practicesoray花生壳
oray花生壳 时间:2021-04-03 阅读:()
U.
S.
Food&DrugAdministration10903NewHampshireAvenueDocID#04017.
02.
12SilverSpring,MD20993www.
fda.
govAugust17,2018GENORAYCo.
,Ltd.
℅Ms.
KaitlynnMinBusinessDevelopmentGENORAYAmericaInc.
147E.
BristolLaneORANGECA92780Re:K181943Trade/DeviceName:OSCAR(OSCARPrime,OSCARClassic)RegulationNumber:21CFR892.
1650RegulationName:Image-intensifiedfluoroscopicx-raysystemRegulatoryClass:IIProductCode:OWB,JAADated:July5,2018Received:July20,2018DearMs.
Min:WehavereviewedyourSection510(k)premarketnotificationofintenttomarketthedevicereferencedaboveandhavedeterminedthedeviceissubstantiallyequivalent(fortheindicationsforusestatedintheenclosure)tolegallymarketedpredicatedevicesmarketedininterstatecommercepriortoMay28,1976,theenactmentdateoftheMedicalDeviceAmendments,ortodevicesthathavebeenreclassifiedinaccordancewiththeprovisionsoftheFederalFood,Drug,andCosmeticAct(Act)thatdonotrequireapprovalofapremarketapprovalapplication(PMA).
Youmay,therefore,marketthedevice,subjecttothegeneralcontrolsprovisionsoftheAct.
Althoughthisletterreferstoyourproductasadevice,pleasebeawarethatsomeclearedproductsmayinsteadbecombinationproducts.
The510(k)PremarketNotificationDatabaselocatedathttps://www.
accessdata.
fda.
gov/scripts/cdrh/cfdocs/cfpmn/pmn.
cfmidentifiescombinationproductsubmissions.
ThegeneralcontrolsprovisionsoftheActincluderequirementsforannualregistration,listingofdevices,goodmanufacturingpractice,labeling,andprohibitionsagainstmisbrandingandadulteration.
Pleasenote:CDRHdoesnotevaluateinformationrelatedtocontractliabilitywarranties.
Weremindyou,however,thatdevicelabelingmustbetruthfulandnotmisleading.
Ifyourdeviceisclassified(seeabove)intoeitherclassII(SpecialControls)orclassIII(PMA),itmaybesubjecttoadditionalcontrols.
ExistingmajorregulationsaffectingyourdevicecanbefoundintheCodeofFederalRegulations,Title21,Parts800to898.
Inaddition,FDAmaypublishfurtherannouncementsconcerningyourdeviceintheFederalRegister.
PleasebeadvisedthatFDA'sissuanceofasubstantialequivalencedeterminationdoesnotmeanthatFDAhasmadeadeterminationthatyourdevicecomplieswithotherrequirementsoftheActoranyFederalPage2–Ms.
KaitlynnMinK181943statutesandregulationsadministeredbyotherFederalagencies.
YoumustcomplywithalltheAct'srequirements,including,butnotlimitedto:registrationandlisting(21CFRPart807);labeling(21CFRPart801);medicaldevicereporting(reportingofmedicaldevice-relatedadverseevents)(21CFR803)fordevicesorpostmarketingsafetyreporting(21CFR4,SubpartB)forcombinationproducts(seehttps://www.
fda.
gov/CombinationProducts/GuidanceRegulatoryInformation/ucm597488.
htm);goodmanufacturingpracticerequirementsassetforthinthequalitysystems(QS)regulation(21CFRPart820)fordevicesorcurrentgoodmanufacturingpractices(21CFR4,SubpartA)forcombinationproducts;and,ifapplicable,theelectronicproductradiationcontrolprovisions(Sections531-542oftheAct);21CFR1000-1050.
Also,pleasenotetheregulationentitled,"Misbrandingbyreferencetopremarketnotification"(21CFRPart807.
97).
ForquestionsregardingthereportingofadverseeventsundertheMDRregulation(21CFRPart803),pleasegotohttp://www.
fda.
gov/MedicalDevices/Safety/ReportaProblem/default.
htm.
