reviewedregister.com
register.com 时间:2021-01-11 阅读:()
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs1PublicDisclosureofClinicalResearchTheIssueThepharmaceuticalindustrycommunicatestheresultsofitsclinicalresearchbypublishinginscientificjournals;bypresentingresultsatscientificcongresses;and,inlinewithestablishedindustrycommitmentsandevolvinglegalrequirements,bypostinginformationandresultsoninternet-basedpublicregisters.
Thisdisclosureisinadditiontosubmissionofinformationtonationalorregionalregulatoryauthoritiesaspartoftheproductdevelopmentorapprovalprocess.
Despitethis,however,concernshavebeenraisedbysomestakeholdersabout:–whetherstudyresultswhichmaybeviewedas"negative"forcompanies'medicinesarepublishedinscientificliterature;–whetherpublishedstudiesaccuratelyreflecttheconductofthestudy;–whethersomejournalarticlesare"ghost-written"(wheredoctorsputtheirnametoarticleswrittenbypharmaceuticalcompanies);–thelackofaccesstotheunderlyingpatientleveldatathatiscollectedduringthestudy.
ThispaperoutlinesGSK'sapproachtoaddressingtheseissues.
Itisanapproachthatdatesbackto2004whenwebecamethefirstcompanytolaunchaninternet-basedclinicalstudyregisterhttp://www.
gsk-studyregister.
com/providingresultsummariesfromourclinicaltrialsofmarketedmedicines(i.
epharmaceuticalsandvaccines)irrespectiveofthestudyoutcome.
Ithasevolvedsignificantlyaswehavesoughtnewwaysofenhancingthetransparencyofourclinicalresearch,includingthatconductedbyourconsumerhealthcaredivision.
GSK'sdisclosurepolicygoesbeyondwhatisrequiredbylawsandregulations.
GSK'sPosition–Beforethefirstsubjectisenrolledinastudy,wepostprotocolsummariesofGSK-sponsoredinterventionalandnon-interventionalstudiesoninternetregisters.
ThisincludesphaseI-IVclinicaltrialsofbothinvestigationalandmarketedmedicinesandconsumerhealthcareproducts.
Thiscommitmentalsoincludespooledanalyses/meta-analysesthatevaluateourproducts.
–WepostourinterventionalstudiesonClinicalTrials.
gov(http://www.
clinicaltrials.
gov).
Wepostournon-interventional,observationalstudiesonourGSKStudyRegister(http://www.
gsk-studyregister.
com).
–Irrespectiveoftheoutcomeofthestudy,wepostresultsummarieswithin12monthsofprimarycompletiondateforinterventionalstudiesand12monthsfromthecompletionofanalysisfornon-interventionalstudies.
Wedonotwaituntilapprovalorterminationofthemedicinebeforepostingresultsummaries.
Atthetimeofresults'registration,wealsopostthefullprotocolandthestatisticalanalysisplan.
–Ourcommitmentincludespostingresultsfromstudiesofterminatedcompoundsinordertohelpinformthescientificcommunityaboutnon-productiveareasofresearchandtoreduceunnecessaryexposureofstudyparticipantstosimilarcompoundsinotherclinicaltrials.
–GSK'scommitmenttopostprotocolsummaries,analysisplansandresultsummariesforallPhaseIstudies(inadditiontoPhasesII-IV)toClinicalTrials.
gov,goesbeyondwhatiscurrentlyrequiredbylawsandregulationsintheUSandEU.
Equally,ourpolicyofpostingasimilarlevelofdetailforallournon-interventionalstudiestoGSK'sStudyRegistergoesbeyondcurrentlegalandregulatoryrequirements.
–Inordertosupportabroaderunderstandingofourclinicalresearch,in2017webegancreating"plainlanguage"summariesofsomeclinicaltrialresults.
Aswegainexperienceandfeedback,wemayincreasinglycreatethesesummaries.
Wepostourplainlanguagesummariesonwww.
trialsummaries.
com.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs2–Weconsidertheabovepostingsontheinternettobesupplementaryto,andnotareplacementfor,theneedtopublishstudiesinpeerreviewedjournals.
Ourapproachistosubmitstudiesasmorecomprehensivemanuscriptsforpublicationinpeerreviewedjournals,withanincreasingfocusonopenaccessjournals,thatareindexedbyonlinesearchenginessuchasMedlineandEmbase.
Themanuscriptsaresubmittedwithin18monthsofstudycompletion,regardlessofmarketauthorisationortermination.
–Weaimtopublishallinterventionalandnon-interventionalstudiesthatevaluatetheefficacy,safety,oreffectivenessofourproductsinhumans.
