise"MiniatureReactor[*]Dr.
Y.
Xue,Dr.
H.
Wang,Dr.
Y.
Zhao,Prof.
X.
Wang,Prof.
T.
LinCentreforMaterialandFibreInnovationDeakinUniversityGeelong,VIC3217(Australia)E-mail:tong.
lin@deakin.
edu.
auProf.
L.
DaiDepartmentofChemicalEngineeringCaseWesternReserveUniversityCleveland,Ohio44106(USA)Prof.
L.
FengStateKeyLaboratoryofChemicalEngineeringDepartmentofChemicalandBiologicalEngineeringZhejiangUniversityHangzhou310027(P.
R.
China)DOI:10.
1002/adma.
201001898Miniaturizedchemicalprocesseshavemanyadvantages,suchasreduceduseofchemicalreagentsandsolvents,preciselycontrolledreactionconditions,muchshortenedreactiontime,andtheabilitytointegrateintoadigitaldevice.
[1–4]Theyareveryusefulforhigh-throughputanalysesandpuricationsinchemicalandbiologicalprocesses,suchasdrugdiscovery,[5]DNAanalysis,[6,7]proteincrystallization,[8]andthesynthesisofmoleculesorparticles.
[9–11]Indeed,considerableeffortshavebeenmadetominiaturizechemicalprocessesusingvariousprinciples.
Forexample,oil/wateremulsionshavebeenusedtoperformchemicalreactionsinparallelinalargenumberofemulsiedtinydroplets.
[10]Microuidic"lab-on-a-chip"deviceshavebeendesignedtomanipulatechemicalprocesseswithinmicro-channelseitherincontinuousuidordisconnecteduidsegmentsseparatedbyanimmiscibleuidorgas.
[10,12]Althoughthechannel-basedmicro-reactorsshowgreatpoten-tialinhigh-throughputreactionandintegratabilitytoexternalanalysisfacilities,thereaction-specicdevicepreparations,thechannel-associatedcross-contaminations,andtheneedforexternalpumpstoactuatetheuidmotionmakethemneitheruniversalnorminiature.
Also,microuidicsishardtousetohandleasingledroplet.
Recently,freeaqueousdropletsinanimmiscibleuid(channel-freemicrouidics)[9]hasalsobeenproposedasanalternativestrategy,butcross-contaminationstillremainsaconcern.
Inlivingsystems,cellsarewell-knownasthebasicstructuralandfunctionalunits[13]thatmediatevariousbiochemicalreac-tions(e.
g.
,catabolismandanabolism)forsustainingthegrowthanddivisionofcellsandhencelivingsystems.
Althoughtheirstructurelooksverysimple,cellscommunicatewitheachotherinahighlyintelligentway,throughoutthewholelivingsystem.
Inthisregard,howtomimiclivingcells,bothinstructureandfunction,istheultimatechallengetowardthedevelopmentofarticialminiaturereactors.
Liquidmarbles,liquiddropsencapsulatedwithsolidpowderattheliquid–gasinterface,haverecentlybeenreportedasanewapproachtomanipulatingliquid,[14]whichcouldbeusedasminiaturereactors.
Theyarenon-wettingtoanysolidsur-facesandcouldbemanipulatedbyexternalforces,suchasgravity,[14]electrical,[15]andmagnetic,[16]dependingontheircomponents.
[14–19]However,mostliquidmarblesreportedsofarhavebeenbasedonaqueousliquid.
Althoughafewpapers[19]havereportedtheencapsulationoforganicliquidsintoliquidmarbles,theliquidswereconnedtothosehavingahighsur-facetension.
[19]Theformationofstableliquidmarblesfromawiderangeoforganicsolvents,especiallythosethathavealowsurfacetension,isimportantforchemicalreactions,butstillremainsachallenge.