Forcomprehensiveregulatoryinformationaboutmedicaldevicesandradiation-emittingproducts,includinginformationaboutlabelingregulations,pleaseseeDeviceAdvice(https://www.
fda.
gov/MedicalDevices/DeviceRegulationandGuidance/)andCDRHLearn(http://www.
fda.
gov/Training/CDRHLearn).
Additionally,youmaycontacttheDivisionofIndustryandConsumerEducation(DICE)toaskaquestionaboutaspecificregulatorytopic.
SeetheDICEwebsite(http://www.
fda.
gov/DICE)formoreinformationorcontactDICEbyemail(DICE@fda.
hhs.
gov)orphone(1-800-638-2041or301-796-7100).
Sincerely,RobertOchs,Ph.
D.
DirectorDivisionofRadiologicalHealthOfficeofInVitroDiagnosticsandRadiologicalHealthCenterforDevicesandRadiologicalHealthEnclosurenGjUSGsUX^VXZY`jvumpklu{phsK181943Exhibit5510(k)SummaryDateofSummaryPreparation:July.
05,20181.
SubmitterandUSOfficialCorrespondentSubmitter:GENORAYCo.
,Ltd.
Address:512,560,Dunchon-daero,Jungwon-gu,Seongnam-si,Gyeonggi-Do,KoreaTelephoneNo.
:+82-31-740-4100Fax:+82-31-737-8018OfficialCorrespondent(U.
S):KaitlynnMin-BusinessManagerCorrespondent:GENORAYAmericaInc.
Address:147E.
BristolLane,Orange,CA92865USATelephoneNo.
:+1-855-436-6729Fax:+1-714-786-8919Email:kaitlynn@genorayamerica.
com2.
EstablishmentRegistrationNumber30058434183.
DeviceInformationTrade/DeviceName:OSCAROSCARPrime,OSCARClassicRegulationName:Image-IntensifiedfluoroscopicX-raySystemClassificationName:InterventionalFluoroscopicX-RaySystemProductCode:OWB/InterventionalFluoroscopicX-RaySystemSubsequenceproductcode:JAA/System,X-Ray,Fluoroscopic,Image-IntensifiedDeviceClass:ClassIIperregulation21CFR892.
16504.
PredicateDevice(EquivalentLegallyMarketedDevice)Manufacturer:GENORAYCo.
,LtdDeviceName:ZEN-2090Pro510(k)Number:K091918(DecisionDate–October07,2009)ClassificationName:Image–IntensifiedFluoroscopicX-RaySystemPrimaryProductCode:OWB/InterventionalFluoroscopicX-raySystemSecondaryproductcode:JAA/system,x-ray,fluoroscopic,image-intensifiedOXO/image-intensifiedfluoroscopicx-raysystem,mobileDeviceClass:ClassIIperregulation21CFR892.
16505.
DescriptionoftheDeviceOSCARPrimeandOSCARClassicareclassifiedaccordingtotheoptionofimageacquisitionparts.
FlatpaneldetectorisOSCARPrime,andImageintensifierisOSCARClassic.
AndtheyarecalledOSCARasthebrandname.
OSCARisconsistofX-rayTube,X-raytubeassembly,x-raycontroller,imagereceptorandsomeaccessories.
Thereisnowirelessfunctioninthisdevice.
TheOSCAR,C-ArmMobileisthedeviceintendedtovisualizeanatomicalstructuresbyconvertingapatternofx-radiationintoavisibleimagethroughelectronicamplification.
Thisdeviceisusedforprovidingfluoroscopicandradiographicimagesofpatientanatomy,especiallyduringthespecialproceduresinahospitalormedicalclinics.
Thefluoroscopicmodeofoperationisveryusefultotheattendingphysiciantoseetheimagesonrealtimewithouttheneedtodevelopindividualfilms.
6.
IndicationsforuseOSCARisamobilefluoroscopysystemdesignedtoprovidefluoroscopicandspotfilmimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,urologic,orthopedic,neurologic,vascular,andstonelocalization.