Wealsoaimtopublishotherhumansubjectresearchwhentheresultsprovideimportantscientificknowledgeorarerelevantforpatientcare.
–GSK'spolicyprohibits"ghostwriting"ofjournalmanuscriptsandabstractsbyrequiringauthorshipandacknowledgementsforscientificpublicationsconsistentwiththerequirementsoftheInternationalCommitteeofMedicalJournalEditors(ICMJE).
GSKandexternalmedicalwritersareeithernamedasauthorsorincludedintheacknowledgementsectionofmanuscripts.
–SinceDecember2013,wehavemadeClinicalStudyReports(CSRs1)publiclyavailablethroughourClinicalStudyRegister,oncethemedicinebeingstudiedisapprovedorterminatedfromdevelopment.
WehavealsopostedCSRsforinterventionalclinicaltrials(phaseI-IV)forapprovedandterminatedmedicinesdatingbacktotheformationofGSKin2000,aswellallnon-interventionalstudiesconductedsince2009thathaveevaluatedthesafetyandefficacyofourmedicines.
–ThenamesoftheinvestigatorswhoparticipateinourclinicalstudiesthatwereinitiatedafterJanuary2009areavailableonourClinicalStudyRegister.
GSKalsodisclosespaymentsmadetohealthcareprofessionals(HCPs)andhealthcareorganisations(HCOs)involvedinGSK-sponsoredclinicaltrialsinmarketsincludingtheUSA,Europe,JapanandAustralia.
ThiscommitmentbeganinMarch2011,withdisclosureofindividualpaymentsmadetoUSHCPsandtheirHCOsforresearchstudiesinitiatedfromJanuary2010onwards.
WhileinEurope,wehavepublishedourtransfersofvaluemadetoHCPsandtheirHCOsforresearchactivitiesasanaggregatefiguresince2016.
–InMay2013,GSKlaunchedanonlinesystemtoenableresearcherstorequestaccesstoanonymisedpatientleveldatafromgloballyconductedGSKclinicaltrials(phaseI-IV)ofmedicinesstartedsince2000.
Studiesarelistedonthissystemwithinsixmonthsofpublication.
RequestsforaccesstoparticipantleveldatafromConsumerHealthcarestudiesmayalsobesubmitted.
Thissystemisnowaconsortiumofseveralindustrysponsorsandnon-industryresearchfunders.
Itcanbefoundatwww.
clinicalstudydatarequest.
com.
–Wereinforcethesecommitmentsbycontinuallyassessingourperformance.
Amonthlydashboardofmetricsonourtransparencyactivitiesismaintainedaspartofourinternalbusinessmonitoring.
BackgroundTheClinicalDevelopmentProcessEvaluationofaninvestigationalproductisdonethroughinterventionalclinicaltrialsandisusuallyconductedinfourmainphases.
Eachphaseaddressesdifferentquestionsthatdetermineiftestingshouldproceedtothenextphase.
PhaseI:PhaseIstudiesareprimarilyconcernedwithassessingtheinvestigationalproduct'ssafetyusuallyinasmallnumberofhealthyhumanvolunteers(typicallybetween20and100people)andaredesignedtodeterminewhathappenstotheinvestigationalproductinthehumanbody.
1CSRsareformalstudyreportsthatprovidemoredetailsonthedesign,methodsandresultsofclinicaltrialsandformthebasisofsubmissionstotheUSFDA,EMAandotherregulatoryagencies.
PatientdataintheCSRsandappendiceswithpatientdatalistingsareremovedtoprotectpatientconfidentiality.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs3PhaseII:AninvestigationalproductthatpassesPhaseItestingthenmovesontoPhaseII,whichusuallyincludesthe"proofofconcept"stage.
Hereforthefirsttime,itisgenerallyadministeredtocarefullyselectedpatientssufferingfromthediseasewhichtheproductwillpotentiallytreat.
Generally100–300patientsareenrolledinthesePhaseIIstudies.
Prophylacticvaccinetrialsenrolluptoseveralhundredhealthyvolunteers.
Therapeuticvaccinetrialsenrolvolunteerswhoarealreadyinfectedorhavethedisease.
Theaimofthestudiesistodetermineiftheinvestigationalproductaddressestheillnessitisintendedfor,aswellastheamountandfrequencyofdosingnecessarytoachievetheoptimalbenefitsforpatientswiththefewestsideeffects.
PhaseIII:InPhaseIIIstudies,theinvestigationalproductisgiventohundredsandfrequentlythousandsofpatients.