Inthisstudy,wedemonstratetheincredibleabilityofauor-inateddecylpolyhedraloligomericsilsesquioxane(FD-POSS,structureshowninFigure1a)anditscombinationwithhydro-phobicmagneticnanoparticlestoactasencapsulatingagentstopreparestableliquidmarblesfrombothaqueoussolutionandorganicliquidswithasurfacetensionaslowas20.
1dynecm1,andthefeasibilityofusingthemagneticliquidmarblesasuni-versal"smart"miniaturereactors.
FD-POSSwassynthesizedbythereportedmethod[20,21]andaneFD-POSSpowderwaspreparedbyagrindingtechnique.
TheFD-POSS/Fe3O4nanoparticlecompositepowdersofdif-ferentratios(1:10to1:1,w/w)werepreparedbyco-dispersingtheneFD-POSSpowderwithhydrophobicFe3O4nanopar-ticlesinethanol.
AsshowninthephotographsinFigure1b,FD-POSSpowdersarewhiteandhaveacertaintransparencytovisiblelightbutarenotresponsivetoamagneticeld.
Inethanol,FD-POSSandFe3O4nanoparticleshadastrongafnity,andtheytendedtoaggregateintopowderswhichshowedasuperparamagneticproperty(SupportingInfor-mation).
TheopticalmicrographsinFigure1cindicatethatFD-POSSandFD-POSS/Fe3O4compositepowdersarelessthan70μm,andtheFD-POSSandFe3O4nanoparticlesformedarandomlyblendedcompositestructure.
Whenathinbedofthepowderswerecoatedonasolidsurface,thesurfacebecamehighlyrepellenttoliquids.
Water,dimethylsulfoxide(DMSO),toluene,hexadecane,andethanolallformedrounddropletsonthepowdercoatedsurfacewithoverallcontactanglesof171.
1°,166.
2°,155.
7°,154.
4°,and142.
8°,respectively(Figure1d).
Thepowderlayershowedstrongrepellencetootherorganicsolvents(SupportingInformation).
ThecompositepowdersinthestudiedFD-POSS/Fe3O4ratiorangeshowedaverysimilaruidrepellencytothepureFD-POSSgrains.
Liquidmarbleswerepreparedsimplybydroppingasmallvolumeofliquidontothepowdersurfaceandsubsequentlyshakingthedropletgentlysothatthepowderscoveredtheentiredropletsurface.
Theas-preparedpowders,bothFD-POSS4815www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818Severalwaterdropletswereaddedtotheopenedmarble(10μL)toformawater-in-oilbicomponentliquidmarble.
Oil-in-waterbicomponentliquidmarblescanalsobeproducedinasimilarway.
Thesebicomponentliquidmarblesshowtheabilitytopre-ventthecoreliquidfromrapidevaporation.
Todemonstratetheabilitytomediatechemicalreactions,weusedtwodifferentwaystoundertakeachemiluminescencereaction.
[22,23]Onemethodusedtwomagneticliquidmarblestocarrydifferentreactiveagents,oneforhydrogenperoxideandtheotherforbis(2,4,6-trichlorophenyl)oxalateanddye.
AsshowninFigure2f(toppanel),thetwoliquidmarbleswereactuatedunderamagneticeldtomovetogetherandtheythencoalescedintoalargerliquidmarble,initiatingthechemilumi-nescencereactionwhichcanbeprovenbyanimmediatelightemissionfromthecoalescedliquidmarble.
Anothermethodinvolvedusingabicomponentliquidmarbletohosttwocoredroplets,onefortheoxidantandtheotherfortheuorescer,inanimmiscibleuid.
Undergentlemovementactuatedbyamagnetbar,thetwocoredropletsjointedtogethertriggeringthechemiluminescencereaction.
TheimageinFigure2f(bottompanel)clearlyindicatesthelightemittingjustfromthecombinedcoredroplet.
Besidesthechemiluminescencereaction,otherchemicalreactionssuchasphotochemicalpolymerization,nanoparticlesynthesis,andanacid–basereactionhavealsobeentrialedandproventobesuccessful(SupportingInformation).