ThepatientgroupusingOSCARisallexceptchildren.
7.
SubstantialequivalencechartNameProposeddeviceOSCARPredicatedeviceZEN-2090ProManufacturerGENORAYCo.
,Ltd.
GENORAYCo.
,Ltd.
510(k)No.
-K091918IndicationsforuseOSCARisamobilefluoroscopysystemdesignedtoprovidefluoroscopicandspotfilmimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,urologic,orthopedic,neurologic,vascular,cardiacandcriticalcare.
ThepatientgroupusingOSCARisallexceptchildren.
ZEN-2090ProisamobiledigitalC-armdesignedtoprovidefluoroscopicandradiographicimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,orthopedic,neurologic,stonelocalization,criticalcareandemergencyroomproceduresi.
e.
surgicalinterventionsneedingX-rayimagingand/orguidanceandinterventionsinsideandoutsidetheoperatingroom.
GeneratorHighFrequencyInverterHighFrequencyInverterMax.
outputpower4.
5kW2.
2kWX-raytubeStationarytubeStationarytubeLarge:1.
4/Small:0.
5Large:1.
5mm/Small:0.
5mmDetectorOSCARPrime(Flatpanel)OSCARClassic(Imageintensifier)ZEN-2090Pro(ImageIntensifier)OptionA:GFD-200(DualRay-Q)(ClearedunderK172180)Activeimagearea:260x256mmCentralResolution:4.
6lp/mmType:CMOSResolution:2600x2560Pixelsamplingresolution:14bitsPixelpitch:100μmMTF:56%DQE:59%Scintillator:CsIOptionA:E5830SD-P4A(ClearedunderK140041)Activeimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)E5830SD-P4AActiveimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)OptionB:VIVIX-D0909GActiveimagearea:229.
12x229.
12mmCentralResolution:2.
7lp/mmType:a-SiTFTResolution:2600x2560Pixelsamplingresolution:16bitPixelpitch:179μmMTF:45%DQE:45%Scintillator:CsIOptionB:TH9438QX(ClearedunderK140041).
Activeimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)Fluoroscopy40-120kV/0.
2-6.
0mA40-110kV/0.
2-6.
0mAPulsedFluoroscopy0.
2-10.
0mA0.
2-6.
0mARadiography40-120kV/0.
6-200mAs40-110kV/0.
4-100mAsDimensionsSID:1000mmSID:960mmPanningRotation:±12.
5PanningRotation:±12.
5OrbitalRotation:155OrbitalRotation:120Vert.
Travel:500mmVert.
Travel:400mmHoriz.
Travel:200mmHoriz.
Travel:200mmTheproposedOSCARisbasedonthepredicatedevice,ZEN-2090Pro(K091918).
ThereareseveraldifferencebetweenOSCAR(Prime,Classic)andpredicatedevice(ZEN-2090Pro)istheoptionsofImageacquisitionparts.
OSCARPrimeistheflatpaneldetectorandpredicatedeviceisImageIntensifier.
Asmentionedinthecomparisontable,predicatedeviceDQEishigherthanOSCARPrime.
However,predicatedeviceDQEistheDQEoftheimageintensifieritself.
Ingeneral,whenimageintensifieriscombinedwithCCDcameratheDQEdecreases.
Inconclusion,theDQEofthecompletepredicatedeviceis51%.
SotheDQEoftheOSCARPrimeismoreeffectiveandsafetythanpredicatedevice.
Alsoflatpaneldetectortypehasexcellentimageuniformity,nogeometricdistortion,noveilingglareorvignetting,smallandthinphysicalsizeascomparedtotheImageIntensifiertype.
TheOSCARissubstantiallyequivalenttothepredicatedevice,ZEN-2090Pro(K091918).
8.
Safety,EMCandPerformancedatacomparisontoPredicateOSCARcomplieswithindustrystandardssuchasIEC60601-1Seriesand21CFR1020.
30,21CFR1020.
31and21CFR1020.
32tominimizeelectrical,mechanicalandradiationhazards.