PhaseIIIstudiesrequiredifferingperiodsoftimetocomplete,dependingonthediseasebeingstudied.
PhaseIV:Trialsofaninvestigationalproductmaycontinueafterithasbeenapprovedformarketing.
KnownasPhaseIVtrials,theymayfurtherevaluatetheeffectoftheproductfortheapproveduse;assessotherpotentialuses;oryieldadditionalsafetydata.
Regulatoryagenciesmayrequirethesetrialstoaddressspecificquestions.
Othertypesofclinicalresearch,suchasnon-interventionalresearch(usingdatacollectedduringtheprovisionofroutinehealthcare)andanalysesofdatathatiscombinedfromanumberofclinicaltrials(e.
g.
meta-analyses),areincreasinglyseenasimportantevidenceintheevaluationoftherisksandbenefitsofproducts.
GSK&ClinicalResearchDataTheguidingprincipleforGSKistodisclosepubliclytheresultsofGSK-sponsoredclinicalresearchthatevaluatesourproducts,irrespectiveofwhethertheresultsarelikelytobeperceivedaspositiveornegative.
Likewise,werequireinvestigatorsponsoredstudiessupportedbyGSKtobeconductedandpubliclydisclosedconsistentwithourpolicies.
ToinforminvestigatorsoftheoutcomeofaGSK-sponsoredtrial,GSKaimstoprovidesthemwithasummaryoftheoveralltrialresultswhichweencouragethemtosharewithstudyparticipants.
Internet-basedRegistrationofOngoingClinicalTrialsPubliclyavailableinternet-basedregistrationofongoingclinicalresearchcanhelptoincreaseparticipation.
Italsoprovidesanimportantreferencepointsointerestedpartiescantrackthesubsequentpublicdisclosureoftheresults.
PublicationandInternet-basedPostingofClinicalResearchResultsTraditionally,researchresultshavebeenpubliclydisclosedviapublicationinpeerreviewedscientificliterature;however,therearewellrecognisedconstraintsassociatedwiththisapproach.
Withlimitedjournalcapacity,somestudiesoranalysesmaynotbeconsideredaprioritybysomejournals,andthereforemaynotbeacceptedforpublication.
Attimes,throughagovernancereviewprocess,wealsorecognisethatapublicationattemptwouldbefutileforreasonssuchasearlystudyterminationwithlimitedenrollment.
Postingresultsummariesoninternet-basedregistersispartofasolutionasitensuresthattheresultsofclinicalstudiesareavailableinthepublicdomainwhetherornottheyareacceptedforpublication.
Regulatoryrequirementstodiscloseresearchresultshavethereforearisenandnumerouspublicregisters[e.
g.
http://www.
clinicaltrials.
gov/]havebeenestablishedtoserveasrepositoriesforthisinformation.
Wherewearenotthesponsorofastudy,forexample,wherewesupportastudybyprovidingaGSKproduct,weseekagreementthatexternalresearcherswillpostprotocolandresultsummariesoninternet-basedregistersandsubmittheresultsforpublicationinasearchablepeer-reviewedjournal.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs4AuthorshipAuthorshipandacknowledgementsformanuscriptsfollowICMJEcriteria(http://www.
icmje.
org)andaredeterminedbasedonthelevelofintellectualcontributiontostudydesign,dataacquisition,analysisandinterpretation,andwritingorrevisingthemanuscript.
Somejournals,however,haveamorenarrowdefinitionofauthorshipandthisconventionisfollowedforsuchjournals.
Thenamedprimaryauthorforapapermustactivelyparticipateinthedraftingprocessandleadthecontentdevelopmentofmanuscripts.
Theprimaryauthorworkscloselywithco-authorsandtogethertheyhavefinalapprovalauthorityforthemanuscript.
AnyGSKstafforcontractorssuchasprofessionalmedicalwriterswhocontributetothedevelopmentofmanuscriptsforauthors(e.
g.
assistanceinassemblinginitialdrafts,tablesandfigures,collatingco-authorcommentsandrevisingthedocumentbasedonauthorinput)arenamedinthearticleeitherasauthorswhentheircontributionmeetsauthorshipcriteria,orbydescriptionoftheircontributionwithintheacknowledgementssection.
GSKwillprovideauthorsfullaccesstodatasupportingthepublicationincludingaccesstodatatables,finalstudyreports,casereportformsandrawdataasneeded.
Wedonotsuppressorvetosubmissionofmanuscripts;thoughthetimingofsubmissionsmayonoccasionneedtobedelayedtoallowGSKtheopportunitytoseeknecessaryintellectualpropertyprotection.