Oneimportantaspectforminiaturechemicalreactorsistheabilitytointegratewithpuricationandanalysisfacilities.
SincethemagneticliquidmarblescanbeopenedandclosedreversiblyandFD-POSS/Fe3O4composite,canencapsulatenotonlywaterbutalsoorganicsolventstoformstableliquidmarbles.
Figure2ashowsphotosofliquidmarblespreparedfromwater,DMSO,toluene,ethanol,andoctane(3μL).
Manyotherorganicsol-ventshavealsobeentestedsofarandthepowderswereabletoencapsulatealiquidofsurfacetensionaslowas20.
1dynescm1(SupportingInformation).
Theliquidmarblesareverystableandtheycanoatnotonlyonwaterbutalsoonauidoflowsurfacetension(Figure2b).
ThesuperparamagneticpropertyoftheFe3O4nanoparticlesimpartstheFD-POSS/Fe3O4powder-encapsulatedliquidmar-bleswithamagnetresponsiveability.
Asaresult,theliquidmarblescanbeactuatedtomoveindifferentdirections,oropenedandclosedreversibly,underamagneticeld.
Figure2cshowsmagnetopenedliquidmarblesfromdifferentliquids.
Theopenedliquidmarbleallowsittobecoveredwithanothertypeofhydrophobicmaterial.
Todemonstratethis,weusedpureFD-POSSpowdertocovertheopenedliquidsurfaceofamagneticliquidmarble,andthecoveredpartshowedadifferentcolortothemagneticpowdercoveredsurface.
Consequently,themotionofliquidcanbetracedeasily.
Figure2dillustratesthemotionofawhiteFD-POSS-stainedFD-POSS/Fe3O4hexa-decanemarbleunderamagneticeld.
Itcanbeclearlyseenthatthemarblerotatesonasolidsurface(toppanel),butshiftssidewaysonaliquidsurface(bottompanel).
Themagnetopenedliquidmarblealsoallowstheliquidtobetakenfromthemarbleorthemarbletobereplenishedwithotherliquids.
Figure2edemonstratestheadditionofcolorfulwaterdropletsintoamagnetopenedhexadecanemarble.
Figure1.
a)StructureofFD-POSS.
b)ProcesstomaketheFD-POSS/Fe3O4nanoparticlecomposite.
Theinsertimagetotherightshowsthesuspen-sionsofDP-POSSandtheFD-POSS/Fe3O4nanoparticlecompositepowdersinsolventandtheFD-POSS/Fe3O4underamagneticeld.
c)OpticalmicroscopyimagesofFD-POSSandFD-POSS/Fe3O4powders(FD-POSS:Fe3O4=1:1,w/w).
d)Water,dimethylsulfoxide(DMSO),toluene,hexade-cane,andethanoldropletsontheFD-POSS/Fe3O4powder-coatedsiliconsurface.
4816www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818thatpushestheliquidoutofthepowderlayer.
Whetheraliquidmarblecanremainstableonasolidsurfaceornotisdeterminedbytheabilityofthenon-wettingpowderlayertopreventtheliquidbreakthrough(i.
e.
,breakthroughpressure,Pbreakthrough)andtheactualliquidpressuregeneratedbytheliquiddroplet(Pdroplet).
IfPbreakthroughPdroplet,theactualpressuregeneratedbytheliquiddropletisnotenoughtoforcethroughthepowderlayerhencetheliquidmarbleisstable.
WhenPdropletisclosetoPbreakthrough,itiseasyfortheliquidtobreakthepowderlayer,whichleadstobreakageoftheliquidmarble.
Basedonthecapillaryforceandtheliquidpressurethatforcesliquidtosagintothepowderypores(Figure3b,c),theratiobetweenthebreakthroughpressureandthemaximumliquidpressureformedbythedroplet(Pmaxdroplet=Dgl,whereρisthedensityoftheliquid,gistheconstantduetogravity,andl=γlv/ρgiscapillarylengthoftheliquid),PbreakthroughPmaxdroplet/,canbeestimatedas:PbreakthroughPmaxdroplet=A=H.