-Electrical,mechanical,environmentalsafetyandperformancetestingaccordingtostandardIEC60601-1,IEC60601-1-3,IEC60601-1-6,IEC60601-2-28,IEC60601-2-43,IEC60601-2-54andIEC62366wereperformed.
-EMCtestingwasconductedinaccordancewithstandardIEC60601-1-2.
-OSCARmeetstheEPRCstandards(21CFR1020.
30,31,32).
-FDAguidance"guidanceforSSXIdevices",and"guidancefortheContentofPremarketSubmissionsforSoftwareContainedinMedicaldevices",wasperformedforOSCAR.
Thesoftwarefeatureshasbeenchangedcomparetopredicatedeviceasbelow:EnhanceImageCalibrationfunction.
DisplayexposuremodesshowsontheLCDtouchpanel.
AddAngiographyfunctions.
Changestothepredicatedevicesoftwareweretestedandtheydonotaffectthedevicesafetyandeffectiveness.
Also,thedevicesoftwareismoderatelevelofconcern.
Asaresults,alltestresultsweresatisfactoryandtheresultofbenchandclinicalevaluationindicatesthatthenewdeviceisassafeandeffectiveasthepredicatedevice.
9.
ConclusionInreferencetothecomparisoninformationprovidedinsubstantialequivalencechart,andthemostoffunctionsandelectronicfeaturesaresimilarwithpredicatedevice.
WebelievethattheOSCARissafeandeffectiveaspredicatedevice,andhasnonewindicationforuse.
Therefore,OSCARissubstantiallyequivalenttopredicatedevice.
S.
Food&DrugAdministration10903NewHampshireAvenueDocID#04017.
02.
12SilverSpring,MD20993www.
fda.
govAugust17,2018GENORAYCo.
,Ltd.
℅Ms.
KaitlynnMinBusinessDevelopmentGENORAYAmericaInc.
147E.
BristolLaneORANGECA92780Re:K181943Trade/DeviceName:OSCAR(OSCARPrime,OSCARClassic)RegulationNumber:21CFR892.
1650RegulationName:Image-intensifiedfluoroscopicx-raysystemRegulatoryClass:IIProductCode:OWB,JAADated:July5,2018Received:July20,2018DearMs.
Min:WehavereviewedyourSection510(k)premarketnotificationofintenttomarketthedevicereferencedaboveandhavedeterminedthedeviceissubstantiallyequivalent(fortheindicationsforusestatedintheenclosure)tolegallymarketedpredicatedevicesmarketedininterstatecommercepriortoMay28,1976,theenactmentdateoftheMedicalDeviceAmendments,ortodevicesthathavebeenreclassifiedinaccordancewiththeprovisionsoftheFederalFood,Drug,andCosmeticAct(Act)thatdonotrequireapprovalofapremarketapprovalapplication(PMA).
Youmay,therefore,marketthedevice,subjecttothegeneralcontrolsprovisionsoftheAct.
Althoughthisletterreferstoyourproductasadevice,pleasebeawarethatsomeclearedproductsmayinsteadbecombinationproducts.
The510(k)PremarketNotificationDatabaselocatedathttps://www.
accessdata.
fda.
gov/scripts/cdrh/cfdocs/cfpmn/pmn.
cfmidentifiescombinationproductsubmissions.
ThegeneralcontrolsprovisionsoftheActincluderequirementsforannualregistration,listingofdevices,goodmanufacturingpractice,labeling,andprohibitionsagainstmisbrandingandadulteration.
Pleasenote:CDRHdoesnotevaluateinformationrelatedtocontractliabilitywarranties.
Weremindyou,however,thatdevicelabelingmustbetruthfulandnotmisleading.
Ifyourdeviceisclassified(seeabove)intoeitherclassII(SpecialControls)orclassIII(PMA),itmaybesubjecttoadditionalcontrols.
ExistingmajorregulationsaffectingyourdevicecanbefoundintheCodeofFederalRegulations,Title21,Parts800to898.
Inaddition,FDAmaypublishfurtherannouncementsconcerningyourdeviceintheFederalRegister.