GSKgenerallydoesnotsupportpublicationofdatafromanindividualcentreinamulti-centretrial.
ItisGSK'spositionthattheresultsfromtheentiretrialshouldbepublishedbeforeinformationfromindividualcentresispublished,andthatindividualcentredatashouldalwaysreferencetheprimarypublicationoftheentirestudy.
AccesstoPatientLevelDataPublicationofclinicalstudiesinthescientificliteratureandresultsummariesonregisterstypicallyonlycontainaggregateddata.
Thesepublicationsthereforehavelimitationsforthosewhowishtoexaminethedatamorecloselyortocombineitwithotherstudiesinmeta-analyses.
Toaddresstheselimitations,thereneedstobegreateraccesstounderlyingpatientleveldata.
InMay2013,wethereforeestablishedanonlinesystemtoallowresearcherstorequestaccesstoanonymisedpatientleveldatafromGSKsponsoredclinicaltrials.
Clinicalstudiesforourmedicinesarelistedonthewebsitewithin6monthsofpublicationoftheprimaryendpointsofthestudy.
Studiesthathavenotbeenacceptedforpublicationandarenolongerbeingprogressedarealsoavailable,asarestudiesofapprovedmedicinesforindicationsthatareterminatedfromdevelopment.
AnonymisedpatientleveldataismadeavailablefromGSKmedicines'studiesprovidedthatanexternalIndependentReviewPanelapprovesanassociatedresearchproposalandtheinvestigatorsignsaDataSharingAgreement.
TheIndependentReviewPanelacceptsorrejectsproposalsbasedonthescientificrationaleandrelevancetomedicalscienceorpatientcare.
ThePanelalsoconsidersthequalificationsoftheinvestigators,themanagementofpotentialconflictsofinterestandpublicationplans.
Accesstothedataisprovidedinasecuremannertohelpensurepatientprivacyisprotectedandthedataisusedonlyfortheintendedpurpose.
Togetherwithothersponsorsofclinicaltrials,GSKisencouragingabroadersharingofclinicaltrialdatatoenableresearcherstocombinedatafrommultiplesponsors(bothindustryandnon-industry),andtohelpensurethatthecontributionofstudyvolunteerstomedicalinnovationisfullyrealised.
June2019
Thisdisclosureisinadditiontosubmissionofinformationtonationalorregionalregulatoryauthoritiesaspartoftheproductdevelopmentorapprovalprocess.
Despitethis,however,concernshavebeenraisedbysomestakeholdersabout:–whetherstudyresultswhichmaybeviewedas"negative"forcompanies'medicinesarepublishedinscientificliterature;–whetherpublishedstudiesaccuratelyreflecttheconductofthestudy;–whethersomejournalarticlesare"ghost-written"(wheredoctorsputtheirnametoarticleswrittenbypharmaceuticalcompanies);–thelackofaccesstotheunderlyingpatientleveldatathatiscollectedduringthestudy.
ThispaperoutlinesGSK'sapproachtoaddressingtheseissues.
Itisanapproachthatdatesbackto2004whenwebecamethefirstcompanytolaunchaninternet-basedclinicalstudyregisterhttp://www.
gsk-studyregister.
com/providingresultsummariesfromourclinicaltrialsofmarketedmedicines(i.
epharmaceuticalsandvaccines)irrespectiveofthestudyoutcome.
Ithasevolvedsignificantlyaswehavesoughtnewwaysofenhancingthetransparencyofourclinicalresearch,includingthatconductedbyourconsumerhealthcaredivision.
GSK'sdisclosurepolicygoesbeyondwhatisrequiredbylawsandregulations.
GSK'sPosition–Beforethefirstsubjectisenrolledinastudy,wepostprotocolsummariesofGSK-sponsoredinterventionalandnon-interventionalstudiesoninternetregisters.
ThisincludesphaseI-IVclinicaltrialsofbothinvestigationalandmarketedmedicinesandconsumerhealthcareproducts.
Thiscommitmentalsoincludespooledanalyses/meta-analysesthatevaluateourproducts.
–WepostourinterventionalstudiesonClinicalTrials.
gov(http://www.
clinicaltrials.
gov).
Wepostournon-interventional,observationalstudiesonourGSKStudyRegister(http://www.
gsk-studyregister.
com).
–Irrespectiveoftheoutcomeofthestudy,wepostresultsummarieswithin12monthsofprimarycompletiondateforinterventionalstudiesand12monthsfromthecompletionofanalysisfornon-interventionalstudies.
Wedonotwaituntilapprovalorterminationofthemedicinebeforepostingresultsummaries.
Atthetimeofresults'registration,wealsopostthefullprotocolandthestatisticalanalysisplan.