TH+T/(1)byamagneticeld,thereactionliquidcanberemovedeasilybyacapillarydeviceforfurtheranalysesordirectlytestedbyanopticalspectrometer(e.
g.
,UVanduorescentspectrom-eters).
Figure2galsoshowsthedirectpuricationofareac-tionproductthroughanarrowchromatographicaluminasheetpluggedintotheopenedliquidmarble.
Theabilityofahydrophobicpowdertorepelliquiduidismainlydeterminedbythesurfacechemistryandphysicaltopologyofthepowdermaterial,thesurfacetensionofaliquid,andthecontactmodebetweentheliquidandpowder.
TheFD-POSSmoleculecompriseseightuorinateddecylgroupswhichinclude136uorineatomssurroundingthePOSScage,makingthemoleculeextremelylowinsurfacefreeenergy.
Theroughsurfaceattributabletotheparticulatestructurefacilitatestheformationofanon-wettingpowderysurface.
[24–26]Inthestaticstate,theencapsulatingpowdersbetweenthedropletandthesolidsupport(Figure3a)receivethelargestliquidpressure.
Becauseofthenon-wettingfeature,theliquidonapowderyporouslayerreceivesanegativecapillarypressureFigure2.
a)DigitalgraphicimagesofliquidmarblesproducedfromdifferentliquidsandFD-POSSpowder(dropletsize3μL).
Foreasyobservation,asmallamountofdyewasaddedtotheliquidandtheexistenceofthedyehasnoinuenceonthestabilityoftheliquidmarbles.
b)Liquidmarblesoatingonwaterandhexadecanesurfaces(dropletsize3μL).
c)Magnetopenedliquidmarbles(dropletsize7μL).
d)Magnet-drivenmotionsofaFD-POSS-stainedFD-POSS/Fe3O4hexadecanemarbleonaglassslide(top)andwatersurface(bottom)(dropletsize7μL).
e)Magnetopenedhexa-decanemarbleswithdifferentnumbersofcoloredwaterdropletsadded(overalldropletsize10μL).
f)Achemiluminescencereactionthatoccursasaresultofcoalescingoftwomagneticliquidmarblesthatcontaindifferentreagents(top),andthesamechemiluminescencereactionhappeningwithinasingleliquidmarble(bottom)(dropletsize10μL).
g)Chromatographicanalysisofliquidintheopenedliquidmarble(dropletsize10μL).
Scalebar:1mm.
4817www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818liquids.
Magneticmanipulationenablestheliquidinthemarbletocommunicatewiththeoutsideenvironmentondemand,whichprovidesasimplewaytointegratewithportablepurica-tionandanalysisfacilities.
Allthesefeaturescouldleadtonewuniversal"lab-on-chip"reactorsystemswithexcellentintegrationabilitytoincorporatesamplefabrication,actuation,purication,andanalysisintoasingleelectronicchipfordiverseapplications.
ExperimentalSectionPreparationofFD-POSS/Fe3O4NanoparticleCompositePowders:(1H,1H,2H,2H-heptadecauorodecyl)8Si8O12(FD-POSS),synthesizedaccordingtotheliteraturemethod,[20]andhydrophobicFe3O4nanoparticles,preparedbyaliteraturemethod,[16]weredispersedin20mLofethanolandcontinuouslystirredfor24h.
Themixturewasthenisolatedfromthesolutionwithabarmagnetanddriedundervacuumat60°C.
PreparationandMagneticManipulationofLiquidMarbles:LiquidmarbleswerepreparedbydroppingliquiddropletsontoabedofFD-POSSorFD-POSS/Fe3O4nanoparticlegrains.
Upongentleshakingofthedroplet,thesolidpowdersbecamestucktothedropletsurfaceformingapowder-encapsulatedliquiddroplet(i.
e.