PleasebeadvisedthatFDA'sissuanceofasubstantialequivalencedeterminationdoesnotmeanthatFDAhasmadeadeterminationthatyourdevicecomplieswithotherrequirementsoftheActoranyFederalPage2–Ms.
KaitlynnMinK181943statutesandregulationsadministeredbyotherFederalagencies.
YoumustcomplywithalltheAct'srequirements,including,butnotlimitedto:registrationandlisting(21CFRPart807);labeling(21CFRPart801);medicaldevicereporting(reportingofmedicaldevice-relatedadverseevents)(21CFR803)fordevicesorpostmarketingsafetyreporting(21CFR4,SubpartB)forcombinationproducts(seehttps://www.
fda.
gov/CombinationProducts/GuidanceRegulatoryInformation/ucm597488.
htm);goodmanufacturingpracticerequirementsassetforthinthequalitysystems(QS)regulation(21CFRPart820)fordevicesorcurrentgoodmanufacturingpractices(21CFR4,SubpartA)forcombinationproducts;and,ifapplicable,theelectronicproductradiationcontrolprovisions(Sections531-542oftheAct);21CFR1000-1050.
Also,pleasenotetheregulationentitled,"Misbrandingbyreferencetopremarketnotification"(21CFRPart807.
97).
ForquestionsregardingthereportingofadverseeventsundertheMDRregulation(21CFRPart803),pleasegotohttp://www.
fda.
gov/MedicalDevices/Safety/ReportaProblem/default.
htm.
Forcomprehensiveregulatoryinformationaboutmedicaldevicesandradiation-emittingproducts,includinginformationaboutlabelingregulations,pleaseseeDeviceAdvice(https://www.
fda.
gov/MedicalDevices/DeviceRegulationandGuidance/)andCDRHLearn(http://www.
fda.
gov/Training/CDRHLearn).
Additionally,youmaycontacttheDivisionofIndustryandConsumerEducation(DICE)toaskaquestionaboutaspecificregulatorytopic.
SeetheDICEwebsite(http://www.
fda.
gov/DICE)formoreinformationorcontactDICEbyemail(DICE@fda.
hhs.
gov)orphone(1-800-638-2041or301-796-7100).
Sincerely,RobertOchs,Ph.
D.
DirectorDivisionofRadiologicalHealthOfficeofInVitroDiagnosticsandRadiologicalHealthCenterforDevicesandRadiologicalHealthEnclosurenGjUSGsUX^VXZY`jvumpklu{phsK181943Exhibit5510(k)SummaryDateofSummaryPreparation:July.
05,20181.
SubmitterandUSOfficialCorrespondentSubmitter:GENORAYCo.
,Ltd.
Address:512,560,Dunchon-daero,Jungwon-gu,Seongnam-si,Gyeonggi-Do,KoreaTelephoneNo.
:+82-31-740-4100Fax:+82-31-737-8018OfficialCorrespondent(U.
S):KaitlynnMin-BusinessManagerCorrespondent:GENORAYAmericaInc.
Address:147E.
BristolLane,Orange,CA92865USATelephoneNo.
:+1-855-436-6729Fax:+1-714-786-8919Email:kaitlynn@genorayamerica.
com2.
EstablishmentRegistrationNumber30058434183.
DeviceInformationTrade/DeviceName:OSCAROSCARPrime,OSCARClassicRegulationName:Image-IntensifiedfluoroscopicX-raySystemClassificationName:InterventionalFluoroscopicX-RaySystemProductCode:OWB/InterventionalFluoroscopicX-RaySystemSubsequenceproductcode:JAA/System,X-Ray,Fluoroscopic,Image-IntensifiedDeviceClass:ClassIIperregulation21CFR892.
16504.
PredicateDevice(EquivalentLegallyMarketedDevice)Manufacturer:GENORAYCo.