–Ourcommitmentincludespostingresultsfromstudiesofterminatedcompoundsinordertohelpinformthescientificcommunityaboutnon-productiveareasofresearchandtoreduceunnecessaryexposureofstudyparticipantstosimilarcompoundsinotherclinicaltrials.
–GSK'scommitmenttopostprotocolsummaries,analysisplansandresultsummariesforallPhaseIstudies(inadditiontoPhasesII-IV)toClinicalTrials.
gov,goesbeyondwhatiscurrentlyrequiredbylawsandregulationsintheUSandEU.
Equally,ourpolicyofpostingasimilarlevelofdetailforallournon-interventionalstudiestoGSK'sStudyRegistergoesbeyondcurrentlegalandregulatoryrequirements.
–Inordertosupportabroaderunderstandingofourclinicalresearch,in2017webegancreating"plainlanguage"summariesofsomeclinicaltrialresults.
Aswegainexperienceandfeedback,wemayincreasinglycreatethesesummaries.
Wepostourplainlanguagesummariesonwww.
trialsummaries.
com.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs2–Weconsidertheabovepostingsontheinternettobesupplementaryto,andnotareplacementfor,theneedtopublishstudiesinpeerreviewedjournals.
Ourapproachistosubmitstudiesasmorecomprehensivemanuscriptsforpublicationinpeerreviewedjournals,withanincreasingfocusonopenaccessjournals,thatareindexedbyonlinesearchenginessuchasMedlineandEmbase.
Themanuscriptsaresubmittedwithin18monthsofstudycompletion,regardlessofmarketauthorisationortermination.
–Weaimtopublishallinterventionalandnon-interventionalstudiesthatevaluatetheefficacy,safety,oreffectivenessofourproductsinhumans.
Wealsoaimtopublishotherhumansubjectresearchwhentheresultsprovideimportantscientificknowledgeorarerelevantforpatientcare.
–GSK'spolicyprohibits"ghostwriting"ofjournalmanuscriptsandabstractsbyrequiringauthorshipandacknowledgementsforscientificpublicationsconsistentwiththerequirementsoftheInternationalCommitteeofMedicalJournalEditors(ICMJE).
GSKandexternalmedicalwritersareeithernamedasauthorsorincludedintheacknowledgementsectionofmanuscripts.
–SinceDecember2013,wehavemadeClinicalStudyReports(CSRs1)publiclyavailablethroughourClinicalStudyRegister,oncethemedicinebeingstudiedisapprovedorterminatedfromdevelopment.
WehavealsopostedCSRsforinterventionalclinicaltrials(phaseI-IV)forapprovedandterminatedmedicinesdatingbacktotheformationofGSKin2000,aswellallnon-interventionalstudiesconductedsince2009thathaveevaluatedthesafetyandefficacyofourmedicines.
–ThenamesoftheinvestigatorswhoparticipateinourclinicalstudiesthatwereinitiatedafterJanuary2009areavailableonourClinicalStudyRegister.
GSKalsodisclosespaymentsmadetohealthcareprofessionals(HCPs)andhealthcareorganisations(HCOs)involvedinGSK-sponsoredclinicaltrialsinmarketsincludingtheUSA,Europe,JapanandAustralia.
ThiscommitmentbeganinMarch2011,withdisclosureofindividualpaymentsmadetoUSHCPsandtheirHCOsforresearchstudiesinitiatedfromJanuary2010onwards.
WhileinEurope,wehavepublishedourtransfersofvaluemadetoHCPsandtheirHCOsforresearchactivitiesasanaggregatefiguresince2016.
–InMay2013,GSKlaunchedanonlinesystemtoenableresearcherstorequestaccesstoanonymisedpatientleveldatafromgloballyconductedGSKclinicaltrials(phaseI-IV)ofmedicinesstartedsince2000.
Studiesarelistedonthissystemwithinsixmonthsofpublication.
RequestsforaccesstoparticipantleveldatafromConsumerHealthcarestudiesmayalsobesubmitted.
Thissystemisnowaconsortiumofseveralindustrysponsorsandnon-industryresearchfunders.
Itcanbefoundatwww.
clinicalstudydatarequest.
com.
–Wereinforcethesecommitmentsbycontinuallyassessingourperformance.
Amonthlydashboardofmetricsonourtransparencyactivitiesismaintainedaspartofourinternalbusinessmonitoring.
BackgroundTheClinicalDevelopmentProcessEvaluationofaninvestigationalproductisdonethroughinterventionalclinicaltrialsandisusuallyconductedinfourmainphases.