,liquidmarble).
Magneticmanipulationoftheliquidmarbleswasperformedusinganeodymiumcylindermagnet(diameter10mmandlength12mm).
Thedirectedmovementoftheliquidmarbleswasrecordedusingadigitalmicroscope(Dino-LiteAM313).
SupportingInformationSupportingInformationisavailablefromtheWileyOnlineLibraryorfromtheauthor.
H=2(1cosθ)lcapD(2√3D2ππ)(2)T=Blcapsin(2Rmin)R(2√3DB)(3)HandTaretherobustnessheightandangledeterminedbytheliquid–gassurfacetension(γlv),theparticleradius(R),theinterparticledistance(2D),andtheapparentcontactangleofliquidonthepowdermaterial(θ)(fordetailsseetheSupportingInformation).
ThedependencyofPbreakthroughPmaxdroplet/onthepar-ticleradiusandthehalfinter-particledistanceforthepowderarrangementisgiveninFigure3d(thecalculationontheotherparticlearrangementisgivenintheSupportingInformation).
ItisinterestingtonotethatamaximumPbreakthroughPmaxdroplet/ratiooccurswhenthepowderradiusisabout35μmandtheinter-powderdistanceissmall.
Basedontheactualpowderdimen-sionandsurfaceproperties,thePbreakthroughPmaxdroplet/Dforthreestudiedliquidsystemswascomputed(Figure3e).
Whenthisinter-powderdistanceislessthan160μm(D<80μm),thePbreakthroughPmaxdroplet/islargerthanunityeveniftheliquidsurfacetensionisverylow,suggestingtheformationofaverystableliquidmarble.
Insummary,wehavedemonstratedthepotentialofusinguorinatedPOSSandmagneticnanoparticlesasauniversalencapsulatingagenttoprepareliquidmarblesfromvariousFigure3.
a)Idealizedliquidmarblewithasinglelayerofpowderscoveringthedropletsurface.
b)Fluidsaggingintotwoparticlesandthedimen-sionsforcalculationofthebreakthroughpressure.
c)Onepowderarrangementusedforcalculationofbreakthroughpressure.
d)DependenciesofPbreakthroughPmax/dropletratioonRandDforoctane(θ=55°).
e)PbreakthroughPmax/dropletDdependencycalculatedbasedontheactualpowderdimensionforwater(θ=120°),hexadecane(θ=80°),andoctane.
4818www.
advmat.
dewww.
MaterialsViews.
com2010WILEY-VCHVerlagGmbH&Co.
KGaA,WeinheimCOMMUNICATIONwileyonlinelibrary.
comAdv.
Mater.
2010,22,4814–4818AcknowledgementsTheauthorsacknowledgethefundingsupportfromDeakinUniversityundertheAlfredDeakinPostdoctoralFellowshipschemeandStateKeyLaboratoryofChemicalEngineering(SKL-ChE-09D05).
Received:May22,2010Publishedonline:September5,2010[1]K.
Jensen,Nature1998,393,735.
[2]A.
J.
deMello,Nature2006,442,394.
[3]S.
-Y.
Teh,R.
Lin,L.
-H.
Hung,A.
P.
Lee,LabChip2008,8,198.
[4]M.
Abdelgawad,A.
R.
Wheeler,Adv.
Mater.
2009,21,920.
[5]P.
S.
Dittrich,A.
Manz,Nat.
Rev.
DrugDiscov.
2006,5,210.
[6]R.
H.
Liu,J.
Yang,R.
Lenigk,J.
Bonanno,P.
Grodzinski,Anal.
Chem.
2004,79,1824.
[7]M.
A.
Burns,B.
N.
Johnson,S.
N.
Brahmasandra,K.
Handique,J.
R.
Webster,M.
Krishnan,T.
S.
Sammarco,P.
M.
Man,D.
Jones,D.
Heldsinger,C.