,LtdDeviceName:ZEN-2090Pro510(k)Number:K091918(DecisionDate–October07,2009)ClassificationName:Image–IntensifiedFluoroscopicX-RaySystemPrimaryProductCode:OWB/InterventionalFluoroscopicX-raySystemSecondaryproductcode:JAA/system,x-ray,fluoroscopic,image-intensifiedOXO/image-intensifiedfluoroscopicx-raysystem,mobileDeviceClass:ClassIIperregulation21CFR892.
16505.
DescriptionoftheDeviceOSCARPrimeandOSCARClassicareclassifiedaccordingtotheoptionofimageacquisitionparts.
FlatpaneldetectorisOSCARPrime,andImageintensifierisOSCARClassic.
AndtheyarecalledOSCARasthebrandname.
OSCARisconsistofX-rayTube,X-raytubeassembly,x-raycontroller,imagereceptorandsomeaccessories.
Thereisnowirelessfunctioninthisdevice.
TheOSCAR,C-ArmMobileisthedeviceintendedtovisualizeanatomicalstructuresbyconvertingapatternofx-radiationintoavisibleimagethroughelectronicamplification.
Thisdeviceisusedforprovidingfluoroscopicandradiographicimagesofpatientanatomy,especiallyduringthespecialproceduresinahospitalormedicalclinics.
Thefluoroscopicmodeofoperationisveryusefultotheattendingphysiciantoseetheimagesonrealtimewithouttheneedtodevelopindividualfilms.
6.
IndicationsforuseOSCARisamobilefluoroscopysystemdesignedtoprovidefluoroscopicandspotfilmimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,urologic,orthopedic,neurologic,vascular,andstonelocalization.
ThepatientgroupusingOSCARisallexceptchildren.
7.
SubstantialequivalencechartNameProposeddeviceOSCARPredicatedeviceZEN-2090ProManufacturerGENORAYCo.
,Ltd.
GENORAYCo.
,Ltd.
510(k)No.
-K091918IndicationsforuseOSCARisamobilefluoroscopysystemdesignedtoprovidefluoroscopicandspotfilmimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,urologic,orthopedic,neurologic,vascular,cardiacandcriticalcare.
ThepatientgroupusingOSCARisallexceptchildren.
ZEN-2090ProisamobiledigitalC-armdesignedtoprovidefluoroscopicandradiographicimagesofthepatientduringdiagnostic,surgicalandinterventionalprocedures.
Examplesofclinicalapplicationmayincludecholangiography,endoscopy,orthopedic,neurologic,stonelocalization,criticalcareandemergencyroomproceduresi.
e.
surgicalinterventionsneedingX-rayimagingand/orguidanceandinterventionsinsideandoutsidetheoperatingroom.
GeneratorHighFrequencyInverterHighFrequencyInverterMax.
outputpower4.
5kW2.
2kWX-raytubeStationarytubeStationarytubeLarge:1.
4/Small:0.
5Large:1.
5mm/Small:0.
5mmDetectorOSCARPrime(Flatpanel)OSCARClassic(Imageintensifier)ZEN-2090Pro(ImageIntensifier)OptionA:GFD-200(DualRay-Q)(ClearedunderK172180)Activeimagearea:260x256mmCentralResolution:4.
6lp/mmType:CMOSResolution:2600x2560Pixelsamplingresolution:14bitsPixelpitch:100μmMTF:56%DQE:59%Scintillator:CsIOptionA:E5830SD-P4A(ClearedunderK140041)Activeimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)E5830SD-P4AActiveimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)OptionB:VIVIX-D0909GActiveimagearea:229.
12x229.
12mmCentralResolution:2.
7lp/mmType:a-SiTFTResolution:2600x2560Pixelsamplingresolution:16bitPixelpitch:179μmMTF:45%DQE:45%Scintillator:CsIOptionB:TH9438QX(ClearedunderK140041).
Activeimagearea:9"(9/6/4.
5)Min.
CentralResolutionatthemonitor:-9"(23cm):2.
2lp/mm-6"(15cm):2.
8lp/mm-6"(15cm):3.
0lp/mmDQE:65%(typically)Fluoroscopy40-120kV/0.
2-6.
0mA40-110kV/0.
2-6.
0mAPulsedFluoroscopy0.