Eachphaseaddressesdifferentquestionsthatdetermineiftestingshouldproceedtothenextphase.
PhaseI:PhaseIstudiesareprimarilyconcernedwithassessingtheinvestigationalproduct'ssafetyusuallyinasmallnumberofhealthyhumanvolunteers(typicallybetween20and100people)andaredesignedtodeterminewhathappenstotheinvestigationalproductinthehumanbody.
1CSRsareformalstudyreportsthatprovidemoredetailsonthedesign,methodsandresultsofclinicaltrialsandformthebasisofsubmissionstotheUSFDA,EMAandotherregulatoryagencies.
PatientdataintheCSRsandappendiceswithpatientdatalistingsareremovedtoprotectpatientconfidentiality.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs3PhaseII:AninvestigationalproductthatpassesPhaseItestingthenmovesontoPhaseII,whichusuallyincludesthe"proofofconcept"stage.
Hereforthefirsttime,itisgenerallyadministeredtocarefullyselectedpatientssufferingfromthediseasewhichtheproductwillpotentiallytreat.
Generally100–300patientsareenrolledinthesePhaseIIstudies.
Prophylacticvaccinetrialsenrolluptoseveralhundredhealthyvolunteers.
Therapeuticvaccinetrialsenrolvolunteerswhoarealreadyinfectedorhavethedisease.
Theaimofthestudiesistodetermineiftheinvestigationalproductaddressestheillnessitisintendedfor,aswellastheamountandfrequencyofdosingnecessarytoachievetheoptimalbenefitsforpatientswiththefewestsideeffects.
PhaseIII:InPhaseIIIstudies,theinvestigationalproductisgiventohundredsandfrequentlythousandsofpatients.
PhaseIIIstudiesrequiredifferingperiodsoftimetocomplete,dependingonthediseasebeingstudied.
PhaseIV:Trialsofaninvestigationalproductmaycontinueafterithasbeenapprovedformarketing.
KnownasPhaseIVtrials,theymayfurtherevaluatetheeffectoftheproductfortheapproveduse;assessotherpotentialuses;oryieldadditionalsafetydata.
Regulatoryagenciesmayrequirethesetrialstoaddressspecificquestions.
Othertypesofclinicalresearch,suchasnon-interventionalresearch(usingdatacollectedduringtheprovisionofroutinehealthcare)andanalysesofdatathatiscombinedfromanumberofclinicaltrials(e.
g.
meta-analyses),areincreasinglyseenasimportantevidenceintheevaluationoftherisksandbenefitsofproducts.
GSK&ClinicalResearchDataTheguidingprincipleforGSKistodisclosepubliclytheresultsofGSK-sponsoredclinicalresearchthatevaluatesourproducts,irrespectiveofwhethertheresultsarelikelytobeperceivedaspositiveornegative.
Likewise,werequireinvestigatorsponsoredstudiessupportedbyGSKtobeconductedandpubliclydisclosedconsistentwithourpolicies.
ToinforminvestigatorsoftheoutcomeofaGSK-sponsoredtrial,GSKaimstoprovidesthemwithasummaryoftheoveralltrialresultswhichweencouragethemtosharewithstudyparticipants.
Internet-basedRegistrationofOngoingClinicalTrialsPubliclyavailableinternet-basedregistrationofongoingclinicalresearchcanhelptoincreaseparticipation.
Italsoprovidesanimportantreferencepointsointerestedpartiescantrackthesubsequentpublicdisclosureoftheresults.
PublicationandInternet-basedPostingofClinicalResearchResultsTraditionally,researchresultshavebeenpubliclydisclosedviapublicationinpeerreviewedscientificliterature;however,therearewellrecognisedconstraintsassociatedwiththisapproach.
Withlimitedjournalcapacity,somestudiesoranalysesmaynotbeconsideredaprioritybysomejournals,andthereforemaynotbeacceptedforpublication.
Attimes,throughagovernancereviewprocess,wealsorecognisethatapublicationattemptwouldbefutileforreasonssuchasearlystudyterminationwithlimitedenrollment.
Postingresultsummariesoninternet-basedregistersispartofasolutionasitensuresthattheresultsofclinicalstudiesareavailableinthepublicdomainwhetherornottheyareacceptedforpublication.
Regulatoryrequirementstodiscloseresearchresultshavethereforearisenandnumerouspublicregisters[e.
g.
http://www.
clinicaltrials.
gov/]havebeenestablishedtoserveasrepositoriesforthisinformation.