H.
Mastrangelo,D.
T.
Burke,Science1998,282,484.
[8]B.
Zheng,L.
S.
Roach,R.
F.
Ismagilov,J.
Am.
Chem.
Soc.
2003,125,11170.
[9]O.
D.
Velev,B.
G.
Prevo,K.
H.
Bhatt,Nature2003,426,515.
[10]M.
Joanicot,A.
Ajdari,Science2005,309,887.
[11]H.
Song,D.
L.
Chen,R.
F.
Ismagilov,Angew.
Chem.
Int.
Ed.
2006,45,7336.
[12]A.
Günther,M.
Jhunjhunwala,M.
Thalmann,M.
A.
Schmidt,K.
F.
Jensen,Langmuir2005,21,1547.
[13]B.
Alberts,A.
Johnson,J.
Lewis,M.
Raff,K.
Roberts,P.
Walter,MolecularBiologyoftheCell,GarlandScience,NewYork2002.
[14]P.
Aussillous,D.
Quere,Nature2001,411,924.
[15]P.
Aussillous,D.
Quere,Proc.
R.
Soc.
A2006,462,973.
[16]Y.
Zhao,J.
Fang,H.
Wang,X.
Wang,T.
Lin,Adv.
Mater.
2010,22,707.
[17]A.
V.
Rao,M.
M.
Kulkarni,S.
D.
Bhagat,J.
ColloidInterfaceSci.
2005,285,413.
[18]G.
McHale,D.
L.
Herbertson,S.
J.
Elliott,N.
J.
Shirtcliffe,M.
I.
Newton,Langmuir2007,23,918.
[19]L.
Gao,T.
J.
Mccarthy,Langmuir2007,23,10445.
[20]J.
M.
Mabry,A.
Vij,S.
T.
Iacono,B.
D.
Viers,Angew.
Chem.
Int.
Ed.
2008,47,4137.
[21]A.
Tuteja,w.
Choi,M.
Ma,J.
M.
Mabry,S.
A.
Mazzella,G.
C.
Rutledge,G.
H.
Mckinley,R.
E.
Cohen,Science2007,318,1618.
[22]R.
B.
Thompson,S.
E.
S.
McBee,Langmuir1988,4,106.
[23]R.
E.
Milofsky,J.
W.
Birks,J.
Am.
Chem.
Soc.
1991,113,9715.
[24]X.
Feng,L.
Jiang,Adv.
Mater.
2006,18,3063.
[25]P.
Roach,N.
J.
Shirtcliffe,M.
I.