2-10.
0mA0.
2-6.
0mARadiography40-120kV/0.
6-200mAs40-110kV/0.
4-100mAsDimensionsSID:1000mmSID:960mmPanningRotation:±12.
5PanningRotation:±12.
5OrbitalRotation:155OrbitalRotation:120Vert.
Travel:500mmVert.
Travel:400mmHoriz.
Travel:200mmHoriz.
Travel:200mmTheproposedOSCARisbasedonthepredicatedevice,ZEN-2090Pro(K091918).
ThereareseveraldifferencebetweenOSCAR(Prime,Classic)andpredicatedevice(ZEN-2090Pro)istheoptionsofImageacquisitionparts.
OSCARPrimeistheflatpaneldetectorandpredicatedeviceisImageIntensifier.
Asmentionedinthecomparisontable,predicatedeviceDQEishigherthanOSCARPrime.
However,predicatedeviceDQEistheDQEoftheimageintensifieritself.
Ingeneral,whenimageintensifieriscombinedwithCCDcameratheDQEdecreases.
Inconclusion,theDQEofthecompletepredicatedeviceis51%.
SotheDQEoftheOSCARPrimeismoreeffectiveandsafetythanpredicatedevice.
Alsoflatpaneldetectortypehasexcellentimageuniformity,nogeometricdistortion,noveilingglareorvignetting,smallandthinphysicalsizeascomparedtotheImageIntensifiertype.
TheOSCARissubstantiallyequivalenttothepredicatedevice,ZEN-2090Pro(K091918).
8.
Safety,EMCandPerformancedatacomparisontoPredicateOSCARcomplieswithindustrystandardssuchasIEC60601-1Seriesand21CFR1020.
30,21CFR1020.
31and21CFR1020.
32tominimizeelectrical,mechanicalandradiationhazards.
-Electrical,mechanical,environmentalsafetyandperformancetestingaccordingtostandardIEC60601-1,IEC60601-1-3,IEC60601-1-6,IEC60601-2-28,IEC60601-2-43,IEC60601-2-54andIEC62366wereperformed.
-EMCtestingwasconductedinaccordancewithstandardIEC60601-1-2.
-OSCARmeetstheEPRCstandards(21CFR1020.
30,31,32).
-FDAguidance"guidanceforSSXIdevices",and"guidancefortheContentofPremarketSubmissionsforSoftwareContainedinMedicaldevices",wasperformedforOSCAR.
Thesoftwarefeatureshasbeenchangedcomparetopredicatedeviceasbelow:EnhanceImageCalibrationfunction.
DisplayexposuremodesshowsontheLCDtouchpanel.
AddAngiographyfunctions.
Changestothepredicatedevicesoftwareweretestedandtheydonotaffectthedevicesafetyandeffectiveness.
Also,thedevicesoftwareismoderatelevelofconcern.
Asaresults,alltestresultsweresatisfactoryandtheresultofbenchandclinicalevaluationindicatesthatthenewdeviceisassafeandeffectiveasthepredicatedevice.
9.
ConclusionInreferencetothecomparisoninformationprovidedinsubstantialequivalencechart,andthemostoffunctionsandelectronicfeaturesaresimilarwithpredicatedevice.
WebelievethattheOSCARissafeandeffectiveaspredicatedevice,andhasnonewindicationforuse.
Therefore,OSCARissubstantiallyequivalenttopredicatedevice.
- practicesoray花生壳相关文档
- 地址oray花生壳
- 配置oray花生壳
- oray花生壳花生壳怎么用 花生壳动态域名使用方法
国内云服务器 1核 2G 2M 15元/月 萤光云
标题【萤光云双十二 全场6折 15元/月 续费同价】今天站长给大家推荐一家国内云厂商的双十二活动。萤光云总部位于福建福州,其成立于2002 年。主打高防云服务器产品,主要提供福州、北京、上海 BGP 和香港 CN2 节点。萤光云的高防云服务器自带 50G 防御,适合高防建站、游戏高防等业务。这家厂商本次双十二算是性价比很高了。全线产品6折,上海 BGP 云服务器折扣更大 5.5 折(测试了一下是金...