Wherewearenotthesponsorofastudy,forexample,wherewesupportastudybyprovidingaGSKproduct,weseekagreementthatexternalresearcherswillpostprotocolandresultsummariesoninternet-basedregistersandsubmittheresultsforpublicationinasearchablepeer-reviewedjournal.
GSKPublicpolicypositionsApublicationofGlaxoSmithKlineCommunicationsandGovernmentAffairs4AuthorshipAuthorshipandacknowledgementsformanuscriptsfollowICMJEcriteria(http://www.
icmje.
org)andaredeterminedbasedonthelevelofintellectualcontributiontostudydesign,dataacquisition,analysisandinterpretation,andwritingorrevisingthemanuscript.
Somejournals,however,haveamorenarrowdefinitionofauthorshipandthisconventionisfollowedforsuchjournals.
Thenamedprimaryauthorforapapermustactivelyparticipateinthedraftingprocessandleadthecontentdevelopmentofmanuscripts.
Theprimaryauthorworkscloselywithco-authorsandtogethertheyhavefinalapprovalauthorityforthemanuscript.
AnyGSKstafforcontractorssuchasprofessionalmedicalwriterswhocontributetothedevelopmentofmanuscriptsforauthors(e.
g.
assistanceinassemblinginitialdrafts,tablesandfigures,collatingco-authorcommentsandrevisingthedocumentbasedonauthorinput)arenamedinthearticleeitherasauthorswhentheircontributionmeetsauthorshipcriteria,orbydescriptionoftheircontributionwithintheacknowledgementssection.
GSKwillprovideauthorsfullaccesstodatasupportingthepublicationincludingaccesstodatatables,finalstudyreports,casereportformsandrawdataasneeded.
Wedonotsuppressorvetosubmissionofmanuscripts;thoughthetimingofsubmissionsmayonoccasionneedtobedelayedtoallowGSKtheopportunitytoseeknecessaryintellectualpropertyprotection.
GSKgenerallydoesnotsupportpublicationofdatafromanindividualcentreinamulti-centretrial.
ItisGSK'spositionthattheresultsfromtheentiretrialshouldbepublishedbeforeinformationfromindividualcentresispublished,andthatindividualcentredatashouldalwaysreferencetheprimarypublicationoftheentirestudy.
AccesstoPatientLevelDataPublicationofclinicalstudiesinthescientificliteratureandresultsummariesonregisterstypicallyonlycontainaggregateddata.
Thesepublicationsthereforehavelimitationsforthosewhowishtoexaminethedatamorecloselyortocombineitwithotherstudiesinmeta-analyses.
Toaddresstheselimitations,thereneedstobegreateraccesstounderlyingpatientleveldata.
InMay2013,wethereforeestablishedanonlinesystemtoallowresearcherstorequestaccesstoanonymisedpatientleveldatafromGSKsponsoredclinicaltrials.
Clinicalstudiesforourmedicinesarelistedonthewebsitewithin6monthsofpublicationoftheprimaryendpointsofthestudy.
Studiesthathavenotbeenacceptedforpublicationandarenolongerbeingprogressedarealsoavailable,asarestudiesofapprovedmedicinesforindicationsthatareterminatedfromdevelopment.
AnonymisedpatientleveldataismadeavailablefromGSKmedicines'studiesprovidedthatanexternalIndependentReviewPanelapprovesanassociatedresearchproposalandtheinvestigatorsignsaDataSharingAgreement.
TheIndependentReviewPanelacceptsorrejectsproposalsbasedonthescientificrationaleandrelevancetomedicalscienceorpatientcare.
ThePanelalsoconsidersthequalificationsoftheinvestigators,themanagementofpotentialconflictsofinterestandpublicationplans.
Accesstothedataisprovidedinasecuremannertohelpensurepatientprivacyisprotectedandthedataisusedonlyfortheintendedpurpose.
Togetherwithothersponsorsofclinicaltrials,GSKisencouragingabroadersharingofclinicaltrialdatatoenableresearcherstocombinedatafrommultiplesponsors(bothindustryandnon-industry),andtohelpensurethatthecontributionofstudyvolunteerstomedicalinnovationisfullyrealised.