Newton,SoftMatter2008,4,224.
[26]X.
Zhang,F.
Shi,J.
Niu,Y.
Jiang,Z.
Wang,J.
Chem.
Mater.
2008,18,621.
- preparedwww.55fang.com相关文档
- 深圳市www.55fang.com
- boardwww.55fang.com
- analyzedwww.55fang.com
- fullywww.55fang.com
- 随着知识资本流经消息发送系统以及员工的移动性越来越高,企业面临
- leachingwww.55fang.com
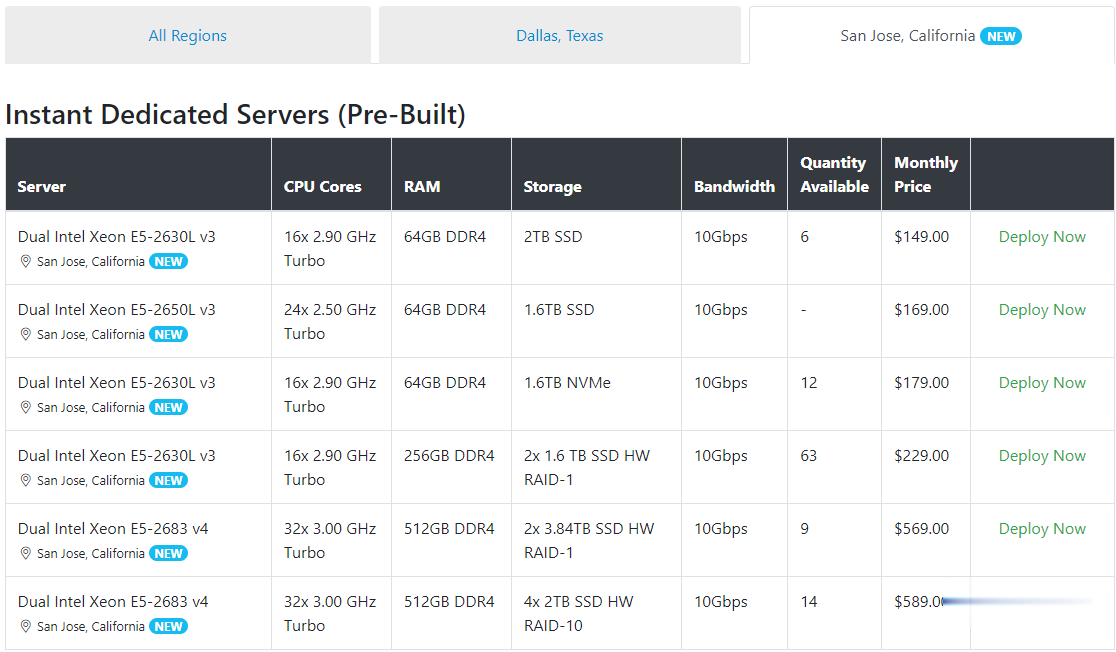
Spinservers美国圣何塞服务器$111/月流量10TB
Spinservers是Majestic Hosting Solutions,LLC旗下站点,主营美国独立服务器租用和Hybrid Dedicated等,数据中心位于美国德克萨斯州达拉斯和加利福尼亚圣何塞机房。TheServerStore.com,自 1994 年以来,它是一家成熟的企业 IT 设备供应商,专门从事二手服务器和工作站业务,在德克萨斯州拥有 40,000 平方英尺的仓库,库存中始终有...
Sharktech云服务器35折年付33美元起,2G内存/40G硬盘/4TB流量/多机房可选
Sharktech又称SK或者鲨鱼机房,是一家主打高防产品的国外商家,成立于2003年,提供的产品包括独立服务器租用、VPS云服务器等,自营机房在美国洛杉矶、丹佛、芝加哥和荷兰阿姆斯特丹等。之前我们经常分享商家提供的独立服务器产品,近期主机商针对云虚拟服务器(CVS)提供优惠码,优惠后XS套餐年付最低仅33.39美元起,支持使用支付宝、PayPal、信用卡等付款方式。下面以XS套餐为例,分享产品配...

SunthyCloud阿里云国际版分销商注册教程,即可PayPal信用卡分销商服务器
阿里云国际版注册认证教程-免绑卡-免实名买服务器安全、便宜、可靠、良心,支持人民币充值,提供代理折扣简介SunthyCloud成立于2015年,是阿里云国际版正规战略级渠道商,也是阿里云国际版最大的分销商,专业为全球企业客户提供阿里云国际版开户注册、认证、充值等服务,通过SunthyCloud开通阿里云国际版只需要一个邮箱,不需要PayPal信用卡就可以帮你开通、充值、新购、续费阿里云国际版,服务...

www.55fang.com为你推荐
-
公司网络被攻击网站总是被攻击,该怎么处理啊?蓝色骨头手机宠物的骨头分别代表几级?商标注册流程及费用商标注册流程及费用?百度关键词价格查询百度关键字如何设定竟价价格?lunwenjiancepaperfree论文检测怎样算合格www.cn12365.orgwww.12365china.net是不是真的防伪网站300373一搓黑是真的吗888300.com请问GXG客服电话号码是多少?汴京清谈求好看的鼠猫文~网页源代码网页的HTML代码百度关键字百度推广中关键词匹配方式分为哪几种?