sharktech:洛杉矶/丹佛/荷兰高防服务器;1G独享$70/10G共享$240/10G独享$800
sharktech怎么样?sharktech (鲨鱼机房)是一家成立于 2003 年的知名美国老牌主机商,又称鲨鱼机房或者SK 机房,一直主打高防系列产品,提供独立服务器租用业务和 VPS 主机,自营机房在美国洛杉矶、丹佛、芝加哥和荷兰阿姆斯特丹,所有产品均提供 DDoS 防护。不知道大家是否注意到sharktech的所有服务器的带宽价格全部跳楼跳水,降幅简直不忍直视了,还没有见过这么便宜的独立服...
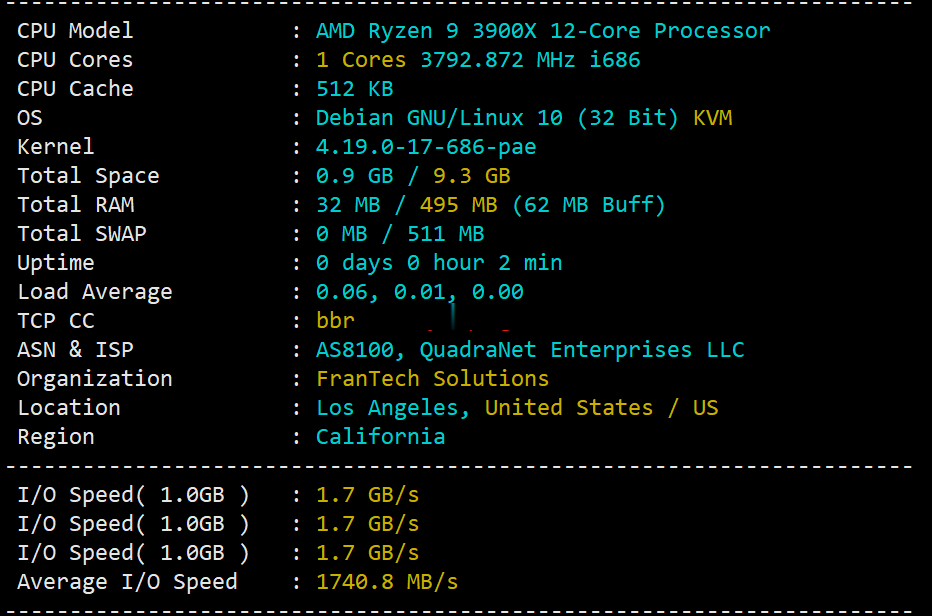
buyvm迈阿密机房VPS国内首发测评,高性能平台:AMD Ryzen 9 3900x+DDR4+NVMe+1Gbps带宽不限流量
buyvm的第四个数据中心上线了,位于美国东南沿海的迈阿密市。迈阿密的VPS依旧和buyvm其他机房的一样,KVM虚拟,Ryzen 9 3900x、DDR4、NVMe、1Gbps带宽、不限流量。目前还没有看见buyvm上架迈阿密的block storage,估计不久也会有的。 官方网站:https://my.frantech.ca/cart.php?gid=48 加密货币、信用卡、PayPal、...
oray花生壳为你推荐
-
摩拜超15分钟加钱摩拜单车不是按骑行时间收费吗,我怎么只要开锁就要支付一元(而且只骑十几分钟)小度商城小度怎么下载app?mathplayer西南交大网页上的 Mathplayer 安装了为什么还是用不了?mathplayer比较word,TeX,MathML中的数学公式处理方式的异同点,尽量详细哦,分数不是问题,谢谢哈,会加分的。mathplayerjavascript 如何判断document.body.innerHTML是否为空access数据库ACCESS数据库有什么用www.haole012.com012qq.com真的假的qq530.com求教:如何下载http://www.qq530.com/ 上的音乐www.7788k.comwww.6601txq.com.有没有这个网站www.zhiboba.com看NBA直播的网站哪个知道