June2019
- reviewedregister.com相关文档
- Frequencyregister.com
- makingregister.com
- unnecessaryregister.com
- emphasisedregister.com
- estimateregister.com
- retainregister.com
香港2GB内存DIYVM2核(¥50月)香港沙田CN2云服务器
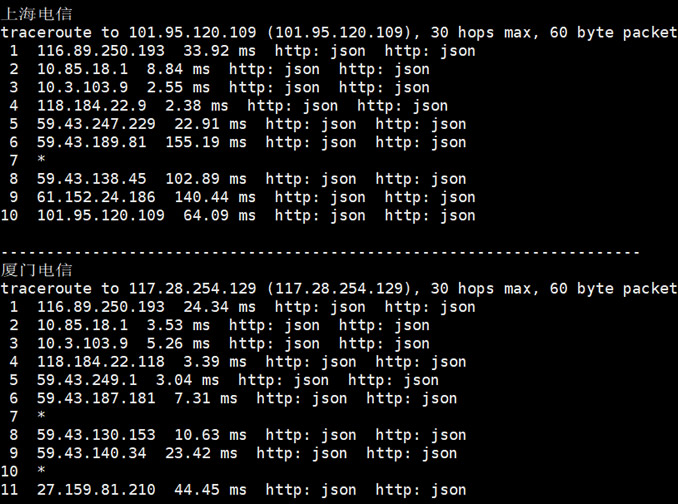
DiyVM 香港沙田机房,也是采用的CN2优化线路,目前也有入手且在使用中,我个人感觉如果中文业务需要用到的话虽然日本机房也是CN2,但是线路的稳定性不如香港机房,所以我们在这篇文章中亲测看看香港机房,然后对比之前看到的日本机房。香港机房的配置信息。CPU内存 硬盘带宽IP价格购买地址2核2G50G2M1¥50/月选择方案4核4G60G3M1¥100/月选择方案4核8G70G3M4¥200/月选择...
PIGYUN:美国联通CUVIPCUVIP限时cuvip、AS9929、GIA/韩国CN2机房限时六折
pigyun怎么样?PIGYunData成立于2019年,2021是PIGYun为用户提供稳定服务的第三年,目前商家提供香港CN2线路、韩国cn2线路、美西CUVIP-9929、GIA等线路优质VPS,基于KVM虚拟架构,商家采用魔方云平台,所有的配置都可以弹性选择,目前商家推出了七月优惠,韩国和美国所有线路都有相应的促销,六折至八折,性价比不错。点击进入:PIGYun官方网站地址PIGYUN优惠...

UCloud新人优惠中国香港/日本/美国云服务器低至4元
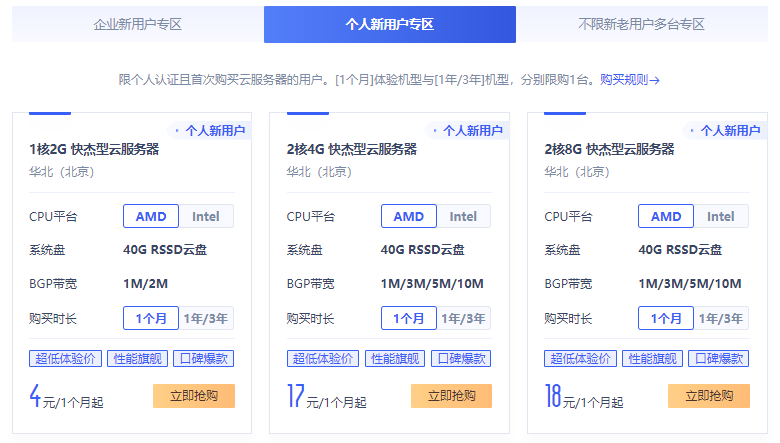
UCloud优刻得商家这几年应该已经被我们不少的个人站长用户认知,且确实在当下阿里云、腾讯云服务商不断的只促销服务于新用户活动,给我们很多老用户折扣的空间不多。于是,我们可以通过拓展选择其他同类服务商享受新人的福利,这里其中之一就选择UCloud商家。UCloud服务商2020年创业板上市的,实际上很早就有认识到,那时候价格高的离谱,谁让他们只服务有钱的企业用户呢。这里希望融入到我们大众消费者,你...
register.com为你推荐
-
台湾主机香港,美国,台湾,韩国,日本主机到底哪个好网站空间价格1M网站空间是多少钱虚拟主机软件谁知道这个虚拟机软件叫什么。虚拟主机软件虚拟主机管理软件那个最好用?北京虚拟主机租用北京云主机租用哪家资质正规,价格便宜,服务好?要真云主机不要那种vps的假云主机,机房要在北京的!虚拟主机mysql怎么管理虚拟主机上的MYSQL?(高分回报)asp虚拟主机ASP源码上传到虚拟主机什么地方jsp虚拟主机java虚拟主机空间怎么选择,国内jsp虚拟主机比较稳定,现在java项目做好后需要推荐一下吧虚拟主机测评我们可以用哪些命令来测试一个虚拟主机的好坏?虚拟主机排名换一台虚拟主机会影响排名吗?